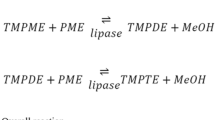Abstract
The objective of this study was to find the optimal parameters for lipase-catalyzed methanolysis of triricinolein to produce 1,2(2,3)-diricinolein. Four different immobilized lipases were tested, Candida antarctica type B (CALB), Rhizomucor miehei (RML), Pseudomonas cepacia (PCL), and Penicillium roquefortii (PRL). n-Hexane and diisopropyl ether (DIPE) were examined as reaction media at three different water activities (a w), 0.11, 0.53, and 0.97. The consumption of triricinolein and the formation of 1,2(2,3)-diricinolein, methyl ricinoleate, and ricinoleic acid were followed for up to 48 h. PRL gave the highest yield of 1,2(2,3)-diricinolein. Moreover, this lipase showed the highest specificity for the studied reaction, i.e., high selectivity for the reaction with triricinolein but low for 1,2(2,3)-diricinolein. Recoveries of 93 and 88% DAG were obtained using PRL in DIPE at a w of 0.11 and 0.53, respectively. Further, NMR studies showed that a higher purity of the 1,2(2,3)-isomer vs. the 1,3-isomer was achieved at higher a w (88% at a w=0.53), compared to lower a w (71% at a w=0.11). The DAG obtained was acylated by the DAG acyltransferase from Arabidopsis thaliana. Therefore, this enzymatic product is a useful enzyme substrate for lipid biosynthesis. Accordingly, the use of PRL in DIPE at a w 0.53 is considered optimal for the synthesis of 1,2(2,3)-diricinolein from triricinolein.
Similar content being viewed by others
Abbreviations
- AtDGAT:
-
cDNA coding DGAT cloned from Arabidopsis thaliana, Columbia
- a w :
-
water activity
- CALB:
-
Candida antarctica type B lipase
- DGAT:
-
DAG acyltransferase
- DIPE:
-
dusopropyl ether
- ESI-MS:
-
electrospray ionization MS
- NP-HPLC:
-
normal-phase HPLC
- PCL:
-
Pseudomonas cepacia lipase
- PRL:
-
Penicillium roquefortii lipase
- RML:
-
Rhizomucor miehei lipase
- RRO:
-
sn-1,2-diricinoleoyl-sn-3-oleoyl TAG
- RT-PCR:
-
reverse-transcriptase PCR
References
Lin, J.-T., Woodruff, C.L., Lagouche, O.J., McKeon, T.A., Stafford, A.E., Goodrich-Tanrikulu, M., Singleton, J.A., and Haney, C.A. (1998) Biosynthesis of Triacylglycerols Containing Ricinoleate in Castor Microsomes Using 1-Acyl-2-oleoyl-sn-glycero-3-phosphocholine as the Substrate of Oleoyl-12-hydroxylase, Lipids 33, 59–69.
Lin, J.-T., Turner, C., Liao, L.P., and McKeon, T.A. (2003) Identification and Quantification of the Molecular Species of Acylglycerols in Castor Oil by HPLC Using ELSD, J. Liq. Chromatogr. Rel. Technol. 26, 759–766.
Caupin, H.-J. (1997) Products from Castor Oil—Past, Present, and Future, in Lipid Technologies and Applications (Gunstone, F.D., and Padley, F.B., eds.), pp. 787–795, Marcel Dekker, New York.
Lord, J.M., Roberts, L.M., and Robertus, J.D. (1994) Ricin: Structure. Mode of Action, and Some Current Applications. FASEB J. 8, 201–208.
Nagao, T., Watanabe, H., Goto, N., Onizawa, K., Taguchi, H., Matsuo, N., Yasukawa, T., Tsushima, R., Shimasaki, H., and Itakura, H. (2000) Dietary Diacylglycerol Suppresses Accumulation of Body Fat Compared to Triacylglycerol in Men: A Double-Blind Controlled Trial, J. Nutr. 130, 792–797.
Lambert, D.M. (2000) Rationale and Applications of Lipids as Prodrug Carriers, Eur. J. Pharm. Sci. 11, S15-S27.
Fureby, A.M., Tian, L., Adlercreutz, P., and Mattiasson, B. (1997) Preparation of Diglycerides by Lipase-Catalyzed Alcoholysis of Triglycerides, Enzyme Microb. Technol. 20, 198–206.
Vogel, G., and Browse, J. (1995) Preparation of Radioactively Labeled Synthetic sn-1.2-Diacylglycerols for Studies of Lipid Metabolism, Anal. Biochem. 224, 61–67.
Gitlesen, T., Bauer, M., and Adlercreutz, P. (1997) Adsorption of Lipase on Polypropylene Powder, Biochim. Biophys. Acta 1345, 188–196.
Halling, P.J. (1992) Salt Hydrates for Water Activity Control with Biocatalysis in Organic Media, Biotechnol. Tech. 6, 271–276.
Urban, P., Werckreichhart, D., Teutsch, H.G., Durst, F., Regnier, S., Kazmaier, M., and Pompon, D. (1994) Characterization of Recombinant Plant Cinnamate 4-Hydroxylase Produced in Yeast—Kinetic and Spectral Properties of the Major Plant P450 of the Phenylpropanoid Pathway, Eur. J. Biochem. 222, 843–850.
Cases, S., Smith, S.J., Zheng, Y.W., Myers, H.M., Lear, S.R., Sande, E., Novak, S., Collins, C., Welch, C.B., Lusis, et al. (1998) Identification of a Gene Encoding an Acyl CoA:Diacylglycerol Acyltransferase, a Key Enzyme in Triacylglycerol Synthesis, Proc. Natl. Acad. Sci. USA 95, 13018–13023.
McKeon, T.A., Lin, J.T., Goodrich-Tanrikulu, M., and Stafford, A.E. (1997) Ricinoleate Biosynthesis in Castor Microsomes, Ind. Crops Prod. 6, 383–389.
Lin, J.-T., McKeon, T.A., Goodrich-Tanrikulu, M., and Stafford, A.E. (1996) Characterization of Oleoyl-12-hydroxylase in Castor Microsomes Using the Putative Substrate, 1-Acyl-2-oleoyl-sn-glycero-3-phosphocholine, Lipids 31, 571–577.
Lin, J.-T., Woodruff, C.L., and McKeon, T.A. (1997) Non-aqueous Reversed-Phase High-Performance Liquid Chromatography of Synthetic Triacylglycerols and Diacylglycerols, J. Chromatogr. A 782, 41–48.
Fureby, A.M., Virto, C., Adlercreutz, P., and Mattiasson, B. (1996) Acyl Group Migrations in 2-Monoolein, Biocatal. Biotransform. 14, 89–111.
Malcata, F.X., Reyes, H.R., Garcia, H.S., Hill, C.G., Jr., and Amundson, C.H. (1992) Kinetics and Mechanisms of Reactions Catalysed by Immobilized Lipases, Enzyme Microb. Technol. 14, 426–446.
Itabashi, Y., Kukis, A., Marai, L., and Takagi, T. (1990) HPLC Resolution of Diacylglycerol Moieties of Natural Triacylglycerols on a Chiral Phase Consisting of Bonded (R)-(+)-1-(1-naphthyl)Ethylamine, J. Lipid Res. 31, 1711–1717.
Turner, C., Persson, M., Mathiasson, L., Adlercreutz, P., and King, J.W. (2001) Lipase-Catalyzed Reactions in Organic and Supercritical Solvents: Application to Fat-Soluble Vitamin Determination in Milk Powder and Infant Formula, Enzyme Microb. Technol. 29, 111–121.
Adlercreutz, P. (2000) Biocatalysis in Non-conventional Media, in Applied Biocatalysis (Straathof, A.J.J., and Adlercreutz, P., eds.), pp. 295–316, Harwood Academic, Newark, NJ.
Svensson, I., Wehtje, E., Adlercreutz, P., and Mattiasson, B. (1994) Effects of Water Activity on Reaction Rates and Equilibrium Positions in Enzymatic Esterifications, Biotechnol. Bioeng. 44, 549–556.
Varian Associates (1962) NMR Spectra Catalog, Varian Associates, Palo Alto, CA.
Author information
Authors and Affiliations
About this article
Cite this article
Turner, C., He, X., Nguyen, T. et al. Lipase-catalyzed methanolysis of triricinolein in organic solvent to produce 1,2(2,3)-diricinolein. Lipids 38, 1197–1206 (2003). https://doi.org/10.1007/s11745-003-1179-5
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11745-003-1179-5