Abstract
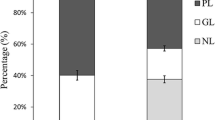
Phospholipids (PL) and glycolipids (GL) FA in the edible Rhodophyta Grateloupia turuturu, from Brittany, France, were investigated over four seasons. The major lipid class was GL in all seasons (around 45 %). More than 80 FA occurred in polar lipids, with chains from C12 to C26, identified as methyl esters and N-acyl pyrrolidides by gas chromatography–mass spectrometry (GC–MS). PUFA occurred at up to 47.1 % (summer) in PL, and up to 43.6 % (summer) in GL. The major PUFA were 20:5n-3 (12.2 % in PL and 29.0 % in GL) and 20:4n-6 (25.6 % in PL and 10.4 % in GL). The unusual 18:3n-7 acid was identified in PL up to 2.2 %. Several minor unsaturated FA were identified in PL and are previously unreported in seaweeds, namely 14-tricosenoic, 15-tetracosenoic, 5,11-octadecadienoic and 5,9-nonadecadienoic. Also unprecedented in seaweeds, ten 2-hydroxy and three 3-hydroxy FA occurred mainly in PL, 13.9 % in spring with the 3-hydroxyhexadecanoic acid as the major one (8.1 % winter). Three n-9 monounsaturated 2-hydroxy FA occurred in PL. The 2-hydroxy-15-tetracosenoic acid was characterized as the dimethyl disulfide adduct of its methyl ester. The 2-hydroxy-16-pentacosenoic and 2-hydroxy-17-hexacosenoic acids were identified by comparison of mass spectra and GC mobilities with those of the 2-hydroxy-15-tetracosenoic acid, and of other homogeneous FA series. These rare n-9 monounsaturated 2-hydroxy FA are unprecedented in seaweeds.




Similar content being viewed by others
Abbreviations
- amu:
-
Atomic mass unit
- ARA:
-
Arachidonic acid (20:4n-6)
- DMA:
-
Aldehyde dimethylacetals
- DMDS:
-
Dimethyl disulfide
- Dry wt:
-
Dry weight
- ECL:
-
Equivalent chain length
- EPA:
-
Icosapentaenoic acid (20:5n-3)
- FA:
-
Fatty acid(s)
- FAME:
-
Fatty acid methyl ester(s)
- GL:
-
Glycolipid(s)
- MUFA:
-
Monounsaturated fatty acid(s)
- NAP:
-
N-acylpyrrolidides
- GC–MS:
-
Gas chromatography–mass spectrometry
- PL:
-
Phospholipid(s)
- PUFA:
-
Polyunsaturated fatty acid(s)
- RT:
-
Retention time(s)
- SFA:
-
Saturated fatty acid(s)
References
Dawczynski C, Schubert R, Jahreis G (2007) Amino acids, fatty acids, and dietary fibre in edible seaweed products. Food Chem 103:891–899
Stengel DB, Connan S, Popper ZA (2011) Algal chemodiversity and bioactivity: sources of natural variability and implications for commercial application. Biotechnol Adv 29:483–501
Bocanegra A, Bastida S, Benedí J, Ródenas S, Sánchez-Muniz FJ (2009) Characteristics and nutritional and cardiovascular-health properties of seaweeds. J Med Food 12:236–258
Kumari P, Kumar M, Gupta V, Reddy CRK, Jha B (2010) Tropical marine macroalgae as potential sources of nutritionally important PUFAs. Food Chem 120:749–757
Khotimchenko SV (2005) Lipids from the marine alga Gracilaria verrucosa. Chem Nat Comp 41:285–288
Hotimchenko SV (2002) Fatty acid composition of algae from habitats with varying amounts of illumination. Russ J Mar Biol 28:218–220
Denis C, Morançais M, Li M, Deniaud E, Gaudin P, Wielgosz-Collin G, Barnathan G, Jaouen P, Fleurence J (2010) Study of the chemical composition of edible red macroalgae Grateloupia turuturu from Brittany (France). Food Chem 119:913–917
Kendel M, Couzinet-Mossion A, Viau M, Fleurence J, Barnathan G, Wielgosz-Collin G (2013) Seasonal composition of lipids, fatty acids and sterols in the edible red alga Grateloupia turuturu. J Appl Phycol 25:425–432. doi:10.1007/s10811-012-9876-3
Simon C, Gall EA, Deslandes E (2001) Expansion of the red alga Grateloupia doryphora along the coast of Brittany, France. Hydrobiologia 443:23–29
Pang SJ, Xiao T, Shan TF, Wang ZF, Gao SQ (2006) Evidences of the intertidal red alga Grateloupia turuturu in turning Vibrio parahaemolyticus into non-culturable state in the presence of light. Aquaculture 260:369–374
Plouguerné E, Hellio C, Deslandes E, Veron E, Stiger-Pouvreau V (2008) Anti-microfouling activities in extracts of two invasive algae: Grateloupia turuturu and Sargassum muticum. Bot Mar 51:202–208
Denis C, Wielgosz-Collin G, Bretéché A, Ruiz N, Rabesaotra V, Boury-Esnault N, Kornprobst JM, Barnathan G (2009) New 17-methyl-13-octadecenoic and 3,16-docosadienoic acids from the sponge Polymastia penicillus. Lipids 44:655–663
Quévrain E, Barnathan G, Meziane T, Domart-Coulon I, Rabesaotra V, Bourguet-Kondracki ML (2012) New 2-methyl-13-eicosenoic acid from the temperate calcisponge Leuconia johnstoni. Lipids 47:345–353
Araki S, Sakurai T, Oohusa T, Kayama M, Nisizawa K (1990) Content of arachidonic and eicosapentaenoic acids in polar lipids from Gracilaria (Gracilariales, Rhodophyta). Hydrobiologia 204(205):513–519
Pettitt TR, Jones AL, Harwood JL (1989) Lipids of the marine red algae, Chondrus crispus and Polysiphonia lanosa. Phytochemistry 28:399–405
Andersson BA (1978) Mass spectrometry of fatty acid pyrrolidides. Prog Chem Fats Lipids 16:279–308
Christie WW (consulted in June 2012) The Lipid library. http://www.lipidlibrary.co.uk Accessed on July 2012
Pond DW, Allen CE, Bell MV, Van Dover CL, Fallick AE, Dixon DR, Sargent JR (2002) Origins of long-chain polyunsaturated fatty acids in the hydrothermal vent worms Ridgea piscesae and Protis hydrothermica. Mar Ecol Prog Ser 225:219–226
Ratnayake WMN, Ackman RG (1979) Identification of novel octadecenoic fatty acids in the seaweed Cladophora rupestris through oxidative ozonolysis of the alcohols prepared from the acids. Lipids 14:580–584
Barnathan G (2009) Non-methylene-interrupted fatty acids from marine invertebrates: occurrence, characterization and biological properties. Biochimie 91:671–678
Barnathan G, Kornprobst JM, Doumenq P, Mirallès J (1996) New unsaturated long-chain fatty acids in the phospholipids from the Axinellida sponges Trikentrion loeve and Pseudaxinella cf. lunaecharta. Lipids 31:193–200
Carballeira NM, Cruz C (1998) 5,9-Nonadecadienoic acids in Malvaviscus arboreus and Allamanda cathartica. Phytochemistry 49:1253–1256
Christie WW, AOCS Lipid Library. Mass spectra of hydroxy fatty acids: Part A. 2- to 9- hydroxy acids. http://lipidlibrary.aocs.org/ms/ms23/index.htm Accessed on August 2012
Barnathan G, Kornprobst JM (1993) Sponge fatty acids, 5. Characterization of complete series of 2-hydroxy long-chain fatty acids in phospholipids of two Senegalese marine sponges from the family Suberitidae: Pseudosuberites sp. and Suberites massa. J Nat Prod 56:2104–2113
Carballeira NM, Emiliano A, Rodriguez J, Reyes ED (1992) Isolation and characterization of novel 2-hydroxy fatty acids from the phospholipids of the sponge Smenospongia aurea. Lipids 27:681–685
Barnathan G, Bourgougnon N, Kornprobst JM (1998) Methoxy fatty acids isolated from the red alga Schizymenia dubyi. Phytochemistry 5:761–765
Carballeira NM, Shalabi F, Reyes M (1994) New 2-hydroxy fatty acids in the Caribbean urchin Tripneustes esculentus. J Nat Prod 57:614–619
Imai H, Yamamoto K, Shibahara A, Miyatani S, Nakayama T (2000) Determining double-bond positions in monoenoic 2-hydroxy fatty acids of glucosylceramides by gas chromatography–mass spectrometry. Lipids 35:233–236
Dagorn F, Dumay J, Wielgosz-Collin G, Rabesaotra V, Viau M, Monniot C, Biard JF, Barnathan G (2010) Phospholipid distribution and phospholipid fatty acids of the tropical tunicates Eudistoma sp. and Leptoclinides uniorbis. Lipids 45:253–261
Fleurence J, Gutbier G, Mabeau S, Leray C (1994) Fatty acids from 11 marine macroalgae of the French Brittany coast. J Appl Phycol 6:527–532
Herbreteau F, Coiffard LJM, Derrien A, De Roeck-Holtzhauer Y (1997) The fatty acid composition of five species of macroalgae. Bot Mar 40:25–27
Mishra VK, Temelli F, Ooraikul B, Shacklock PF, Craigie JS (1993) Lipids of the red alga, Palmaria palmata. Bot Mar 36:169–174
McDermid KJ, Stuercke B (2003) Nutritional composition of edible Hawaiian seaweeds. J Appl Phycol 15:513–524
Nelson MM, Phleger CF, Nichols PD (2002) Seasonal lipid composition in macroalgae of the northeastern Pacific Ocean. Bot Mar 45:58–65
Khotimchenko SV, Yakovleva IM (2005) Lipid composition of the red alga Tichocarpus crinitus exposed to different levels of photon irradiance. Phytochemistry 66:73–79
Araki S, Sakurai T, Omata T, Kawaguchi A, Murata N (1986) Lipid and fatty acid composition in the red algae Porphyra yezoensis. Jpn J Phycol 34:94–100
Sanina NM, Goncharova SN, Kostetsky EY (2004) Fatty acid composition of individual polar lipid classes from marine macrophytes. Phytochemistry 65:721–730
Dembitsky VM, Pechenkina-Shubina EE, Rozentsvet OA (1991) Glycolipids and fatty acids of some seaweeds and marine grasses from the Black Sea. Phytochemistry 30:2279–2283
Khotimchenko SV (2003) The fatty acid composition of glycolipids of marine macrophytes. Russ J Mar Biol 29:126–128
Rubio-Rodríguez N, Beltrán S, Jaime I, de Diego SM, Sanz MT, Rovira J, Carballido JR (2010) Production of omega-3 polyunsaturated fatty acid concentrates: a review. Innovative Food Sci Emerg Technol 11:1–12
Ruxton CHS, Calder PC, Reed SC, Simpson MJA (2005) The impact of long-chain n-3 polyunsaturated fatty acids on human health. Nutrition Res Rev 18:113–129
Mozaffarian D, Wu JHY (2011) Omega-3 fatty acids and cardiovascular disease effects on risk factors, molecular pathways, and clinical events. J Amer Coll Cardiol 58:2047–2067
Calviello G, Serini S, Piccioni E (2007) n-3 Polyunsaturated fatty acids and the prevention of colorectal cancer: molecular mechanisms involved. Curr Med Chem 14:3059–3069
Matsumoto GI, Nagashima H (1984) Occurrence of 3-hydroxy acids in microalgae and cyanobacteria and their geochemical significance. Geochim Cosmochim Acta 48:1683–1687
Volkman JK, Barrett SM, Blackburn SI, Mansour MP, Sikes EL, Gelin F (1998) Microalgal biomarkers: a review of recent research developments. Org Geochem 29:1163–1179
Ratledge C, Wilkinson SG (1988) Microbial lipids, vol 1. Academic Press, Harcourt Brace Jovanovich, London
Alugupalli S, Portaels F, Larsson L (1994) Systematic study of the 3-hydroxy fatty acid composition of mycobacteria. J Bacteriol 176:2962–2969
Rezanka T, Sokolov MY, Viden I (1990) Unusual and very long-chain fatty acids in Desulfotomaculum, a sulfate-reducing bacterium. FEMS Microbiol Ecol 73:231–238
Sjögren J, Magnusson J, Broberg A, Schnürer J, Kenne L (2003) Antifungal 3-hydroxy fatty acids from Lactobacillus plantarum MiLAB 14. Appl Envir Microbiol 69:7554–7557
Kumar GS, Jagannadham MV, Ray MK (2002) Low-temperature-induced changes in composition and fluidity of lipopolysaccharides in the Antarctic psychrotrophic bacterium Pseudomonas syringae. J Bacteriol 184:6746–6749
Colombo ML, Risè P, Giavarini F, De Angelis L, Galli C, Bolis CL (2006) Macroalgae as sources of polyunsaturated fatty acids. Plant Foods Human Nutr 61:67–72
Cole JJ (1982) Interactions between bacteria and algae in aquatic ecosystems. Annu Rev Ecol Evol Syst 13:291–314
Zabeti N, Bonin P, Volkman JK, Guasco S, Rontani JF (2010) Fatty acid composition of bacterial strains associated with living cells of the haptophyte Emiliania huxleyi. Org Geochem 41:627–636
Nagan N, Zoeller RA (2001) Plasmalogens: biosynthesis and functions. Prog Lipid Res 40:199–229
Acknowledgments
The authors thank Dr Claire Denis and Mr Pierre Gaudin from University of Nantes, Laboratory Mer-Molécules-Santé (MMS), EA 2160, for their technical assistance, in collecting alga, measurement, and in data analysis. This work is part of Melha Kendel’s Ph.D. thesis, supported by a grant from the Conseil Général de la Loire Atlantique, France.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.









About this article
Cite this article
Kendel, M., Barnathan, G., Fleurence, J. et al. Non-methylene Interrupted and Hydroxy Fatty Acids in Polar Lipids of the Alga Grateloupia turuturu Over the Four Seasons. Lipids 48, 535–545 (2013). https://doi.org/10.1007/s11745-013-3783-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11745-013-3783-5