Abstract
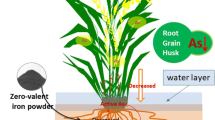
Arsenic contamination is a serious environmental concern in Indo-Gangetic Plains of West Bengal, India and Bangladesh. Productivity of Lentils (Lens culinaris Medik.) is severely affected in this region due to use of contaminated groundwater for irrigation purpose. Precipitation of iron oxides or hydroxides on root surfaces in form of iron plaque is observed as a response to arsenic toxicity in Lentils. Present study was constructed to elucidate the significance of the iron plaque in Lentils (cv. WBL-77) and to find out a possible way to ameliorate arsenic toxicity by inducing iron plaque formation. Lentil seeds were allowed for germination on different concentrations of arsenate, one set of which was supplemented with ferrous sulfate (FeSO4). The supplementation effectively triggered iron plaque formation on emerging radicles and significantly enhanced the rate of successful shoot development. The morphological, physiological and biochemical attributes depicted that the toxic effects of arsenic were considerably less pronounced in those seedlings compared to the set without FeSO4 supplementation. Initially formed surficial plaque was removed from another set of seedlings that significantly restricted their further growth and development compared to the iron plaque containing seedlings. Moreover, arsenic content was higher in iron plaque than the roots and shoots, indicating that the surficial iron plaque attenuated the toxic effects of arsenic by immobilizing and sequestering them. Therefore, FeSO4 alleviated the toxic effects of arsenic in lentils by enhancing the iron plaque formation. The study propounds that FeSO4 supplementation can be an effective measure for stabilization of arsenic in contaminated regions.






Similar content being viewed by others
Availability of data and material
All required data have been provided in supplementary file.
Code availability
Not applicable.
References
Allen SE (1989) Analysis of vegetation and other organic materials. In: Allen SE (ed) Chemical analysis of ecological materials. Blackwell Scientific Publications, Oxford, pp 46–61
Bates LS, Waldren RP, Teare ID (1973) Rapid determination of free proline for water-stress studies. Plant Soil 39:205–207
Bindu N, Gundlur SS (2019) Effect of soil and foliar application of ferrous sulphate on crop growth, seed yield, quality and nutrient uptake by safflower in Vertisol. Environ Ecol 37:68–681
Blute NK, Brabander DJ, Hemond HF, Sutton SR, Newville MG, Rivers ML (2004) Arsenic sequestration by ferric iron plaque on cattail roots. Environ Sci Technol 38:6074–6077
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Dahal BM, Fuerhacker M, Mentler A, Karki KB, Shrestha RR, Blum WEH (2008) Arsenic contamination of soils and agricultural plants through irrigation water in Nepal. Environ Pollut 155:157–163
El-Jendoubi H, Vazquez S, Calatayud A, Vavpetic P, Vogel-Mikus K, Pelicon P, Abadia J, Abadia A, Morales F (2014) The effects of foliar fertilization with iron sulphate in chlorotic leaves are limited to the treated area. A study with peach trees (Prunus persica L. Batsch) grown in the field and sugar beet (Beta vulgaris L.) grown in hydroponics. Front Plant Sci. https://doi.org/10.3389/fpls.2014.00002
Fresno T, Penalosa JM, Santer J, Puschenreiter M, Prohaska T, Moreno-Jimenez E (2016) Iron plaque formed under aerobic conditions efficiently immobilizes arsenic in Lupinus albus L roots. Environ Pollut 216:215–222
Fu Y, Yang X, Shen H (2018) Root iron plaque alleviates cadmium toxicity to rice (Oryza sativa) seedlings. Ecotoxicol Environ Saf 161:534–541
Greipsson S (1994) Effects of iron plaque on roots of rice on growth and metal concentration of seeds and plant tissues when cultivated in excess copper. Commun Soil Sci Plant Anal 25:2761–2769
Greipsson S, Crowder AA (1992) Amelioration of copper and nickel toxicity by iron plaque on roots of rice (Oryza sativa). Can J Bot 70:824–830
Grieve CM, Grattan SR (1983) Rapid assay for determination of water-soluble quaternary-amino compounds. Plant Soil 70:303–307
Hammer O, Harper DAT, Ryan PD (2001) PAST: Paleontological statistics software package for education and data analysis. Paleontol Electron 4:1–9
Hansel CM, La-Force MJ, Fendorf S, Sutton S (2002) Spatial and temporal association of As and Fe species on aquatic plant roots. Environ Sci Technol 36:1988–1994
Hartley W, Edwards R, Lepp NW (2004) Arsenic and heavy metal mobility in iron oxide-amended contaminated soils as evaluated by short- and long-term leaching tests. Environ Pollut 131:495–504
Hayat S, Hayat Q, Alyemeni MN, Wani AS, Pichtel J, Ahmad A (2012) Role of proline under changing environments: a review. Plant Sig Behav 7:1456–1466
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplasts. I. Kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys 125:189–198
Jiang QQ, Singh BR (1994) Effect of different forms and sources of arsenic crop yield and arsenic concentration. Water Air Soil Pollut 74:321–343
Knauer K, Behra R, Hemond H (1999) Toxicity of inorganic and methylated arsenic to algal communities from lakes along an arsenic contamination gradient. Aqua Toxicol 46:221–230
Kumpiene J, Fitts JP, Mench M (2012) Arsenic fractionation in mine spoils 10 years after aided phytostabilization. Environ Pollut 166:82–88
Lee CH, Hsieh YC, Lin TH, Lee DY (2012) Iron plaque formation and its effect on arsenic uptake by different genotypes of paddy rice. Plant Soil 363:231–241
Lin Y, Fan J, Yu J, Jiang S, Yan C, Liu J (2018) Root activities and arsenic translocation of Avicennia marina (Forsk.) Vierh seedlings influenced by sulfur and iron amendments. Marine Pollut Bull 135:1174–1182
Liu JG, Li KQ, Xu JK, Zhang ZJ, Ma TB, Lu XL, Yang JC, Zhu QS (2003) Lead toxicity, uptake and translocation in different rice cultivars. Plant Sci 165:793–802
Liu WJ, Zhu YG, Smith FA, Smith SE (2004) Do iron plaque and genotypes affect arsenate uptake and translocation by rice seedlings (Oryza sativa L.) grown in solution culture? J Exp Bot 55:1707–1713
Liu WJ, Zhu YG, Hu Y, Williams PN, Gault AG, Meharg AA, Charnock JM, Smith FA (2006) Arsenic sequestration in iron plaque, its accumulation and speciation in mature rice plants (Oryza sativa L.). Environ Sci Technol 40:5730–5736
Lurie S, Fallik E, Handros A, Shapira R (1997) The possible involvement of peroxidase in resistance to Botrytis cinerea in heat treated tomato fruit. Physiol Mol Plant Pathol 50:141–149
Makris KC, Punamiya P, Sarkar D, Datta R (2008) Novel colorimetric method overcoming phosphorus interference during trace arsenic analysis in soil solution. Analyst 133:191–196
Meguro R, Asano Y, Odagiri S, Li C, Iwatsuki H, Shoumura K (2007) Nonheme iron histochemistry for light and electron microscopy: a historical, theoretical and technical review. Arch Histol Cytol 70:1–19
Mishra P, Dubey RS (2013) Excess nickel modulates activities of carbohydrate metabolizing -enzymes and induces accumulation of sugars by up-regulating acid invertase and sucrose synthase in rice seedlings. Biometals 26:97–111
Montillet JL, Chamnongpol S, Rust´erucci C, Dat J, van de Cotte B, Agnel JP, Battesti C, Inze D, Breusegem FV, Triantaphylides C (2005) Fatty acid hydroperoxides and H2O2 in the execution of hypersensitive cell death in tobacco leaves. Plant Physiol 138:1516–1526
Moore TJ, Rightmire CM, Vempati RK (2000) Ferrous iron treatment of soils contaminated with wood-preserving solution. Soil Sed Contamin 9:375–405
Mukherjee A, Sengupta MK, Hossain MA, Ahamed S, Das B, Nayak B, Lodh D, Rahman MM, Chakraborti D (2006) Arsenic contamination in groundwater: a global perspective with emphasis on the Asian scenario. J Health Popul Nutr 24:142–163
Oberbacher MF, Vines HM (1963) Spectrophotometric assay of ascorbic acid oxidase. Nature 197:1203–1204
Otte M, Rozema J, Koster L, Haarsma M, Broekman R (1989) Iron plaque on roots of Aster tripolium L.: interaction with zinc uptake. New Phytol 111:309–317
Otte ML, Dekkers MJ, Rozema J, Broekman RA (1991) Uptake of arsenic by Aster tripolium in relation to rhizosphere oxidation. Can J Bot 69:2670–2677
Peng C, Chen S, Shen C, He M, Zhang Y, Ye J, Liu J, Shi J (2018) Iron plaque: a barrier layer to the uptake and translocation of copper oxide nanoparticles by rice plants. Environ Sci Technol 52:12244–12254
Sairam RK (1994) Effect of moisture-stress on physiological activities of two contrasting wheat genotypes. Ind J Exp Biol 31:551–553
Sakamoto A, Murata N (2002) The role of glycine betaine in the protection of plants from stress: clues from transgenic plants. Plant Cell Environ 25:163–171
Sebastian A, Prasad MNV (2016) Iron plaque decreases cadmium accumulation in Oryza sativa L. and serves as a source of iron. Plant Biol 18:1008–1015
Syu CH, Jiang PY, Huang HH, Chen WT, Lin TH, Lee DY (2013) Arsenic sequestration in iron plaque and its effect on As uptake by rice plants grown in paddy soils with high contents of As, iron oxides and organic matter. Soil Sci Plant Nutr 59:463–471
Singh AP, Dixit G, Kumar A, Mishra S, Kumar N, Dixit S, Singh PK, Dwivedi S, Trivedi PK, Pandey V (2017) A protective role for nitric oxide and salicylic acid for arsenite phytotoxicity in rice (Oryza sativa L.). Plant Physiol Biochem 115:163–173
Siqueira-Silva AI, Da Silva LC, Azevedo AA, Oliva MA (2012) Iron plaque formation and orphoanatomy of roots from species of restinga subjected to excess iron. Ecotoxicol Environ Saf 78:265–275
Slama I, Abdelly C, Bouchereau A, Flowers T, Savouré A (2015) Diversity, distribution and roles of osmoprotective compounds accumulated in halophytes under abiotic stress. Ann Bot 115:433–447
Smedley PL, Kinniburgh DG (2013) Arsenic in groundwater and the environment. In: Selinus O (ed) Essentials of Medical Geology. Springer, Amsterdam, pp 279–310
Tang T, Miller DM (1991) Growth and tissue composition of rice grown in soil treated with inorganic copper, nickel, and arsenic. Commun Soil Sci Plant Anal 22:2037–2045
Tripathi RD, Tripathi P, Dwivedi S, Kumar A, Mishra A, Chauhan PS, Norton GJ, Nautiyal CS (2014) Roles for root iron plaque in sequestration and uptake of heavy metals and metalloids in aquatic and wetland plants. Metallomics 6:1789–1800
Ullrich-Eberius C, Sanz A, Novacky A (1989) Evaluation of arsenate-and vanadate-associated changes of electrical membrane potential and phosphate transport in Lemna gibba G1. J Exp Bot 40:119–128
Vangronsveld J, Herzig R, Weyens N, Boulet J, Adriaensen K, Ruttens A, Thewys T, Vassilev A, Meers E, Nehnevajova E, van der Lelie D, Mench M (2009) Phytoremediation of contaminated soils and groundwater: lessons from the field. Environ Sci Pollut Res 16:765–794
Warren GP, Alloway BJ (2003) Reduction of arsenic uptake by lettuce with ferrous sulfate applied to contaminated soil. J Environ Qual 32:767–772
Weatherley PE (1950) Studies in the water relations of the cotton plant: I. The field measurement of water deficits in leaves. New Phytol 49:81–97
Williams PN, Santner J, Larsen M, Lehto NJ, Oburger E, Wenzel W, Glud RN, Davison W, Zhang H (2014) Localized flux maxima of arsenic, lead, and iron around root apices in flooded lowland rice. Environ Sci Technol 48:8498–8506
Wu C, Huang L, Xue S, Pan W, Zou Q, Hartley W, Mo J (2017) Effect of arsenic on spatial pattern of radial oxygen loss and iron plaque formation in rice. Transac Nonferr Metals Soc China 27:413–419
Xu B, Yu JY, Xie T, Li YL, Liu MJ, Guo JX, Li HL, Yu Y, Zheng CY, Chen YH (2018) Brassinosteroids and iron plaque affect arsenic and cadmium uptake by rice seedlings grown in hydroponic solution. Biol Plant 62:362–368. https://doi.org/10.1007/s10535-018-0784-5
Yemm EW, Willis AJ (1954) The estimation of carbohydrates in plant extracts by anthrone. Biochem J 57:508–514
Yu HY, Wang X, Li F, Li B, Liu C, Wang Q, Lei J (2017) Arsenic mobility and bioavailability in paddy soil under iron compound amendments at different growth stages of rice. Environ Pollut 224:136–147
Zhang L, Qin X, Tang J, Liu W, Yang H (2016) Review of arsenic geochemical characteristics and its significance on arsenic pollution studies in karst groundwater, Southwest China. Appl Geochem 77:80–88
Zhao FJ, Ma JF, McGrath SP, Meharg AA (2009) Arsenic uptake and metabolism in plants. New Phytol 181:777–794
Zimmer D, Kruse J, Baum C, Borca C, Laue M, Hause G, Meissner R, Leinweber P (2011) Spatial distribution of arsenic and heavy metals in willow roots from a contaminated floodplain soil measured by X-ray fluorescence spectroscopy. Sci Total Environ 409:4094–4100
Zohary D (1972) The wild progenitor and the place of origin of the cultivated lentil: Lens culinaris. Econ Bot 26:326–332
Acknowledgements
Authors are thankful to The Principal, Darjeeling Government College for providing necessary infrastructural facilities to perform the experimental works.
Funding
This research received no specific grant from any funding agency, commercial or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors declare that they have no conflict of interest.
Ethics approval
Plant samples are not under any protective figure and hence no permission was required.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Communicated by C. L. Cespedes.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mitra, S., Subba, S. Alleviative effects of ferrous sulfate on arsenic toxicity in Lens culinaris Medik. by enhancing iron plaque formation on roots. Acta Physiol Plant 44, 126 (2022). https://doi.org/10.1007/s11738-022-03467-7
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11738-022-03467-7