Abstract
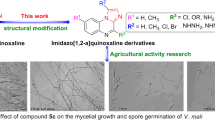
A series of N-(thiazol-4-ylmethyl) benzenesulfonamide derivatives were designed, synthesized, and characterized by 1H NMR, 13C NMR, and HRMS. The evaluation of fungicidal activities against Fusarium oxysporum (F. oxysporum), Sclerotinia sclerotiorum (S. sclerotiorum), Botrytis cinerea (B. cinerea), Colletotrichum fragariae (C. fragariae), Alternaria alternata (A. alternata), Penicillium digitatum (P. digitatum), Curvularia lunata (C. lunata), and Curvularia mebaldsii (C. mebaldsii) for the target compounds was carried out. The results indicated that this series of compounds were sensitive to C. lunata, B. cinerea, and S. sclerotiorum. Especially, 2,4-dichloro-N-((2-phenylthiazol-4-yl)methyl)benzenesulfonamide (5c) exhibited the best bioactivities against C. lunata, B. cinerea, and S. sclerotiorum with EC50 value of 20.8 mg/L (52.3 μM), 24.6 mg/L (61.8 μM), and 9.5 mg/L (23.9 μM), respectively, which has equivalent or superior fungicidal activity to commercial products hymexazol. Furthermore, the structure–activity relationship was given, which might be contribution to the design and discovery of N-(thiazol-4-ylmethyl)benzenesulfonamide derivatives as novel fungicide.




Similar content being viewed by others
References
Cai N, Liu CX, Feng ZH et al (2018) Design, synthesis, and SAR of novel 2-Glycinamide Cyclohexyl Sulfonamide derivatives against botrytis cinerea. Molecules 23:740. https://doi.org/10.1023/A:1008722005570
Fontaine S, Remuson F, Caddoux L, Barres B (2019) Investigation of the sensitivity of Plasmopara viticola to amisulbrom and ametoctradin in French vineyards using bioassays and molecular tools. Pest Manag Sci 75:2115–2123. https://doi.org/10.1002/ps.5461
Hantzsch A, Weber JH (1887) Ueber verbindungen des thiazols (pyridins der thiophenreihe). Ber Dtsch Chem Ges 20:3118–3132. https://doi.org/10.1002/cber.188702002200
Hide GA, Bell F (1980) Effects of treating seed potatoes from commercial and stem cutting stocks with benomyl, thiabendazole and 2-aminobutane on yield and disease. Ann Appl Biol 94:205–214. https://doi.org/10.1111/j.1744-7348.1980.tb03912
Huang DL, Liao M, Zhang TY et al (2020a) Design, synthesis and fungicidal activity of novel 2-aryl-thiazole derivatives containing saccharin motif. ChemistrySelect 5:13179–13182. https://doi.org/10.1002/slct.202003616
Huang DL, Zheng SM, Cheng YX (2020) Design, synthesis and biological evaluation of N-((2-phenyloxazol-4-yl) methyl) pyrimidine Carboxamide derivatives as potential fungicidal agents. Heterocycl Commun 26:185–191. https://doi.org/10.1515/hc-2020b-0117
Huang DL, Zheng SM, Zhang TY, Cheng YX (2021) Design, synthesis, fungicidal activities and structure-activity relationship studies of (-)-borneol derivatives containing 2-aryl-thiazole scaffold. Bioorg Med Chem Lett 45:128006. https://doi.org/10.1016/j.bmcl.2021.128006
Miyake Y, Sakai J, Shibata M et al (2005) Fungicidial activity of benthiavalicarb-isopropyl against Phytophthora infestans and its controlling activity against late blight diseases. J Pestic Sci 30:390–396. https://doi.org/10.1584/jpestics.30.390
Tanaka S, Kochi S, Kunita H et al (1999) Biological mode of action of the fungicide, flusulfamide, against Plasmodiophora brassicae (clubroot). Eur J Plant Pathol 105:577–584. https://doi.org/10.3390/molecules23040740
Wang J, Li C, Song P et al (2022) Molecular and biological characterization of the first mymonavirus identified in Fusarium oxysporum. Front Microbiol 13:870204. https://doi.org/10.3389/fmicb.2022.870204
White DJ, Chen WD, Schroeder KL (2019) Assessing the contribution of ethaboxam in seed treatment cocktails for the management of metalaxyl-resistant Pythium ultimum var. ultimum in Pacific Northwest spring wheat production. Crop Prot 115:7–12. https://doi.org/10.1016/j.cropro.2018.08.026
Yao X, Liu Y, Liu X et al (2022) Effects of thifluzamide on soil fungal microbial ecology. J Hazard Mater 431:128626. https://doi.org/10.1016/10.1016/j.jhazmat.2022.128626
Zhang C, Li T, Xiao L et al (2020) Characterization of tebuconazole resistance in Botrytis cinerea from tomato plants in China. Phytopathology Res 2:25. https://doi.org/10.1186/s42483-020-00064-9
Zhang TY, You SY, Zheng SM et al (2022) Structural modification of saccharin containing 2-aminothiazole as potential fungicidal agents. ChemistrySelect. https://doi.org/10.1002/slct.202103603
Acknowledgments
This work was supported Shenzhen Start-up Funds for Outbound Postdoctoral Research (No. 0001310319).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zheng, S., Huang, D. Design, synthesis and fungicidal activities evaluation of N-(thiazol-4-ylmethyl) benzenesulfonamide derivatives. Chem. Pap. 77, 1059–1066 (2023). https://doi.org/10.1007/s11696-022-02540-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-022-02540-2