Abstract
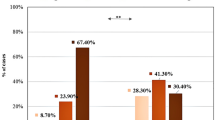
Many studies have pointed out that inflammation plays a pivotal role in pathophysiology of acute coronary syndromes (ACS) because several inflammatory molecules impair the endothelial functions in the coronary circulation and promote atherothrombotic events. Recently, many clinical/experimental evidences indicate that elevated plasma levels of uric acid (UA) might be considered a risk factor for developing ACS. It has been reported that elevated UA doses impair physiologic functions of endothelial cells, shifting them toward a pro atherothrombotic phenotype. In the present manuscript, we investigated the relationship between UA plasma levels, inflammatory burden, and extension of coronary atherosclerotic disease in patients with ACS. Patients with a clinical presentation of ACS (ST-elevated and non-ST-elevated myocardial infarction) admitted to the Vanvitelli Catheterization Laboratory at Monaldi Hospital in 2019, before the COVID-19 pandemia, were retrospectively analyzed. Biochemical profile, type of ACS presentation, as well as extension of coronary atherosclerosis were assessed. A total of 132 ACS patients were included in the analysis, and grouped into 3 tertiles according to the UA values (UA < 4.72 mg/dl, UA between 4.72 and 6.15 mg/dl, and UA > 6.15 mg/dl). Patients with UA plasma levels ≥ 6.15 mg/dL showed higher levels of C-reactive protein (mean of 5.1 mg/dL) as compared to patients with lower UA plasma levels. Moreover, the former group of patients showed higher levels of cardiac troponin and CPK, and presented more often with multivessel disease and complex coronary stenosis (type C of Ellis classification). Even though monocentric and with limited sample size, the present study shows that plasma levels of UA and hs-CRP are elevated in ACS patients and are associated with a more severe coronary disease, suggesting a potential role of UA in the pathophysiology of acute coronary events.

Similar content being viewed by others
References
Visseren FLJ, Mach F, Smulders YM, Carballo D, Koskinas KC, Back M, Benetos A, Biffi A, Boavida JM, Capodanno D et al (2021) 2021 ESC guidelines on cardiovascular disease prevention in clinical practice. Eur Heart J 42:3227–3337. https://doi.org/10.1093/eurheartj/ehab484
Li L, Zhang Y, Zeng C (2020) Update on the epidemiology, genetics, and therapeutic options of hyperuricemia. Am J Transl Res 12:3167–3181
Yu W, Cheng JD (2020) Uric acid and cardiovascular disease: an update from molecular mechanism to clinical perspective. Front Pharmacol 11:582680. https://doi.org/10.3389/fphar.2020.582680
Tian X, Chen S, Zhang Y, Zhang X, Xu Q, Wang P, Wu S, Luo Y, Wang A (2023) Serum uric acid variation and the risk of cardiovascular disease: a prospective cohort study. Eur J Intern Med 112:37–44. https://doi.org/10.1016/j.ejim.2023.02.001
Kanbay M, Afsar B, Covic A (2011) Uric acid as a cardiometabolic risk factor: to be or not to be. Contrib Nephrol 171:62–67. https://doi.org/10.1159/000327160
Culleton BF, Larson MG, Kannel WB, Levy D (1999) Serum uric acid and risk for cardiovascular disease and death: the Framingham heart study. Ann Intern Med 131:7–13. https://doi.org/10.7326/0003-4819-131-1-199907060-00003
Kuwabara M, Hisatome I, Niwa K, Hara S, Roncal-Jimenez CA, Bjornstad P, Nakagawa T, Andres-Hernando A, Sato Y, Jensen T et al (2018) Uric acid is a strong risk marker for developing hypertension from prehypertension: a 5-year japanese cohort study. Hypertension 71:78–86. https://doi.org/10.1161/HYPERTENSIONAHA.117.10370
Kuwabara M, Kuwabara R, Hisatome I, Niwa K, Roncal-Jimenez CA, Bjornstad P, Andres-Hernando A, Sato Y, Jensen T, Garcia G et al (2017) “Metabolically healthy” obesity and hyperuricemia increase risk for hypertension and diabetes: 5-year japanese cohort study. Obesity 25:1997–2008. https://doi.org/10.1002/oby.22000
Kuwabara M, Niwa K, Hisatome I, Nakagawa T, Roncal-Jimenez CA, Andres-Hernando A, Bjornstad P, Jensen T, Sato Y, Milagres T et al (2017) asymptomatic hyperuricemia without comorbidities predicts cardiometabolic diseases: 5-year japanese cohort study. Hypertension 69:1036–1044. https://doi.org/10.1161/HYPERTENSIONAHA.116.08998
Kuwabara M, Niwa K, Nishihara S, Nishi Y, Takahashi O, Kario K, Yamamoto K, Yamashita T, Hisatome I (2017) Hyperuricemia is an independent competing risk factor for atrial fibrillation. Int J Cardiol 231:137–142. https://doi.org/10.1016/j.ijcard.2016.11.268
Huang H, Huang B, Li Y, Huang Y, Li J, Yao H, Jing X, Chen J, Wang J (2014) Uric acid and risk of heart failure: a systematic review and meta-analysis. Eur J Heart Fail 16:15–24. https://doi.org/10.1093/eurjhf/hft132
Kanbay M, Segal M, Afsar B, Kang DH, Rodriguez-Iturbe B, Johnson RJ (2013) The role of uric acid in the pathogenesis of human cardiovascular disease. Heart 99:759–766. https://doi.org/10.1136/heartjnl-2012-302535
Katsiki N, Kouvari M, Panagiotakos DB, Borghi C, Chrysohoou C, Mikhailidis DP, Pitsavos C (2021) The association between serum uric acid levels and 10-year cardiovascular disease incidence: results from the ATTICA prospective study. Rev Cardiovasc Med 22:991–1001. https://doi.org/10.31083/j.rcm2203108
Browne LD, Jaouimaa FZ, Walsh C, Perez-Ruiz F, Richette P, Burke K, Stack AG (2021) Serum uric acid and mortality thresholds among men and women in the Irish health system: a cohort study. Eur J Intern Med 84:46–55. https://doi.org/10.1016/j.ejim.2020.10.001
Rebora P, Centola M, Morici N, Sacco A, Occhino G, Viola G, Oreglia J, Castini D, Persampieri S, Sabatelli L et al (2022) Uric acid associated with acute heart failure presentation in acute coronary syndrome patients. Eur J Intern Med 99:30–37. https://doi.org/10.1016/j.ejim.2022.01.018
Farquharson CA, Butler R, Hill A, Belch JJ, Struthers AD (2002) Allopurinol improves endothelial dysfunction in chronic heart failure. Circulation 106:221–226. https://doi.org/10.1161/01.cir.0000022140.61460.1d
Rekhraj S, Gandy SJ, Szwejkowski BR, Nadir MA, Noman A, Houston JG, Lang CC, George J, Struthers AD (2013) High-dose allopurinol reduces left ventricular mass in patients with ischemic heart disease. J Am Coll Cardiol 61:926–932. https://doi.org/10.1016/j.jacc.2012.09.066
Szwejkowski BR, Gandy SJ, Rekhraj S, Houston JG, Lang CC, Morris AD, George J, Struthers AD (2013) Allopurinol reduces left ventricular mass in patients with type 2 diabetes and left ventricular hypertrophy. J Am Coll Cardiol 62:2284–2293. https://doi.org/10.1016/j.jacc.2013.07.074
Lu J, Sun M, Wu X, Yuan X, Liu Z, Qu X, Ji X, Merriman TR, Li C (2019) Urate-lowering therapy alleviates atherosclerosis inflammatory response factors and neointimal lesions in a mouse model of induced carotid atherosclerosis. FEBS J 286:1346–1359. https://doi.org/10.1111/febs.14768
Xiao J, Zhang XL, Fu C, Han R, Chen W, Lu Y, Ye Z (2015) Soluble uric acid increases NALP3 inflammasome and interleukin-1beta expression in human primary renal proximal tubule epithelial cells through the Toll-like receptor 4-mediated pathway. Int J Mol Med 35:1347–1354. https://doi.org/10.3892/ijmm.2015.2148
Li Z, Shen Y, Chen Y, Zhang G, Cheng J, Wang W (2018) High uric acid inhibits cardiomyocyte viability through the ERK/P38 pathway via oxidative stress. Cell Physiol Biochem 45:1156–1164. https://doi.org/10.1159/000487356
Zhi L, Yuzhang Z, Tianliang H, Hisatome I, Yamamoto T, Jidong C (2016) High uric acid induces insulin resistance in cardiomyocytes in vitro and in vivo. PloS one 11:e0147737. https://doi.org/10.1371/journal.pone.0147737
Maruhashi T, Nakashima A, Soga J, Fujimura N, Idei N, Mikami S, Iwamoto Y, Kajikawa M, Matsumoto T, Hidaka T et al (2013) Hyperuricemia is independently associated with endothelial dysfunction in postmenopausal women but not in premenopausal women. BMJ open 3:e003659. https://doi.org/10.1136/bmjopen-2013-003659
Yan M, Chen K, He L, Li S, Huang D, Li J (2018) Uric acid induces cardiomyocyte apoptosis via activation of calpain-1 and endoplasmic reticulum stress. Cell Physiol Biochem 45:2122–2135. https://doi.org/10.1159/000488048
Cimmino G, Conte S, Marra L, Morello A, Morello M, De Rosa G, Pepe M, Sugralyev A, Golino P, Cirillo P (2022) Uric Acid induces a pro-atherothrombotic phenotype in human endothelial cells by imbalancing TF/TFPI pathway. Thromb Haemost. https://doi.org/10.1055/a-1947-7716
Johnson RJ, Sanchez Lozada LG, Lanaspa MA, Piani F, Borghi C (2023) Uric acid and chronic kidney disease: still more to do. Kidney Int Rep 8:229–239. https://doi.org/10.1016/j.ekir.2022.11.016
Maiuolo J, Oppedisano F, Gratteri S, Muscoli C, Mollace V (2016) Regulation of uric acid metabolism and excretion. Int J Cardiol 213:8–14. https://doi.org/10.1016/j.ijcard.2015.08.109
Sakata K, Hashimoto T, Ueshima H, Okayama A, Group NDR (2001) Absence of an association between serum uric acid and mortality from cardiovascular disease: NIPPON DATA 80, 1980–1994. National integrated projects for prospective observation of non-communicable diseases and its trend in the aged. Eur J Epidemiol 17:461–468. https://doi.org/10.1023/a:1013735717961
Perlstein TS, Gumieniak O, Williams GH, Sparrow D, Vokonas PS, Gaziano M, Weiss ST, Litonjua AA (2006) Uric acid and the development of hypertension: the normative aging study. Hypertension 48:1031–1036. https://doi.org/10.1161/01.HYP.0000248752.08807.4c
Borghi C, Agnoletti D, Cicero AFG, Lurbe E, Virdis A (2022) Uric acid and hypertension: a review of evidence and future perspectives for the management of cardiovascular risk. Hypertension 79:1927–1936. https://doi.org/10.1161/HYPERTENSIONAHA.122.17956
Sanchez-Lozada LG, Rodriguez-Iturbe B, Kelley EE, Nakagawa T, Madero M, Feig DI, Borghi C, Piani F, Cara-Fuentes G, Bjornstad P et al (2020) Uric acid and hypertension: an update with recommendations. Am J Hypertens 33:583–594. https://doi.org/10.1093/ajh/hpaa044
Katsiki N, Dimitriadis GD, Mikhailidis DP (2021) Serum uric acid and diabetes: from pathophysiology to cardiovascular disease. Curr Pharm Des 27:1941–1951. https://doi.org/10.2174/1381612827666210104124320
Afsar B, Sag AA, Oztosun C, Kuwabara M, Cozzolino M, Covic A, Kanbay M (2019) The role of uric acid in mineral bone disorders in chronic kidney disease. J Nephrol 32:709–717. https://doi.org/10.1007/s40620-019-00615-0
Jensen T, Niwa K, Hisatome I, Kanbay M, Andres-Hernando A, Roncal-Jimenez CA, Sato Y, Garcia G, Ohno M, Lanaspa MA et al (2018) Increased serum uric acid over five years is a risk factor for developing fatty liver. Sci Rep 8:11735. https://doi.org/10.1038/s41598-018-30267-2
Mengozzi A, Virdis A (2022) Uric acid and cardiovascular risk stratification in the acute coronary syndromes: a friend we should mind. Eur J Intern Med 99:22–23. https://doi.org/10.1016/j.ejim.2022.02.017
Padda J, Khalid K, Padda S, Boddeti NL, Malhi BS, Nepal R, Cooper AC, Jean-Charles G (2021) Hyperuricemia and its association with ischemic stroke. Cureus 13:e18172. https://doi.org/10.7759/cureus.18172
Li F, Chen S, Qiu X, Wu J, Tan M, Wang M (2021) Serum uric acid levels and metabolic indices in an obese population: a cross-sectional study. Diabetes Metab Syndr Obes Targets Ther 14:627–635. https://doi.org/10.2147/DMSO.S286299
Cimmino G, Loffredo FS, Morello A, D’Elia S, De Palma R, Cirillo P, Golino P (2017) Immune-inflammatory activation in acute coronary syndromes: a look into the heart of unstable coronary plaque. Curr Cardiol Rev 13:110–117. https://doi.org/10.2174/1573403X12666161014093812
Forte L, Cimmino G, Loffredo F, De Palma R, Abbate G, Calabro P, Ingrosso D, Galletti P, Carangio C, Casillo B et al (2011) C-reactive protein is released in the coronary circulation and causes endothelial dysfunction in patients with acute coronary syndromes. Int J Cardiol 152:7–12. https://doi.org/10.1016/j.ijcard.2011.05.062
Danenberg HD, Kantak N, Grad E, Swaminathan RV, Lotan C, Edelman ER (2007) C-reactive protein promotes monocyte-platelet aggregation: an additional link to the inflammatory-thrombotic intricacy. Eur J Haematol 78:246–252. https://doi.org/10.1111/j.1600-0609.2006.00808.x
Cirillo P, Golino P, Calabro P, Cali G, Ragni M, De Rosa S, Cimmino G, Pacileo M, De Palma R, Forte L et al (2005) C-reactive protein induces tissue factor expression and promotes smooth muscle and endothelial cell proliferation. Cardiovasc Res 68:47–55. https://doi.org/10.1016/j.cardiores.2005.05.010
Sattar N, Murray HM, McConnachie A, Blauw GJ, Bollen EL, Buckley BM, Cobbe SM, Ford I, Gaw A, Hyland M et al (2007) C-reactive protein and prediction of coronary heart disease and global vascular events in the Prospective Study of Pravastatin in the Elderly at Risk (PROSPER). Circulation 115:981–989. https://doi.org/10.1161/CIRCULATIONAHA.106.643114
de Carvalho Vidigal F, de Lima LER, Rosado GP, de Ribeiro RDCL, Franceschini SDCC (2014) Serum uric acid can predict higher C-reactive protein levels in apparently healthy men. Nutricion Hospitalaria 29:935–940. https://doi.org/10.3305/nh.2014.29.4.7094
Kang DH, Park SK, Lee IK, Johnson RJ (2005) Uric acid-induced C-reactive protein expression: implication on cell proliferation and nitric oxide production of human vascular cells. J Am Soc Nephrol 16:3553–3562. https://doi.org/10.1681/ASN.2005050572
Ruggiero C, Cherubini A, Ble A, Bos AJ, Maggio M, Dixit VD, Lauretani F, Bandinelli S, Senin U, Ferrucci L (2006) Uric acid and inflammatory markers. Eur Heart J 27:1174–1181. https://doi.org/10.1093/eurheartj/ehi879
Pawlowski T, Prati F, Capodanno D, Tamburino C, Gil RJ (2012) Initial troponin level may predict thrombus burden in patients with acute coronary syndrome. Optical coherence tomography study. Kardiologia Polska 70:457–462
Wasiak J, Law J, Watson P, Spinks A (2012) Percutaneous transluminal rotational atherectomy for coronary artery disease. Cochrane Database Syst Rev 12:CD00334. https://doi.org/10.1002/14651858.CD003334.pub2
Zhang D, Zhang R, Wang N, Lin L, Yu B (2018) Correlation of serum uric acid levels with nonculprit plaque instability in patients with acute coronary syndromes: a 3-vessel optical coherence tomography study. Biomed Res Int 2018:7919165. https://doi.org/10.1155/2018/7919165
Chu X, Lu Y, Mei M, Peng P, Zhao Y, Fu G, Qiu F, Jin C (2022) Correlation between serum uric acid levels and coronary plaque characteristics on optical coherence tomography. Int Heart J 63:806–813. https://doi.org/10.1536/ihj.21-826
He C, Lin P, Liu W, Fang K (2019) Prognostic value of hyperuricemia in patients with acute coronary syndrome: a meta-analysis. Eur J Clin Investig 49:e13074. https://doi.org/10.1111/eci.13074
Zu W, Li CC, Wang XY, Li QS, Liu B (2022) Association of uric acid levels with cardiac syndrome X: a meta-analysis. Front Physiol 13:976190. https://doi.org/10.3389/fphys.2022.976190
Funding
This research was funded by V:ALERE program 2019—OMICs-ACS, from University of Campania Luigi Vanvitelli.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Human and animal rights statement
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the local Ethics Committee for retrospective evaluation of medical records.
Informed consent
Informed consent was obtained from all subjects involved in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cimmino, G., Gallinoro, E., di Serafino, L. et al. Uric acid plasma levels are associated with C-reactive protein concentrations and the extent of coronary artery lesions in patients with acute coronary syndromes. Intern Emerg Med 18, 1751–1757 (2023). https://doi.org/10.1007/s11739-023-03360-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11739-023-03360-2