Abstract
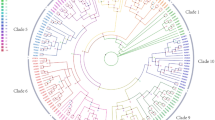
Stone cell which appears in most pear cultivars contributes badly to pear internal quality. Lignin is the important component of stone cells and its first biosynthetic but specific step is catalyzed by cinnamoyl CoA reductase (CCR). In the present study, a cDNA encoding CCR, named as PpCCR (GenBank accession no. GU138672), was isolated from pear (Pyrus pyrifolia) fruit. Its full length is 1,247 bp with a putative maximal open reading frame of 1,017 bp, encoding a protein of 339 amino acids. Sequence alignment revealed that the deduced protein contained a typical CCR catalytic site and NADP(H) binding site. Phylogenetic analysis showed that PpCCR belonged to dicot CCR class II in which most CCRs participated in the regulation of lignin biosynthesis. PpCCR transcript could be detected in leaves, flowers and fruits. In addition, PpCCR expression profiles during fruit development were compared between two cultivars with significant difference in stone cell content in the flesh. Although PpCCR expressed in a similar pattern between the two cultivars and their transcript level consisted strictly with the change of lignin content, its transcript level was always higher in the high-lignin content cultivar than in the low-lignin content cultivar during fruit development. Therefore, it could be suggested that PpCCR is involved in the regulation of lignin biosynthesis and plays an important role in stone cell formation in pear flesh.





Similar content being viewed by others
References
Boerjan W, Ralph J, Baucher M (2003) Lignin biosynthesis. Annu Rev Plant Biol 54:519–546
Bruce RJ, West CA (1989) Elicitation of lignin biosynthesis and isoperoxidase activity by pectic fragments in suspension cultures of castor bean. Plant Physiol 91:889–897
Cai C, Xu CJ, Li X, Ferguson I, Chen KS (2006) Accumulation of lignin in relation to change in activities of lignification enzymes in loquat fruit flesh after harvest. Postharvest Biol Technol 40:163–169
Capeleti I, Bonini EA, Ferrarese MDL, Teixeira ACN, Krzyzanowski FC, Ferrarese-Filho O (2005) Lignin content and peroxidase activity in soybean seed coat susceptible and resistant to mechanical damage. Acta Physiol Plant 27:103–108
Chabannes M, Barakate A, Lapierre C, Marita JM, Ralph J, Pean M, Danoun S, Halpin C, Grima-Pettenati J, Boudet AM (2001) Strong decrease in lignin content without significant alteration of plant development is induced by simultaneous down-regulation of cinnamoyl CoA reductase (CCR) and cinnamyl alcohol dehydrogenase (CAD) in tobacco plants. Plant J 28:257–270
Choi JH, Choi JJ, Hong KH, Kim WS, Lee SH (2007) Cultivar differences of stone cells in pear flesh and their effects on fruit quality. Hortic Environ Biotechnol 48:27–31
Crist JW, Batjer LP (1931) The stone cells of pear fruits, especially the kieffer pear. Mich Agric Exp Station Tech Bull 113:1–55
Goujon T, Ferret V, Mila I, Pollet B, Ruel K, Burlat V, Joseleau JP, Barriere Y, Lapierre C, Jouanin L (2003) Down-regulation of the AtCCR1 gene in Arabidopsis thaliana: effects on phenotype, lignins and cell wall degradability. Planta 217:218–228
Hu CG, Honda C, Kita M, Zhang ZL, Tsuda T, Takaya M (2002) A simple protocol for RNA isolation from fruit trees containing high levels of polysaccharides and polyphenol compounds. Plant Mol Biol Rep 20:69a–69g
Kaczkowski J (2003) Structure, function and metabolism of plant cell wall. Acta Physiol Plant 25:287–305
Kozak M (1984) Compilation and analysis of sequences upstream from the translation start site in eukaryotic messenger-RNAs. Nucleic Acids Res 12:857–872
Kuc J (1997) Molecular aspects of plant responses to pathogens. Acta Physiol Plant 19:551–559
Lacombe E, Hawkins S, VanDoorsselaere J, Piquemal J, Goffner D, Poeydomenge O, Boudet AM, GrimaPettenati J (1997) Cinnamoyl CoA reductase, the first committed enzyme of the lignin branch biosynthetic pathway: cloning, expression and phylogenetic relationships. Plant J 11:429–441
Larsen K (2004a) Molecular cloning and characterization of cDNAs encoding cinnamoyl CoA reductase (CCR) from barley (Hordeum vulgare) and potato (Solanum tuberosum). J Plant Physiol 161:105–112
Larsen K (2004b) Cloning and characterization of a ryegrass (Lolium perenne) gene encoding cinnamoyl-CoA reductase (CCR). Plant Sci 166:569–581
Lauvergeat V, Lacomme C, Lacombe E, Lasserre E, Roby D, Grima-Pettenati J (2001) Two cinnamoyl-CoA reductase (CCR) genes from Arabidopsis thaliana are differentially expressed during development and in response to infection with pathogenic bacteria. Phytochemistry 57:1187–1195
Lee SH, Choi JH, Kim WS, Han TH, Park YS, Gemma H (2006) Effect of soil water stress on the development of stone cells in pear (Pyrus pyrifolia cv. ‘Niitaka’) flesh. Sci Hortic 110:247–253
Leple JC, Dauwe R, Morreel K, Storme V, Lapierre C, Pollet B, Naumann A, Kang KY, Kim H, Ruel K, Lefebvre A, Joseleau JP, Grima-Pettenati J, De Rycke R, Andersson-Gunneras S, Erban A, Fehrle I, Petit-Conil M, Kopka J, Polle A, Messens E, Sundberg B, Mansfield SD, Ralph J, Pilate G, Boerjan W (2007) Downregulation of cinnamoyl-coenzyme A reductase in poplar: multiple-level phenotyping reveals effects on cell wall polymer metabolism and structure. Plant Cell 19:3669–3691
Li YH, Kajita S, Kawai S, Katayama Y, Morohoshi N (2003) Down-regulation of an anionic peroxidase in transgenic aspen and its effect on lignin characteristics. J Plant Res 116:175–182
Liu L, Li J, Qin WM (2005) Stone cell development and its effects on the sarcocarp of Kuerle fragrant pear. Xibei Zhiwu Xuebao 25:1965–1968
Lutcke HA, Chow KC, Mickel FS, Moss KA, Kern HF, Scheele GA (1987) Selection of AUG initiation codons differs in plants and animals. EMBO J 6:43–48
Ma QH (2007) Characterization of a cinnamoyl-CoA reductase that is associated with stem development in wheat. J Exp Bot 58:2011–2021
McInnes R, Lidgett A, Lynch D, Huxley H, Jones E, Mahoney N, Spangenberg G (2002) Isolation and characterization of a cinnamoyl-CoA reductase gene from perennial ryegrass (Lolium perenne). J Plant Physiol 159:415–422
Pichon M, Courbou I, Beckert M, Boudet AM, Grima-Pettenati J (1998) Cloning and characterization of two maize cDNAs encoding Cinnamoyl-CoA Reductase (CCR) and differential expression of the corresponding genes. Plant Mol Biol 38:671–676
Piquemal J, Lapierre C, Myton K, O’Connell A, Schuch W, Grima-Pettenati J, Boudet AM (1999) Down-regulation of Cinnamoyl-CoA Reductase induces significant changes of lignin profiles in transgenic tobacco plants. Plant J 13:71–78
Qiao YJ, Zhang SL, Tao ST, Zhang ZM, Liu ZL (2005) Advances in research on developing mechanism of stone cells in pear fruit. J Fruit Sci 22:367–371 (in Chinese with English abstract)
Ranadive AS, Haard NF (1973) Chemical nature of stone cells from pear fruit. J Food Sci 38:331–333
Selman-Housein G, Lopezm MA, Hernandez D, Civardi L, Miranda F, Rigau J, Puigdomenech P (1999) Molecular cloning of cDNAs coding for three sugarcane enzymes involved in lignification. Plant Sci 143:163–171
Solecka D (1997) Role of phenylpropanoid compounds in plant responses to different stress factors. Acta Physiol Plant 19:257–268
Tamura K, Dudley J, Nei M, Kumar S (2007) MEGA4: molecular evolutionary genetics analysis (MEGA) software version 4.0. Mol Biol Evol 24:1596–1599
Tao ST, Khanizadeh S, Zhang H, Zhang SL (2009) Anatomy, ultrastructure and lignin distribution of stone cells in two Pyrus species. Plant Sci 176:413–419
Wadenback J, von Arnold S, Egertsdotter U, Walter MH, Grima-Pettenati J, Goffner D, Gellerstedt G, Gullion T, Clapham D (2008) Lignin biosynthesis in transgenic Norway spruce plants harboring an antisense construct for cinnamoyl CoA reductase (CCR). Transgenic Res 17:379–392
Acknowledgments
We are grateful to National Sand Pear Germplasm Repository, Wuhan, China, for kindly providing the plant materials. This work was supported by National Natural Science Foundation of China (No. 30921002).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by H. Janska.
Rights and permissions
About this article
Cite this article
Lu, XP., Liu, YZ., An, JC. et al. Isolation of a cinnamoyl CoA reductase gene involved in formation of stone cells in pear (Pyrus pyrifolia). Acta Physiol Plant 33, 585–591 (2011). https://doi.org/10.1007/s11738-010-0583-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11738-010-0583-x