Abstract
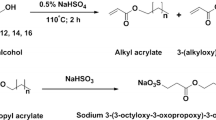
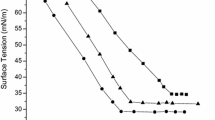
Maleic acid alkyl ester and N-alkyl maleamic acid monomers (RnMa and RnMaAm; n is alkyl chain length; n=6, 8, 10, 12, 14) were synthesized by the reaction of maleic anhydride with alkyl alcohol or alkylamine. The telomerization of RnMa or RnMaAm in the presence of alkanethiol as a chain transfer agent gave telomer-type anionic surfactants (xRnMa, xRnMaAm; x is total average number of alkyl chains; x=2.8–3.3) having multialkyl chains and multicarboxylate groups. Their surface-active properties were investigated by several techniques such as surface tension, foaming property, and emulsification power measurements. Critical micelle concentrations (CMC) of xRnMa were 1/110–1/14 of those of RnMa with the same alkyl chain length. xRnMa and xRnMaAm gave higher efficiencies in lowering the surface tension than RnMa and RnMaAm in aqueous solutions. In particular, the surface tension of 3.2R12MaAm was 24.4 mN m−1 at the CMC. Foaming abilities and foam stabilities of xRnMa and xRnMaAm were higher than those of RnMa and RnMaAm. The addition of 300 ppm of Ca2+ to the aqueous solutions rendered the telomers less surface active. Shaking the aqueous solutions of telomers with toluene emulsified them. The highly stable oil-in-water type emulsion was formed by using 3.0R10MaAm and 3.2R12MaAm, and the degree of emulsification was kept at a level of about 80% after 60 min of standing. Thus, telomer-type surfactants showed excellent surface activities that were superior to the corresponding monomers as well as to conventional surfactants. The relationship between alkyl chain length of the telomers and the properties of surface tension, foaming, and emulsification was unclear.
Similar content being viewed by others
References
Devinsky, F., L. Masarova, and I. Lacko, Surface Activity and Micelle Formation of Some New Bisquaternary Ammonium Salts, J. Colloid Interface Sci. 105:235 (1985).
Menger, F.M., and C.A. Littau, Gemini Surfactants: Synthesis and Properties. J. Am. Chem. Soc. 113:1451 (1991).
Rosen, M.J., Geminis. A New Generation of Surfactants, CHEMTECH 23:30 (1993).
Zana, R., H. Levy, D. Papoutsi, and G. Beinert, Micellization of Two Triquaternary Ammonium Surfactants in Aqueous Solution, Langmuir 11:3694 (1995).
Esumi, K., K. Taguma, and Y. Koide, Aqueous Properties of Multichain Quaternary Cationic Surfactants, Langmuir 12:4039 (1996).
Onitsuka, E., T. Yoshimura, Y. Koide, H. Shosenji, and K. Esumi, Preparation and Surface-Active Properties of Complexane-type Trimeric Surfactants from Tris(2-aminoethyl)amine, J. Oleo Sci. 50:159 (2001).
Strauss, U.P., and E.G. Jackson, Polysoaps. 1. Viscosity and Solubilization Studies on an n-Dodecyl Bromide Addition Compound of Poly-2-vinylpyridine. J. Polymer Sci. 6:649 (1951).
Black, W., in Recent Progress in Surface Science: Surface-Active Substances, edited by J.F. Danielli, K.G.A. Pankhurst, and A.C. Riddiford, Academic Press, New York, 1964, Vol. 1, p. 268.
Piirma, I., Polymeric Surfactants, Marcel Dekker, New York, 1992.
Hanford, W.E., D.E. Sargent, and H. Gilman, Organic Chemistry, John Wiley & Sons, New York, 1953, Vol. 4, p. 52.
Yamada, K., and Y. Koide, Telomer Type Surfactants. J. Jpn. Oil Chem. Soc. (J. Oleo Sci.) 30:2 (1981).
Esumi, K., H. Mizutani, K. Shoji, M. Miyazaki, K. Torigoe, T. Yoshimura, Y. Koide, and H. Shosenji, Physicochemical Propeerties of 2-Vinylpyridine Telomers Possessing Multihydrocarbon Chains in Aqueous Solution and at the Silica/Aqueous Solution Interface, J. Colloid Interface Sci. 220:170 (1999).
Esumi, K., A. Kameo, A. Suzuki, K. Torigoe, T. Yoshimura, Y. Koide, and H. Shosenji, Preparation of Gold Nanoparticles Using 2-Vinylpyridine Telomers Possessing Multi-Hydrocarbon Chains as Stabilizer, Colloids Surf. A 176:233 (2001).
Yoshimura, T., Y. Koide, H. Shosenji, and K. Esumi, Preparation of Cotelomers of Acrylic Acid and n-Hexyl, 2-Ethylhexyl or n-Dodecyl Acrylate and Their Properties as Multi-alkylated Surfactants, J. Jpn. Oil chem. Soc. (J. Oleo Sci.) 49:801 (2000).
Yoshimura, T., R. Tokunaga, H. Shosenji, and K. Esumi, Preparation and Surface Activities of Zwitterionic Surfactants Derived from Cotelomers of 4-Vinylpyridine and Methyl Acrylate, J. Oleo Sci. 50:467 (2001).
Flett, L.H., and W.H. Gardner, Maleic Anhydride Derivatives, John Wiley & Sons, New York, 1952.
Ackermann, R., D. Naegele, and H. Ringsdorf, Polyreactions in Oriented Media. 4. Photoreactions of Fumaric and Maleic Acid Derivatives in Multilayers, Makromol. Chem. 175:699 (1974).
Yoshimura, T., Y. Koide, H. Shosenji, and K. Esumi, Preparation and Surface-Active Properties of N-Alkyl Maleamic Acid Telomer Type Surfactants Having Several Hydrocarbon Chains, J. Oleo Sci. 51:229 (2002).
Rosen, M.J., Surfactants and Interfacial Phenomena, 2nd edn., John Wiley & Sons, New York, 1989, pp. 65, y0, 120, 276.
Nihon-Yukagaku-Kyokai, Yushi-Kagaku-Binran, Maruzen, Tokyo, 1990, p. 480.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Yoshimura, T., Koide, Y., Shosenji, H. et al. Preparation and surface-active properties of telomer-type anionic surfactants from maleic anhydride. J Surfact Deterg 5, 257–262 (2002). https://doi.org/10.1007/s11743-002-0225-x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11743-002-0225-x