Abstract
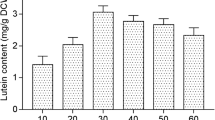
Ultrasound-assisted extraction (UAE) of fucoxanthin and total pigment content was performed from brown, red and green macroalgal species (Fucus virsoides, Amphiroa rigida and Codium bursa) from the Adriatic Sea under different experimental conditions of time (15, 30 and 45 min), temperature (30, 50 and 70 °C) and solvent:solid ratio (10, 20 and 30 mL g−1). UAE was evaluated to determine the conditions influence on fucoxanthin yield (dominant pigment). The results showed that brown macroalga F. virsoides contained the highest fucoxanthin content (1.19–1.90 mg g−1 dry extract), followed by red macroalga A. rigida (0.29–0.90 mg g−1 dry extract) and green macroalga C. bursa (0.07–0.43 mg g−1 dry extract). Additionally, total concentrations of pigments (chlorophyll a, chlorophyll b and carotenoids) were determined by spectrophotometric analysis. Significant dependence on the applied extraction conditions for fucoxanthin and total pigments yields was revealed by the analysis of variance for the response surface models. Accordingly, the results showed differences for each macroalgal species in terms of the optimal UAE parameters. It can be concluded that UAE is successful method for the extraction of fucoxanthin from different macroalgal species, but the extraction optimization is crucial.
Graphical abstract





Similar content being viewed by others
References
Aburai N, Ohkubo S, Miyashita H, Abe K (2013) Composition of carotenoids and identification of aerial microalgae isolated from the surface of rocks in mountainous districts of Japan. Algal Res 2:237–243. https://doi.org/10.1016/j.algal.2013.03.001
Agatonović-Kustrin S, Morton DW (2017) High-performance thin-layer chromatography HPTLC-direct bioautography as a method of choice for alpha-amylase and antioxidant activity evaluation in marine algae. J Chromatogr A 29:197–203. https://doi.org/10.1016/j.chroma.2017.11.024
Bas D, Boyacı IH (2007) Modeling and optimization I: Usability of response surface methodology. J Food Eng 78:836–845. https://doi.org/10.1016/j.jfoodeng.2005.11.024
Bhat I, Haripriya G, Jogi N, Mamatha BS (2021) Carotenoid composition of locally found seaweeds of Dakshina Kannada district in India. Algal Res 53:102154. https://doi.org/10.1016/j.algal.2020.102154
Castro-Puyana M, Perez-Sanchez A, Valdes A, Ibrahim OHM, Suarez-Alvarez S, Ferragut JA, Micol V, Cifuentes A, Ibanez E, Garcia-Canas V (2017) Pressurized liquid extraction of Neochloris oleoabundans for the recovery of bioactive carotenoids with anti-proliferative activity against human colon cancer cells. Food Res Int 99:1048–1055. https://doi.org/10.1016/j.foodres.2016.05.021
Catarino MD, Silva AMS, Cardoso SM (2018) Phycochemical constituents and biological activities of Fucus spp. Mar Drugs 16:249. https://doi.org/10.3390/md16080249
Chemat F, Vian MA, Cravotto G (2012) Green extraction of natural products: concept and principles. Int J Mol Sci 13:8615–8627. https://doi.org/10.3390/ijms13078615
Chen IS, Shen CSJ, Sheppard AJ (1981) Comparison of methylene chloride and chloroform for the extraction of fats from food products. J Am Oil Chem Soc 58:599–601. https://doi.org/10.1007/BF02672373
Cvitković D, Dragović-Uzelac V, Dobrinčić A, Čož-Rakovac R, Balbino S (2021) The effect of solvent and extraction method on the recovery of lipid fraction from Adriatic Sea macroalgae. Algal Res 56:102291. https://doi.org/10.1016/j.algal.2021.102291
Dai YL, Jiang YF, Lu YA, Yu YB, Kang MC, Jeon YJ (2021) Fucoxanthin-rich fraction from Sargassum fusiformis alleviates particulate matter-induced inflammation in vitro and in vivo. Toxicol Rep 6:349–358. https://doi.org/10.1016/j.toxrep.2021.02.005
Dasgupta CN (2015) Algae as a source of phycocyanin and other industrially important pigments. In: Das D (ed) Algal biorefinery: an integrated approach. Springer, Cham, Switzerland, pp 253–276
Deenu A, Naruenartwongsakul S, Kim SM (2013) Optimization and economic evaluation of ultrasound extraction of lutein from Chlorella vulgaris. Biotechnol Bioprocess Eng 18:1151–1162
Dey S, Rathod VK (2013) Ultrasound assisted extraction of β-carotene from Spirulina platensis. Ultrason Sonochem 20:271–276. https://doi.org/10.1016/j.ultsonch.2012.05.010
Fariman GA, Shastan SJ, Zahedi MM (2016) Seasonal variation of total lipid, fatty acids, fucoxanthin content, and antioxidant properties of two tropical brown algae (Nizamuddinia zanardinii and Cystoseira indica) from Iran. J Appl Phycol 28:1323–1331. https://doi.org/10.1007/s10811-015-0645-y
Foo SC, Khoo KS, Ooi CW, Show PL, Khong N, Yusoff FM (2021) Meeting sustainable development goals: alternative extraction processes for fucoxanthin in algae. Front Bioeng Biotechnol 8:546067. https://doi.org/10.3389/fbioe.2020.546067
Fratianni A, Cinquanta L, Panfili G (2010) Degradation of carotenoids in orange juice during microwave heating. Lwt–food Sci Technol 43:867–871. https://doi.org/10.1016/j.lwt.2010.01.011
Fung A, Hamid N, Lu J (2013) Fucoxanthin content and antioxidant properties of Undaria pinnatifida. Food Chem 136:1055–1062. https://doi.org/10.1016/j.foodchem.2012.09.024
Jaeschke DP, Rech R, Marczak LDF, Mercali GD (2017) Ultrasound as an alternative technology to extract carotenoids and lipids from Heterochlorella luteoviridis. Bioresour Technol 224:753–757. https://doi.org/10.1016/j.biortech.2016.11.107
Kanazawa K, Ozaki Y, Hashimoto T, Swadesh K, Das S, Matsushita M, Hirano T, Okada AK, Mori N, Nakatsuka M (2008) Commercial-scale preparation of biofunctional fucoxanthin from waste parts of brown sea algae Laminaria japonica. Food Sci Technol Res 14:573–582. https://doi.org/10.3136/fstr.14.573
Kim SM, Jung YJ, Kwon ON, Cha KH, Um BH, Chung D, Pan CH (2012) A potential commercial source of fucoxanthin extracted from the microalga Phaeodactylum tricornutum. Appl Biochem Biotechnol 166:1843–1855. https://doi.org/10.1007/s12010-012-9602-2
Kong W, Liu N, Zhang J, Yang Q, Hua S, Song H, Xia C (2014) Optimization of ultrasound-assisted extraction parameters of chlorophyll from Chlorella vulgaris residue after lipid separation using response surface methodology. J Food Sci Technol 17:2006–2013. https://doi.org/10.1007/s13197-012-0706-z
Lalegerie F, Gager L, Stiger-Pouvreau V, Connan S (2020) The stressful life of red and brown seaweeds on the temperate intertidal zone: effect of abiotic and biotic parameters on the physiology of macroalgae and content variability of particular metabolites. In: Bourgougnon N (ed) Advances in Botanical Research—Seaweeds around the World: state of Art and Perspectives. Elsevier, London, United Kingdom, 247–287. https://doi.org/10.1016/bs.abr.2019.11.007
Lee IS, Lee HY, Kim HL, Ko KH, Chang HC, Kim IC (2008) Effect of metal ions on stabilization of Codium fragile pigments. Korean J Food Preserv 15:352–360
Liao J, Qu B, Liu D, Zheng N (2015) New method to enhance the extraction yield of rutin from Sophora japonica using a novel ultrasonic extraction system by determining optimum ultrasonic frequency. Ultrason Sonochem 27:110–116. https://doi.org/10.1016/j.ultsonch.2015.05.005
Liao J, Zheng N, Qu B (2016) An improved ultrasonic-assisted extraction method by optimizing the ultrasonic frequency for enhancing the extraction efficiency of lycopene from tomatoes. Food Anal Methods 9:2288–2298. https://doi.org/10.1007/s12161-016-0419-4
Liao J, Guo Z, Yu G (2021) Process intensification and kinetic studies of ultrasound-assisted extraction of flavonoids from peanut shells. Ultrason Sonochem 76:105661. https://doi.org/10.1016/j.ultsonch.2021.105661
Liao J, Peng B, Chu X, Yu G (2022a) Effects of process parameters on the extraction of total anthocyanins from purple sweet potatoes by ultrasound with wide frequency and its kinetics study. J Food Process Preserv 46:e16732. https://doi.org/10.1111/jfpp.16732
Liao J, Xue H, Li J, Peng L (2022b) Effects of ultrasound frequency and process variables of modified ultrasound-assisted extraction on the extraction of anthocyanin from strawberry fruit. Food Sci Technol. https://doi.org/10.1590/fst.20922
Lichtenthaler HK, Buschmann C (2001) Chlorophylls and carotenoids–measurement and characterisation by UV-VIS. Curr Protoc Food Anal Chem 1, F4.3.1-F4.3.8. https://doi.org/10.1002/0471142913.faf0403s01
Macías-Sánchez MD, Mantell C, Rodríguez M, de la Ossa EJM, Lubián LM, Montero O (2009) Comparison of supercritical fluid and ultrasound-assisted extraction of carotenoids and chlorophyll a from Dunaliella salina. Talanta 77:948–952
Marques R, Cruz S, Calado R, Lillebo A, Abreu H, Pereira R, Pitarma B, da Silva JM, Cartaxana P (2021) Effects of photoperiod and light spectra on growth and pigment composition of the green macroalga Codium tomentosum. J Appl Phycol 33:471–480
Matsuno T (2001) Aquatic animal carotenoids. Fish Sci 67:771–783. https://doi.org/10.3390/md9020278
Mittal R, Tavanandi HA, Mantri VA, Raghavarao KSMS (2017) Ultrasound assisted methods for enhanced extraction of phycobiliproteins from marine macro-algae, Gelidium pusillum (Rhodophyta). Ultrason Sonochem 38:92–103. https://doi.org/10.1016/j.ultsonch.2017.02.030
Mohibbullah M, Haque MN, Khan MNA, Park IS, Moon IS, Hong YK (2018) Neuroprotective effects of fucoxanthin and its derivative fucoxanthinol from the phaeophyte Undaria pinnatifida attenuate oxidative stress in hippocampal neurons. J Appl Phycol 30:3243–3252. https://doi.org/10.1007/s10811-018-1458-6
Nunes N, Leça JM, Pereira AC, Pereira V, Ferraz S, Barreto MC, Marques JC, de Carvalho MAAP (2019) Evaluation of fucoxanthin contents in seaweed biomass by vortex-assisted solid-liquid microextraction using high-performance liquid chromatography with photodiode array detection. Algal Res 42:101603. https://doi.org/10.1016/j.algal.2019.101603
Oliyaei N, Moosavi-Nasab M (2021) Ultrasound-assisted extraction of fucoxanthin from Sargassum angustifolium and Cystoseira indica brown algae. J Food Process Preserv. https://doi.org/10.1111/jfpp.15929
Osório C, Machado S, Peixoto J, Bessada S, Pimentel FB, Alves RC, Oliveira MBPP (2020) Pigments content (chlorophylls, fucoxanthin and phycobiliproteins) of different commercial dried algae. Separations 7:33. https://doi.org/10.3390/separations7020033
Parniakov O, Apicella E, Koubaa M, Barba FJ, Grimi N, Lebovka N, Pataro G, Ferrari G, Vorobiev E (2015) Ultrasound-assisted green solvent extraction of high-added value compounds from microalgae Nannochloropsis spp. Bioresour Technol 198:262–267. https://doi.org/10.1016/j.biortech.2015.09.020
Pasquet V, Chérouvrier JR, Farhat F, Thiéry V, Piot JM, Bérard JB, Kaas R, Serive B, Patrice T, Cadoret JP, Picot L (2011) Study on the microalgal pigments extraction process: Performance of microwave assisted extraction. Process Biochem 46:59–67. https://doi.org/10.1016/j.procbio.2010.07.009
Peng J, Yuan JP, Wu CF, Wang JH (2011) Fucoxanthin, a marine carotenoid present in brown seaweeds and diatoms: metabolism and bioactivities relevant to human health. Mar Drugs 9:1806–1828. https://doi.org/10.3390/md9101806
Pingret D, Fabiano-Tixier AS, Chemat F (2013) Degradation during application of ultrasound in food processing: a review. Food Control 31:593–606. https://doi.org/10.1016/j.foodcont.2012.11.039
Plaza M, Santoyo S, Jaime L, Avalo B, Cifuentes A, Reglero G, García-Blairsy Reina G, Señorans FJ, Ibáñez E (2012) Comprehensive characterization of the functional activities of pressurized liquid and ultrasound-assisted extracts from Chlorella vulgaris. LWT—Food Sci Technol, 46, 245–253 https://doi.org/10.1016/j.lwt.2011.09.024
Poojary MM, Barba FJ, Aliakbarian B, Donsì F, Pataro G, Dias DA, Juliano P (2016) Innovative alternative technologies to extract carotenoids from microalgae and seaweeds. Mar Drugs 22:214. https://doi.org/10.3390/md14110214
Raguraman V, Abraham L, Mubarakali D, Narendrakumar G, Thirugnanasambandam R, Kirubagaran R, Thajuddin N (2018) Unraveling rapid extraction of fucoxanthin from Padina tetrastromatica: Purification, characterization and biomedical application. Process Biochem 73:211–219. https://doi.org/10.1016/j.procbio.2018.08.006
Raji V, Loganathan C, Sadhasivam G, Kandasamy S, Poomani K, Thayumanavan P (2020) Purification of fucoxanthin from Sargassum wightii Greville and understanding the inhibition of angiotensin 1-converting enzyme: an in vitro and in silico studies. Int J Biol Macromol 1:696–703
Ritchie RJ (2008) Universal chlorophyll equations for estimating chlorophylls a, b, c, and d and total chlorophylls in natural assemblages of photosynthetic organisms using acetone, methanol, or ethanol solvents. Photosynthetica 46:115–126. https://doi.org/10.1007/s11099-008-0019-7
Roh MK, Uddin MS, Chun BS (2008) Extraction of fucoxanthin and polyphenol from Undaria pinnatifida using supercritical carbon dioxide with co-solvent. Biotechnol Bioprocess Eng 13:724–729. https://doi.org/10.1007/s12257-008-0104-6
Roselló-Soto E, Galanakis C, Brnčić M, Orlien V, Trujillo F, Mawson R, Knoerzer K, Tiwari B, Barba F (2015) Clean recovery of antioxidant compounds from plant foods, by-products and algae assisted by ultrasounds processing. Modeling approaches to optimize processing conditions. Trends Food Sci Technol 42:134–149. https://doi.org/10.1016/j.tifs.2015.01.002
Roy S (2020) Screening and partial characterization of natural antioxidants from seaweeds collected from Rameshwaram Southeast Coast of India. J Mar Sci Res Oceanogr 3:1–12
Shannon E, Abu-Ghannam N (2017) Optimisation of fucoxanthin extraction from Irish seaweeds by response surface methodology. J Appl Phycol 29:1027–1036. https://doi.org/10.1007/s10811-016-0983-4
Silva AFR, Abreu H, Silva AMS, Cardoso SM (2019) Effect of oven-drying on the recovery of valuable compounds from Ulva rigida, Gracilaria sp. and Fucus vesiculosus. Mar Drugs 17, 90.
Sivagnanam SP, Yin S, Choi JH, Park YB, Woo HC, Chun BS (2015) Biological properties of fucoxanthin in oil recovered from two brown seaweeds using supercritical CO2 extraction. Mar Drugs 29:3422–3442. https://doi.org/10.3390/md17020090
Soares AT, Marques Júnior JG, Lopes RG, Derner RB, Antoniosi Filho NR (2016) Improvement of the extraction process for high commercial value pigments from Desmodesmus sp. microalgae. J Brazilian Chem Soc 277:1083–1093. https://doi.org/10.5935/0103-5053.20160004
Sugawara T, Baskaran V, Tsuzuki W, Nagao A (2002) Brown algae fucoxanthin is hydrolyzed to fucoxanthinol during absorption by Caco-2 human intestinal cells and mice. J Nutr 132:946–951. https://doi.org/10.1093/jn/132.5.946
Sun Y, Ma G, Ye X, Kakuda Y, Meng R (2010) Stability of all-trans-β-carotene under ultrasound treatment in a model system: effects of different factors, kinetics and newly formed compound. Ultrason Sonochem 17:654–661. https://doi.org/10.1016/j.ultsonch.2009.12.005
Terasaki M, Kawagoe C, Ito A, Kumon H, Narayan B, Hosokawa M, Miyashita K (2017) Spatial and seasonal variations in the biofunctional lipid substances (fucoxanthin and fucosterol) of the laboratory-grown edible Japanese seaweed (Sargassum horneri Turner) cultured in the open sea. Saudi J Biol Sci 24:1475–1482. https://doi.org/10.1016/j.sjbs.2016.01.009
Verma P, Kumar M, Mishra G, Sahoo D (2017) Multivariate analysis of fatty acid and biochemical constitutes of seaweeds to characterize their potential as bioresource for biofuel and fine chemicals. Bioresour Technol 226:132–144. https://doi.org/10.1016/j.biortech.2016.11.044
Vimala T, Poonghuzhali TV (2013) Estimation of pigments from seaweeds by using acetone and DMSO. Int J Sci Res 4:1850–1854
Wang X, Li H, Wang F, Xia G, Liu H, Cheng X, Kong M, Liu Y, Feng C, Chen X, Wang Y (2017) Isolation of fucoxanthin from Sargassum thunbergii and preparation of microcapsules based on palm stearin solid lipid core. Front Mater Sci 11:66–74. https://doi.org/10.1007/s11706-017-0372-1
Wang J, Ma Y, Yang J, Jin L, Gao Z, Xue L, Hou L, Sui L, Liu J, Zou X (2019) Fucoxanthin inhibits tumour-related lymphangiogenesis and growth of breast cancer. J Cell Mol Med 23:2219–2229. https://doi.org/10.1111/jcmm.14151
Wiltshire KH, Boersma M, Möller A, Buhtz H (2000) Extraction of pigments and fatty acids from the green alga Scenedesmus obliquus (Chlorophyceae). Aquat Ecol 34:119–126. https://doi.org/10.1023/A:1009911418606
Yalçın S, Karakaş Ö, Okudan EŞ, Başkan KS, Çekiç SD, Apak R (2021) HPLC Detection and antioxidant capacity determination of brown, red and green algal pigments in seaweed extracts. J Chromatogr Sci 19:325–337. https://doi.org/10.1093/chromsci/bmaa107
Yang F, Wei Z, Long L (2021) Transcriptomic and physiological responses of the tropical reef calcified macroalga Amphiroa fragilissima to elevated temperature. J Phycol 57:1254–1265. https://doi.org/10.1111/jpy.13158
Zhang R, Yuen AKL, de Nys R, Masters AF, Maschmeyer T (2020) Step by step extraction of bio-actives from the brown seaweeds, Carpophyllum flexuosum, Carpophyllum plumosum, Ecklonia radiata and Undaria pinnatifida. Algal Res 52:102092. https://doi.org/10.1016/j.algal.2020.102092
Zhu J, Sun X, Chen X, Wang S, Wang D (2016) Chemical cleavage of fucoxanthin from Undaria pinnatifida and formation of apo-fucoxanthinones and apo-fucoxanthinals identified using LC-DAD-APCI-MS/MS. Food Chem 15:365–373. https://doi.org/10.1016/j.foodchem.2016.05.064
Zhu Z, Wu Q, Di X, Li S, Barba F, Koubaa M, Roohinejad S, Xiong X, He J (2017) Multistage recovery process of seaweed pigments: Investigation of ultrasound assisted extraction and ultra-filtration performances. Food Bioprod Process 104:40–47. https://doi.org/10.1016/j.fbp.2017.04.008
Zou TB, Jia Q, Li HW, Wang CX, Wu HF (2013) Response surface methodology for ultrasound-assisted extraction of astaxanthin from Haematococcus pluvialis. Mar Drugs 11:1644–1655. https://doi.org/10.3390/md11051644
Acknowledgements
We would like to thank the Croatian Government and the European Union through the European Regional Development Fund—the Competitiveness and Cohesion Operational Programme (KK.01.1.1.01) for funding The Scientific Centre of Excellence for Marine Bioprospecting—BioProCro. We thank also Donat Petricioli for the sample collection and identification.
Funding
This research was funded by the Croatian Government and the European Union through the European Regional Development Fund—the Competitiveness and Cohesion Operational Programme (KK.01.1.1.01), The Scientific Centre of Excellence for Marine Bioprospecting—BioProCro.
Author information
Authors and Affiliations
Contributions
A-MC took part in conceptualization, methodology, formal analysis, investigation, data curation, writing—original draft preparation. KA involved in methodology, formal analysis, investigation, data curation. DV took part in writing—review and editing, and supervision. ST took part in writing—review and editing, and supervision. PL involved in formal analysis and investigation. IJ involved in conceptualization, investigation, writing—original draft preparation, writing—review and editing, supervision.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cikoš, AM., Aladić, K., Velić, D. et al. Evaluation of ultrasound-assisted extraction of fucoxanthin and total pigments from three croatian macroalgal species. Chem. Pap. 77, 1545–1559 (2023). https://doi.org/10.1007/s11696-022-02524-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-022-02524-2