Abstract
Background
Metabolic bone disease is frequently found in patients with coeliac disease (CD). Despite its high prevalence, international guidelines are partially discordant about its management due to the lack of long-term data.
Methods
We retrospectively evaluated a large dataset of prospectively collected data of CD patients assessing the variation of DXA parameters and estimated fracture risk according to the FRAX® score in a 10-year follow-up. Incident fractures are reported, and the predictive ability of the FRAX® score is verified.
Results
We identified 107 patients with low bone density (BMD) at the diagnosis of CD and a 10-year follow-up. After improving at the first follow-up, T-scores slowly reduced over time but with no clinically relevant differences between the first and last examination (lumbar spine: from − 2.07 to − 2.07, p = 1.000; femoral neck: from − 1.37 to − 1.55, p = 0.006). Patients with osteoporosis at the index measurement had more marked fluctuations than those with osteopenia; the latter group also showed minimal modifications of the FRAX® score over time. Six incident major fragility fractures occurred, with a good predictive ability of the FRAX® (AUC 0.826).
Conclusion
Adult CD patients with osteopenia and no risk factors had substantially stable DXA parameters and fracture risk during a 10-year follow-up. A dilated interval between follow-up DXA for these patients could be considered to reduce diagnosis-related time and costs, maintaining a 2-year interval for patients with osteoporosis or risk factors.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
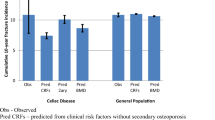
Osteoporosis can be found in 26–40% of patients at the diagnosis of coeliac disease (CD) [1,2,3], resulting in an excess risk of fractures of 320–480/100000 person-years in adults [4,5,6,7]. Treatment with a gluten-free diet (GFD) improves bone mineral density after 1–2 years of adherence [8,9,10,11], but longer term data are lacking. The effects of the GFD on reducing the risk of fragility fractures are still debated, as population-based studies found a similar incidence ratio for fractures before and after CD diagnosis [6]. Thus, the protective role of GFD on subsequent fracture risk may not be universal [12]
As a consequence of these uncertainties and lack of long-term data, the main international guidelines provide partially discordant recommendations. The American College of Gastroenterology (ACG) guidelines recommend a dual-energy X-ray absorptiometry (DXA) at the diagnosis of CD and every 2–3 years [12]. The European Society for the Study of Coeliac Disease (ESsCD) also recommends DXA at the diagnosis of CD, but limits follow-up DXAs to patients with abnormal index values [13]. Conversely, the British Society of Gastroenterology (BSG) recommend a DXA evaluation in patients with risk factors for osteoporosis or if > 55 year-old, repeating the investigation only in patients with low bone density on index measurement [14]. Finally, the National Institute for Health and Care Excellence (NICE) recommend a preliminary risk assessment through clinical scores, prescribing DXA only over certain risk thresholds and monitoring only patients receiving treatments [15].
Answering the call for research favoring an optimized management of bone disease in CD [13] and trying to provide data helping in standardizing the current recommendations, we designed a study gathering information starting from the index DXA and extending for 10 years.
Methods
Clinical setting
We retrospectively analyzed the medical records of patients consecutively diagnosed with CD in our outpatient clinic (IRCCS Azienda Ospedaliero-Universitaria di Bologna, Bologna, Italy) between January 2004 and December 2020. The database was locked in February 2022. The target population was represented by CD patients who had low BMD at the index DXA examination and received regular follow-up DXAs every 2–3 years as part of everyday clinical practice, according to the European recommendations.
Overtime variations of the main DXA parameters and risk of fracture estimated according to the Fracture Risk Assessment Tool (FRAX®) score were the main aim. The secondary aim was to assess the actual rate of major fragility fractures and the predictive ability of the FRAX score in identifying patients who had incident fractures. FRAX® is a tool based on individual patient models that integrate the risks associated with both clinical risk factors and bone mineral density [16]. The output is a 10-year probability of a major osteoporotic fracture. The calculated probability can be used to guide treatment strategies. A > 20% probability of major osteoporotic fractures is widely recognized as a threshold to define high-risk patients which should be considered for antiresorptive treatments. More recently, a 10% probability threshold has been proposed to discriminate low-risk and moderate-risk (10–20% probability) patients [17]. This proposal reflected the fact that more osteoporotic fractures occur in the moderate-risk group than the high-risk group as an absolute value (because there are more individuals in the moderate-risk group), even though the individual risk of fracture remains higher in the high-risk group [18]. Also, according to a recent consensus [19], a 10% risk in a relatively young population is high enough to consider pharmacological treatment in selected cases. On the contrary, some agencies are known to be reluctant to reimburse treatments on the basis of fracture probability at younger ages when the 10-year probability of a major osteoporotic fracture is less than 10% [19]. For these reasons, a 10% risk threshold was set as a cut-off for the subgroup analyses in this study, which included a relatively young population.
Inclusion and exclusion criteria
Coeliac disease was diagnosed by serologic testing of coeliac-specific antibodies (anti- transglutaminase IgA, or IgG in patients with IgA deficiency) and confirmed by duodenal mucosal biopsies [12]. Classical (i.e., with signs and symptoms of malabsorption) and non-classical CD were defined according to the Oslo criteria [20]. We included patients with the following additional criteria: (1) availability of an index DXA, performed within 12 months from the diagnosis of CD; (2) low BMD, defined as abnormal T-score values (< − 1.0) at the index DXA; (3) a minimum 10-year follow-up after the diagnosis of CD; (4) follow-up DXAs regularly performed at 24–36 months intervals after the index examination.
Patients were excluded in case of: (1) index or follow-up DXAs performed outside of the timeframe; (2) incomplete medical records.
The following data were available for all patients: age at the diagnosis, sex, weight, height, family history of CD, clinical presentation (classical vs non-classical), adherence to the GFD (defined as no reported intentional or accidental gluten ingestion in the last 6 months, absence of CD‐related symptoms, and negative anti‐transglutaminase IgA antibodies) [21]. Medical records systematically included information about: family history for fragility fractures, personal history regarding smoke and alcohol habits, bone fractures, concurrent illnesses, and ongoing medications.
Clinical, laboratory, and DXA evaluations
Clinical evaluations were scheduled according to the Italian Protocol for the Diagnosis and Follow-Up of Coeliac Disease (a first follow-up visit 6 months after the start of the GFD, then every 18–24 months) [22].
Blood tests were repeated before each evaluation, including blood cell count, ferritin, TSH, 25-OH vitamin D, total calcium, and anti-tissue transglutaminase IgA.
An index DXA exploring the lumbar spine and hip was prescribed at the diagnosis of CD. Differences between the bone mineral density of each patient and the young adult reference (T-score) or the same age and sex reference (Z-score) were expressed as standard deviations. If the T-score was below the normal range (i.e., < − 1.0) in at least one site, follow-up DXAs were prescribed every 24–36 months. On the contrary, no further DXAs were scheduled for patients with normal values unless new risk factors appeared [12, 13, 22]. Osteopenia (T-score between − 1.0 and − 2.5), osteoporosis (T-score ≤ − 2.5), and BMD below/within the expected range for age (Z-score ≤ or > 2.0, respectively) were defined according to the classification of the World Health Organization and the International Society for Clinical Densitometry (ISCD) [23, 24] DXA scans were performed and analyzed following manufacturer recommendations [25]. Patients were strongly suggested to perform follow-up DXAs with the same machine as the one used at their index examination to minimize the risk of non-comparable results. BMD values were expressed as either T-scores or Z-scores to allow BMD comparison over time. Of note, BMD values derived from different DXA machines are still well correlated [26].
Risk of fracture
Fracture risk was retrospectively calculated at each timepoint using the Fracture Risk Assessment Tool (FRAX.®) for the Italian population (available at https://www.sheffield.ac.uk/FRAX/tool.aspx?lang=en) [16].
For patients younger than 40-year old, the calculator automatically inserted 40 years. Coeliac disease was considered a cause of secondary osteoporosis in calculating the FRAX score to maximize its predictive abilities [27].
Fractures
During each clinical evaluation, patients were asked whether they had experienced a bone fracture. Medical records were also checked to reduce the risk of unreported events. According to the definitions of the World Health Organization, a fragility fracture was recorded if it resulted from mechanical forces that would not ordinarily result in a fracture. Fractures of the spine (clinical), hip, wrist, and humerus were considered major osteoporotic fractures (MOF). Asymptomatic vertebral fractures found at the X-ray examinations were considered previous fractures [23].
Statistics
Categorical variables have been reported as frequencies (percentage). Continuous variables are expressed as mean and standard deviation (SD). Mixed ANOVA for repeated measures was used to assess the overtime variations of the DXA parameters and the possible influence of clinical factors. T- and Z-scores at each of the five timepoints were used as the within-subjects factor. At the same time, clinical variables (age, sex, osteoporosis at the diagnosis, menopause) were considered between-subjects factors. Mauchly’s Test was used to assess sphericity, and a Greenhouse–Geisser correction was applied in case of violation of sphericity. Post hoc analyses were performed with a Bonferroni adjustment. A p < 0.05 was considered statistically significant for all the analyses.
The diagnostic power for the detection of osteoporosis-related fracture of FRAX was estimated using the receiver operator characteristic (ROC) curve analysis.
Ethics
This study was approved by the Institutional Review Board of the Bologna Authority S.Orsola-Malpighi Hospital (Protocol 243/2013/O/OssN) and performed according to the Declaration of Helsinki guidelines. Informed consent was obtained according to Institutional Review Board instructions.
Patient and public involvement
Patients or the public were not involved in the design, or conduct, or reporting, or dissemination plans of our research.
Results
Study population
Among 1202 patients in our database, 1066 had an index DXA at the diagnosis of CD. Of them, 601 (56.4%) had normal results. The remaining 465 (43.4%) had a T-score < − 1.0 in at least one site and were followed up. Among them, 120 patients had a follow-up > 10 years. Thirteen patients, however, had missing or out-of-window data and were excluded. Thus, the final study population included 107 patients.
Baseline characteristics
Most of patients were females (n = 88, 82.2%), among whom 35 were in menopause. The mean age was 43.4 years (SD 13.3), with 50.5% of patients being > 40-year-old. Thirty-four (31.8%) patients had a classical presentation of CD. The prevalence of factors favoring a condition of osteoporosis is reported in Table 1.
The mean T-scores were − 2.1 (SD 0.8) and − 1.4 (SD 0.7) at the lumbar spine and hip, respectively. According to their T-scores, 32 (29.9%) patients had osteoporosis, and 75 (70.1%) had osteopenia. Among the 32 patients with osteoporosis, 6 had T-scores < − 2.5 at both spine and hip, while 26 only at the spine.
In a multivariable model including age at the diagnosis (categorized as < 40 or ≥ 40 years), sex, body mass index, family history of CD, and clinical presentation (classical vs. non-classical), both age ≥ 40 years [odds ratio 5.11, 95% confidence interval 1.86–14.03, p = 0.002] and classical presentation [odds ratio 4.95, 95% confidence interval 1.87–13.17, p = 0.001] were independently associated with osteoporosis.
The mean Z-scores were − 1.6 (SD 0.7) and − 1.0 (SD 0.7) at the lumbar spine and total hip, respectively. Based on their Z-scores, 31 (29.0%) patients had an inadequate bone mineral density for age. In this case, the alterations were found in both sites in 5 patients and in a single site in 26 patients (spine n = 25, hip n = 1). Classical presentation was the only factor independently associated with BMD below the expected range for age according to the multivariable regression model [odds ratio 2.81, 95% confidence interval 1.17–6.84, p = 0.021].
The mean estimated 10-year risk of major fragility fractures was 4.0% according to the FRAX algorithm, with eight (7.5%) patients exceeding the 10% risk threshold.
After the baseline evaluation, Vitamin D and calcium were supplemented in 83 (77.6%) and 67 (62.6%) patients. Twenty-two (20.6%) patients were prescribed with bisphosphonates.
Follow-up—changes in clinical data and evolution of DXA-based parameters
The follow-up DXA were performed 2.4 (SD 0.5), 5.0 (SD 1.0), 7.4 (SD1.1), and 10.3 (SD 1.1) years after the first examination. During the follow-up, 18 women transitioned to menopause, with a medium age at menopause of 50.2 (SD 2.1) years. Additional risk factors for fracture appeared in six patients (in most cases, corticosteroids were started to treat concurrent conditions). Six (5.6%) patients had an incomplete adherence to the GFD. Prescription patterns of calcium, vitamin D, and antiresorptive drugs are reported in Fig. 1.
From a descriptive point of view, T- and Z-scores at the lumbar spine and hip had an upward trend at the first follow-up, with different subsequent patterns according to the site (Fig. 2).
The mixed ANOVA for repeated measures confirmed variation of T-scores over time, both at lumbar spine [F (2.563, 271.687) = 4.929, p 0.001] and at the hip [F (2.763, 292.887) = 9.873, p < 0.001]. Post hoc analysis revealed that values at the lumbar spine improved from the baseline to the first follow-up (p = 0.002) and that there were no significant differences between baseline and the last follow-up (p = 1.000). Instead, the differences between the baseline and the first (p = 0.089) and last follow-up (p = 0.072) were borderline significant at the hip.
The ANOVA also confirmed Z-scores variation over time, both at lumbar spine [F (2.686, 265.906) = 20.038, p < 0.001] and at the hip [F (2.642, 280.037) = 6.091, p = 0.001]. The post hoc analysis at the lumbar spine showed that all follow-up values significantly increased (p < 0.001 in all cases). At the hip, the scores improved at the first follow-up (p = 0.002); this benefit remained steady, with a significant difference at the last follow-up (p = 0.006).
Finally, the mixed ANOVA model did not show significant interactions between variations in DXA parameters and sex (T-score lumbar spine p = ; T-score hip p = ; Z-score lumbar spine p = 0.938; Z-score hip p = 0.699), age > 40 years at the diagnosis (T-score lumbar spine p = 0.273; T-score hip p = 0.390; Z-score lumbar spine p = 0.508; Z-score hip p = 0.206), or menopause (T-score lumbar spine p = 0.878; T-score hip p = 0.812; Z-score lumbar spine p = 0.910; Z-score hip p = 0.924). With the strong limitation of the small number of non-adherent patients (n = 6), no statistically significant interactions were found for dietary adherence. Instead, DXA parameters variations were influenced by osteoporosis at the diagnosis (T-score lumbar spine p = 0.002; T-score hip p = 0.496; Z-score lumbar spine p < 0.001; Z-score hip p < 0.001).
Consequently, separated subgroup analyses for patients with osteopenia vs. osteoporosis were performed. These analyses confirmed different trajectories of the DXA parameters. In particular, patients with osteoporosis had sharper increases in both T- and Z-scores; on the contrary, patients with osteopenia had relatively more stable values across the 10-year observation (Fig. 3).
Follow-up—changes in the FRAX score
The evolution of the fracture risk estimated according to the FRAX algorithm (both reported as a continuous variable and categorized according to different thresholds) is reported in Table 2.
A subgroup analysis was performed considering: (1) patients with osteopenia at the diagnosis and no risk factors (standard-risk group); (2) patients with osteoporosis at the index DXA or at least one risk factor (increased-risk group). In the standard-risk group (n = 64), the FRAX score was always < 10% at the baseline and never exceeded the 10% threshold at the end of observation (range 1.7–9.6%). Also, only one patient (1.6%) increased his score by more than 5% (Fig. 4). In the high-risk group (n = 43), nine (20.9%) patients started above the 10% threshold. Among them, seven remained over this threshold, and two reduced their risk; seven patients started with a < 10% risk and surpassed this threshold over time. Overall, the number of patients with a > 10% risk of major fractures was stable over time (p = 0.182). Moreover, ten (23.3%) patients increased their score by > 5%.
Image of individual variations in fracture risk according to the FRAX algorithm after 10 years from the start of the gluten-free diet. Percentage changes from the baseline score are reported, with patients being stratified as increased-risk group (i.e., patients with osteoporosis at the baseline dual X-ray absorptiometry or risk factors for fragility fracture—n = 43) or standard-risk group (n = 64)
Follow-up—incident major fragility fractures
Six major fragility fractures occurred in five (4.6%) patients during the 10-year observation time (Supplementary material). Four patients had osteoporosis and a high FRAX score at the index examination. The remaining fractured patient had low bone mineral density at the baseline DXA and began a long-term oral corticosteroid therapy for a severe case of Sjogren syndrome diagnosed 2 years after the diagnosis of CD. Among the 102 non-fractured patients, 4 had a high FRAX score at the diagnosis. Therefore, the positive predictive value, negative predictive value, sensitivity, and specificity of the 10% risk threshold were 50%, 99.0%, 80%, and 96.1%, respectively. The ROC curve analysis confirmed a good discriminative power (area under the curve = 0.826).
Discussion
Metabolic bone disease is a relevant problem for CD patients. Still, the current guidelines for CD acknowledge that some points remain elusive and provide discordant recommendations as a consequence [28]. This study was designed to fill these gaps in knowledge and provide information to refine and, possibly, standardize these recommendations. We performed this task by conjugating a large population and an unprecedented long-term follow-up, covering more than 100,000 patient years and testing the risk estimates of the FRAX score against the actual number of fractures. Our findings bring information about the timing of the first and subsequent DXA examinations.
Regarding the timing of the first evaluation, our results confirmed a relatively high prevalence of osteoporosis at the diagnosis of CD [29], even in non-negligible proportion of younger patients. Therefore, our results support the ACG and ESsCD recommendation to perform a DXA in all adult CD patients at the diagnosis [12, 13], especially in patients > 40-year-old or with malabsorption symptoms.
However, the novel information of our study regards the follow-up. First, T-scores did not overtly deteriorate over time. This result derived from Z-scores improving, which can be explained in two ways: regularized intestinal absorption due to mucosal healing, and prompt detection and management of low bone mass. Unlike the general population with similar demographic characteristics, in fact, CD patients are continuously monitored for vitamin D and calcium levels [12,13,14,15]. Notably, the largest fluctuations in BMD were seen in patients with osteoporosis at the index measurement, while patients with osteopenia had more stable scores. Similarly, the fluctuations of the FRAX score were extremely limited in patient with osteopenia and no additional risk factors. Since the FRAX score confirmed its prognostic abilities in the setting of CD (both in this study and in a recent registry-based cohort study [27]), some implications can be drawn. In fact, our data seem to suggest that patients with osteoporosis at the index DXA should be constantly monitored, but applying the same follow-up schedule in patients with osteopenia and no risk factors could bring more limited information and benefits.
Before generalizing this hypothesis to the whole CD population, some limitations of this study should be discussed. First, the proportion of non-adherent patients was similar to that previously reported [30, 31] but still too low to analyze the relationship between adherence and densitometry modifications properly. Therefore, no inferences should be drawn from the apparent lack of correlation between non-adherence DXA parameters. These patients should continue a close monitoring regardless of their basal BMD, as only a strict GFD can avoid a persistent malabsorption [29]. Second, our study population included only a minority of post-menopausal women. Even if menopause and older age were not found to affect the BMD dynamic changes independently, we feel that future studies specifically dedicated to this population are warranted before drawing strong conclusions.
Even considering these limitations, our results still apply to a vast majority of the CD population, which usually receives a diagnosis well before the pre-menopause and adhere to the GFD. Also, our findings are corroborated by a slim proportion of the target population being excluded for incomplete data (thus making a sample bias very unlikely).
Based on these premises, a mixed approach might be proposed. DXA scans could be prescribed to all adult CD patients at the diagnosis for a comprehensive risk stratification, but with different follow-up programs in patients at high risk (which should maintain a strict 2–3 year interval) and low risk (in which a more relaxed follow-up is unlikely to lead to a loss of relevant clinical information). The FRAX score could be used at each follow-up visit to re-evaluate the timing of the next DXA. This suggestion basically consists in the application of the BSG and NICE guidelines (which suggest calculating the FRAX score without BMD information and prescribe DXA only to patients over predetermined risk thresholds) [14, 15] to the sole follow-up setting. This proposal is also in line with the recently advocated proposal of reducing the number of DXA scans in CD patients, to abate diagnosis-related time and costs [32].
In conclusion, we demonstrated that 10 years after the start of the gluten-free diet, the DXA parameters and the FRAX scores of CD patients are substantially stable compared to those found at the diagnosis. Patients without osteoporosis at the index DXA or other risk factors for fragility fracture had remarkably stable parameters and fracture risk. To reduce the time and cost of the procedures related to CD, this subgroup of patients might benefit from more relaxed follow-up without fears of losing crucial clinical information.
Data availability
The authors confirm that the data supporting the findings of this study are available within the article and its supplementary materials.
References
Kemppainen T, Kröger H, Janatuinen E et al (1999) Osteoporosis in adult patients with celiac disease. Bone 24:249–255. https://doi.org/10.1016/s8756-3282(98)00178-1
Meyer D, Stavropolous S, Diamond B et al (2001) Osteoporosis in a north american adult population with celiac disease. Am J Gastroenterol 96:112–119. https://doi.org/10.1111/j.1572-0241.2001.03507.x
Sategna-Guidetti C, Grosso SB, Grosso S et al (2000) The effects of 1-year gluten withdrawal on bone mass, bone metabolism and nutritional status in newly-diagnosed adult coeliac disease patients. Aliment Pharmacol Ther 14:35–43. https://doi.org/10.1046/j.1365-2036.2000.00671.x
Sánchez MIP, Mohaidle A, Baistrocchi A et al (2011) Risk of fracture in celiac disease: gender, dietary compliance, or both? World J Gastroenterol 17:3035–3042. https://doi.org/10.3748/wjg.v17.i25.3035
Jafri MR, Nordstrom CW, Murray JA et al (2008) Long-term fracture risk in patients with celiac disease: a population-based study in Olmsted County, Minnesota. Dig Dis Sci 53:964–971. https://doi.org/10.1007/s10620-007-9976-0
Ludvigsson JF, Michaelsson K, Ekbom A, Montgomery SM (2007) Coeliac disease and the risk of fractures—a general population-based cohort study. Aliment Pharmacol Ther 25:273–285. https://doi.org/10.1111/j.1365-2036.2006.03203.x
West J, Logan RFA, Card TR et al (2003) Fracture risk in people with celiac disease: a population-based cohort study. Gastroenterology 125:429–436. https://doi.org/10.1016/s0016-5085(03)00891-6
Bai JC, Gonzalez D, Mautalen C et al (1997) Long-term effect of gluten restriction on bone mineral density of patients with coeliac disease. Aliment Pharmacol Ther 11:157–164. https://doi.org/10.1046/j.1365-2036.1997.112283000.x
Valdimarsson T, Löfman O, Toss G, Ström M (1996) Reversal of osteopenia with diet in adult coeliac disease. Gut 38:322–327. https://doi.org/10.1136/gut.38.3.322
Ciacci C, Maurelli L, Klain M et al (1997) Effects of dietary treatment on bone mineral density in adults with celiac disease: factors predicting response. Am J Gastroenterol 92:992–996
Mautalen C, González D, Mazure R et al (1997) Effect of treatment on bone mass, mineral metabolism, and body composition in untreated celiac disease patients. Am J Gastroenterol 92:313–318
Rubio-Tapia A, Hill ID, Kelly CP et al (2013) ACG clinical guidelines: diagnosis and management of celiac disease. Am J Gastroenterol 108:656–676. https://doi.org/10.1038/ajg.2013.79. (quiz 677)
Al-Toma A, Volta U, Auricchio R et al (2019) European Society for the Study of Coeliac Disease (ESsCD) guideline for coeliac disease and other gluten-related disorders. United European Gastroenterol J 7:583–613. https://doi.org/10.1177/2050640619844125
Ludvigsson JF, Bai JC, Biagi F et al (2014) Diagnosis and management of adult coeliac disease: guidelines from the British Society of Gastroenterology. Gut 63:1210–1228. https://doi.org/10.1136/gutjnl-2013-306578
Downey L, Houten R, Murch S et al (2015) Recognition, assessment, and management of coeliac disease: summary of updated NICE guidance. BMJ 351:h4513. https://doi.org/10.1136/bmj.h4513
Kanis JA, Oden A, Johnell O et al (2007) The use of clinical risk factors enhances the performance of BMD in the prediction of hip and osteoporotic fractures in men and women. Osteoporos Int 18:1033–1046. https://doi.org/10.1007/s00198-007-0343-y
Papaioannou A, Morin S, Cheung AM et al (2010) 2010 clinical practice guidelines for the diagnosis and management of osteoporosis in Canada: summary. CMAJ 182:1864–1873. https://doi.org/10.1503/cmaj.100771
Leslie WD, Berger C, Langsetmo L et al (2011) Construction and validation of a simplified fracture risk assessment tool for Canadian women and men: results from the CaMos and Manitoba cohorts. Osteoporos Int 22:1873–1883. https://doi.org/10.1007/s00198-010-1445-5
Kanis JA, Harvey NC, McCloskey E et al (2020) Algorithm for the management of patients at low, high and very high risk of osteoporotic fractures. Osteoporos Int 31:1–12. https://doi.org/10.1007/s00198-019-05176-3
Ludvigsson JF, Leffler DA, Bai JC et al (2013) The Oslo definitions for coeliac disease and related terms. Gut 62:43–52. https://doi.org/10.1136/gutjnl-2011-301346
Silvester JA, Graff LA, Rigaux L et al (2016) Symptomatic suspected gluten exposure is common among patients with coeliac disease on a gluten-free diet. Aliment Pharmacol Ther 44:612–619. https://doi.org/10.1111/apt.13725
Protocollo diagnosi e follow-up celiachia. In: AIC—Associazione Italiana Celiachia. https://www.celiachia.it/celiachia/diagnosi/protocollo-diagnosi-e-follow-up-celiachia/. Accessed 22 Jul 2022
WHO Scientific Group on the Prevention and Management of Osteoporosis (2000) : Geneva S (2003) Prevention and management of osteoporosis : report of a WHO scientific group. World Health Organization
Hamdy RC (2019) The 2019 ISCD position development conference. J Clin Densitom 22:451–452. https://doi.org/10.1016/j.jocd.2019.09.002
Leslie WD, Caetano PA, Macwilliam LR, Finlayson GS (2005) Construction and validation of a population-based bone densitometry database. J Clin Densitom 8:25–30. https://doi.org/10.1385/jcd:8:1:025
Hui SL, Gao S, Zhou XH et al (1997) Universal standardization of bone density measurements: a method with optimal properties for calibration among several instruments. J Bone Miner Res 12:1463–1470. https://doi.org/10.1359/jbmr.1997.12.9.1463
Duerksen DR, Lix LM, Johansson H et al (2021) Fracture risk assessment in celiac disease: a registry-based cohort study. Osteoporos Int 32:93–99. https://doi.org/10.1007/s00198-020-05579-7
Raiteri A, Granito A, Giamperoli A et al (2022) Current guidelines for the management of celiac disease: a systematic review with comparative analysis. World J Gastroenterol 28:154–175. https://doi.org/10.3748/wjg.v28.i1.154
Catassi C, Verdu EF, Bai JC, Lionetti E (2022) Coeliac disease. Lancet 399:2413–2426. https://doi.org/10.1016/S0140-6736(22)00794-2
Kurppa K, Lauronen O, Collin P et al (2012) Factors associated with dietary adherence in celiac disease: a nationwide study. Dig 86:309–314. https://doi.org/10.1159/000341416
Fernández Miaja M, Díaz Martín JJ, Jiménez Treviño S et al (2021) Study of adherence to the gluten-free diet in coeliac patients. Anales de Pediatría (English Edition) 94:377–384. https://doi.org/10.1016/j.anpede.2020.06.012
Tortora R, Imperatore N, Capone P et al (2018) Frax score can be used to avoid superfluous DXA scans in detecting osteoporosis in celiac disease: accuracy of the FRAX score in celiac patients. J Clin Densitom 21:315–321. https://doi.org/10.1016/j.jocd.2017.05.010
Funding
Open access funding provided by Alma Mater Studiorum - Università di Bologna within the CRUI-CARE Agreement. No funding to declare.
Author information
Authors and Affiliations
Contributions
FT: conceptualization, formal analysis, original draft preparation; DPP: conceptualization, data curation; AG: data curation; GZ: methodology, writing—review and editing; KS: data curation; AR: data curation; CF: data curation; IK-K: methodology, writing—review and editing; AG: writing—review and editing. All authors approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All authors have declared no conflicts of interest.
Human and animal rights
All procedures performed in this study including huma participants were in accordance with the ethical standards of the institution and national research committee and with the 1964 Helsinki declaration and its later amendments.
Informed consent
All participants provided informed consent prior to thier participation.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tovoli, F., Pallotta, D.P., Giamperoli, A. et al. Evolution of bone densitometry parameters and risk of fracture in coeliac disease: a 10-year perspective. Intern Emerg Med 18, 1405–1414 (2023). https://doi.org/10.1007/s11739-023-03307-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11739-023-03307-7