Abstract
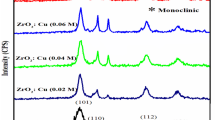
The present work demonstrates a facile preparation and characterization of polyvinyl pyrrolidine surfactant mediated coupled oxide semiconductor nanoparticles. 0, 10, 20, 30 wt% of Copper was added to Zinc Oxide by shallow chemical precipitation method for the coupled oxide nanoparticles preparation. The influence of PVP and Cu made a significant changes in the morphology of ZnO nanoparticles. FESEM and TEM analysis confirmed the morphology of the prepared samples. The more proportion of Cu concentration in ZnO made significant collapse from flakes to stacked nanoplates, oval shapes and nanorods. The XRD patterns confirm the coupled phases such as hexagonal and monoclinic structures. The effectual change in the bandgap energy was observed due to an increase in minority peaks of CuO rather than the majority peaks of ZnO nanoparticles. The DLE (Deep Level Emission) and violet emission were observed by PL analysis. The distribution of Cu level in ZnO lattice by incorporating Zn2+ sites was identified in XPS analysis. The prepared samples were tested for photodegradation of methylene blue dye under visible light irradiation for 120 min. The increase in photocatalytic degradation efficiency was observed due to the addition of a higher level of Cu.












Similar content being viewed by others
References
Abel MJ, Pramothkumar A et al (2019) Flake-like CuMn2O4 nanoparticles synthesized via co-precipitation method for photocatalytic activity. Phy B Con Matt 572:117–124. https://doi.org/10.1016/j.physb.2019.07.047
Ahlam Z, Be H (2019) Synthesis and characterization of Cu doped chromium oxide thin films. High Temp Mater Proc 38:806–812. https://doi.org/10.1515/htmp-2019-0037
Akin S, Liu Y et al (2018) Hydrothrmally processed CuCrO2 nanoparticles as an inorganic hole transporting material for low – cost perovskite solar cells with superior stability. J Mat Chem A 6:20327–20337. https://doi.org/10.1039/C8TA07368F
Alp E, Essgin H et al (2019) Synergetic activity enhancement in 2D CuO–Fe2O3 nanocomposites for the photodegradation of rhodamine B. Ceramic Int 45:9714–9178. https://doi.org/10.1016/j.ceramint.2019.01.258
Antonoglou O, Lafazanis K et al (2019) Biological relevance of CuFeO2 nanoparticles: antibacterial and anti- inflammatory activity, genotoxicity, DNA and protein interactions. Mat Sci Eng C 99:264–274. https://doi.org/10.1016/j.msec.2019.01.112
Ashraf W, Fatima T, Srivastava K et al (2019) Superior photocatalytic activity of tungsten disulfide nanostructures: role of morphology and defects. Appl Nanosci 9:1515–1529. https://doi.org/10.1007/s13204-019-00951-4
Bandekar G, Rajurkar NS et al (2014) Synthesis, characterization and photocatalytic activity of PVP stabilized ZnO and modified ZnO nanostructures. Appl Nanosci 4:199–208. https://doi.org/10.1007/s13204-012-0189-2
Chen C, Huang Y et al (2016) Controllable Synthesis of Cu doped CoO hierarchical structure for high performance lithium—ion battery. J Pow Sou 314:66–75. https://doi.org/10.1016/j.jpowsour.2016.02.085
Dandia A, Jain A et al (2013) CuFe2O4 nanoparticles as a highly efficient and magnetically recoverable catalyst for the synthesis of medicinally privileged spiropyrimidine scaffolds. RSC Adv 3:2924–2934. https://doi.org/10.1039/C2RA22477A
Dehghan A, Ganjali M (2019) Application of copper vanadate nanoparticles for removal of methylene blue from aqueous solution: kinetics, equilibrium and thermodynamic studies. Iran J Chem Chem Eng 38(6):83–92. https://doi.org/10.30492/ijcce.2019.32990
Dhamodharan P, Gobi R et al (2014) Synthesis and characterization of surfactants assisted Cu2+ doped ZnO nanocrystals. Spec Acta Part A 131:125–131. https://doi.org/10.1016/j.saa.2014.04.083
Diamandescu L, Cernea M, Trusca R et al (2018) Effects of a surfactant on the morphology and photocatalytic properties of polycrystalline Fe doped ZnO powders. J Phys Chem Solids 121:319–328. https://doi.org/10.1016/j.jpcs.2018.05.041
Ensafi A, Moosavifard SE et al (2018) Engineering onion – like nanoporous CuCo2O4 hollow spheres derived from bimetal—organic frameworks for high-performance asymmetric supercapacitors. J Mater Chem A 6:10497–10506. https://doi.org/10.1039/C8TA02819B
Fangsheng W et al (2017) Solid-state preparation of CuO/ZnO nanocomposites functional super capacitor electrodes and photocatalysts with enhanced photocatalytic properties. Int J Hyd Ene 42(51):30098–30108. https://doi.org/10.1016/j.ijhydene.2017.10.064
Ferin Fathima A, Jothi Mani R, Sakthipandi K et al (2020) Enhanced antifungal activity of pure and iron—doped ZnO nanoparticles prepared in the absence of reducing agents. J Inorg Orgnomet Polym 30:2397–2405. https://doi.org/10.1007/s10904-019-01400-z
Fernandes DM, Silva R, Custodio MA et al (2009) Synthesis and characterization of ZnO, CuO and a mixed Zn and Cu oxide. Mat Chem Phy 115(1):110–115. https://doi.org/10.1016/j.matchemphys.2008.11.038
Fu M, Li Y, Wu S et al (2011) Sol–gel preparation and enhanced photocatalytic performance of Cu doped ZnO nanoparticles. Appl Surf Sci 258:1587–1591. https://doi.org/10.1016/j.apsusc.2011.10.003
Gajendiran J, Rajendran V (2014) Synthesis and characterization of coupled semiconductor metal oxide (ZnO/CuO) nanocomposite. Mater Lett 116:311–313. https://doi.org/10.1016/j.matlet.2013.11.063
Gopi S, Rajan J (2018) Effectual role of abelmoschusesculentus (Okra) extract on morphology, microbial and photocatalytic activities of CdO tetrahedral clogs. J Inorg Organomet Polym 28:152–167. https://doi.org/10.1007/s10904-017-0695-5
Harish S, Archana J, Sabarinathan M et al (2017) Controlled structural and compositional characteristic of visible light active ZnO/CuO photocatalyst for the degradation of organic pollutant. Appl Sur Sci 418:103–112. https://doi.org/10.1016/j.apsusc.2016.12.082
Hosseini S, Abazari G (2014) Pure CuCr2O4 nanoparticles: synthesis, characterization and their morphological and size effects on the catalytic thermal decomposition of ammonium perchlorate. Solid State Sci 37:72–79. https://doi.org/10.1016/j.solidstatesciences.2014.08.014
Jayaprakash J, Srinivasan N et al (2015) Synthesis and characterization of cluster of grapes like pure and Zinc doped CuO nanoparticles by sol-gel method. Spec Acta A Mol Bio Spec 136:1803–1806. https://doi.org/10.1016/j.saa.2014.10.087
Lassoued A, Lassoued MS et al (2017) Structural, optical and morphological characterization of Cu-doped Fe2O3 nanoparticles synthesized through co-precipitation technique. J Mol Str 1148:276–281. https://doi.org/10.1016/j.molstruc.2017.07.051
Li Z, Xiong YJ et al (2003) Selected-control synthesis of ZnO nanowires and nanorods via a PEG-assisted route. Inorg Chem 42:8105–8109. https://doi.org/10.1021/ic034029q
Lim S, Hui Hong S, Ho S et al (2015) Synthesis of Al doped ZnO nanorods via microemulsion method and their application as a CO sensor j Mater. Sci Technol 31(6):639–644. https://doi.org/10.1016/j.jmst.2014.12.004
Logamani P, Rajeswari R et al (2018) Synthesis, characterization and application of Cu doped ZnO nanocatalyst for photocatalytic ozonation of textile dye and study of its reusability. Mater Res Express 5:115505. https://doi.org/10.1088/2053-1591/aadcdf
Lu P, Zhou W et al (2017) Abnormal room temperature ferromagnetism in CuO/ZnO nanocomposites via hydrothermal method. Applied Sur Sci 399:396–402. https://doi.org/10.1016/j.apsusc.2016.12.113
Mao L, Mohan S et al (2019) Delafossite CuMnO2 as an efficient bifunctional oxygen and hydrogen evolution reaction electrocatalyst for water splitting. J Eletrochem Soci 166(6):H233–H242. https://doi.org/10.1149/2.1181906jes
Norton D, Budai JD et al (1994) SrCuO2/(Sr, Ca) CuO2 superlattice growth by pulsed–laser deposition. Appl Phys Lett 65:2869–3287. https://doi.org/10.1063/1.112519
Perumal V, Sabarinathan A et al (2021) Facile synthesis of cationic surfactant-treated hierarchial nanorods SnO2 @ CdO nanocomposite photocatalyst for degradation of methylene blue under sunlight. https://doi.org/10.1016/j.matpr.2021.10.344
Pinho L, Mosquera MJ (2013) Photocatalytic activity of TiO2 – SiO2 nanocomposites applied to buildings: influence of particle size and loading. Appl Cata b: Envi 134–135:205–221. https://doi.org/10.1016/j.apcatb.2013.01.021
Poloju M, Jayababu N, Ramana Reddy MV (2018) Improved gas sensing performance of Al doped ZnO/ CuO nanocomposite based ammonia gas sensor. Mat Sci Eng B 277:61–67. https://doi.org/10.1016/j.mseb.2017.10.012
Prakash T, Jayaprakash R et al (2014) The effect of surfactants and co-reagents on the microstructure, morphology and optical properties of ZnO nanoparticles synthesized by microwave irradiation. Nano Sci Nano Tec Asia 4(1):49–59. https://doi.org/10.2174/2210681204666140716183120
Rackesh K, Indirajith R et al (2018) A high sensitivity isopropanol vapor sensor based on Cr2O3-SnO2 heterojunction nanocomposites via chemical precipitation route. J Nanosci Nanotec 18(8):5454–5460. https://doi.org/10.1166/jnn.2018.15396
Rajaboopathi S, Thambidurai S (2017) Green synthesis of seaweed surfactant based CdO–ZnO nanoparticles for better thermal and photocatalytic activity. Curr Appl Phys 17(12):1622–1638. https://doi.org/10.1016/j.cap.2017.09.006
Ruhua Z et al (2015) Ultraviolet photocatalytic degradation of methyl orange by nanostructured TiO2/ZnO heterojunctions. J Mater Chem A 3:6565–6574. https://doi.org/10.1039/C5TA00
Sabarinathan A, Jayaprakash R et al (2021) The augmentation of photocatalytic efficiency due to the transition effect between spherical shape and rod-like structure of Sn levels in ZnO nanoparticles. J Inorg Orgnomet Polym Mat 31:1480–1490. https://doi.org/10.1007/s10904-020-01769-2
Saha S, Abd hamid S (2017) CuZrO3 nanoparticles catalyst in aerobic oxidation of vanillyl alcohol. RSC Adv 7:9914–9925. https://doi.org/10.1039/C6RA26370D
Shanmugam V, Jeyaperumal KS (2018) Investigations of visible light driven Sn and Cu doped ZnO hybrid nanoparticles for photocatalytic performance and antibacterial activity. Appl Surf Sci 449:617–630. https://doi.org/10.1016/j.apsusc.2017.11.167
Sin JC, Lam SM (2016) Hydrothermal synthesis of europium-doped flower-like ZnO hierarchical structures with enhanced sunlight photocatalytic degradation of phenol. Mater Lett 182:223–226. https://doi.org/10.1016/j.matlet.2016.06.126
Sin JC, Lam SM et al (2013) Fabrication of erbium-doped spherical-like ZnO hierarchical nanostructures with enhanced visible light-driven photocatalytic activity. Mater Lett 91:1–4. https://doi.org/10.1016/j.matlet.2012.09.049
Singhal S, Namgyal T, Sharma R et al (2012) Cu doped ZnO nanoparticles: synthesis, structural and electrical properties. Physica B 407:1223–1226. https://doi.org/10.1016/j.physb.2012.01.103
Sini K, Biswarup S et al (2015) highly efficient photocatalytic degradation of organic dyes by Cu doped ZnO nanostructures. Phy Che Che Phy 17(38):25172–25181. https://doi.org/10.1039/C5CP01681A
Soleimani F, Salehi M et al (2017) Hydrothermal synthesis, structural and catalytic studies of CuBi2O4 nanoparticles. J Nano analysis 4(3):239–246. https://doi.org/10.22034/jna.2017.542053.1019
Subash B, Krishnakumar B et al (2012) Synthesis of Ce co-doped Ag–ZnO photocatalyst with excellent performance for NBB dye degradation under natural sunlight illumination. Catal Sci Technol 2:2319–2326. https://doi.org/10.1039/C2CY20254A
Toloman D, Pana O et al (2019) Photocatalytic activity of SnO2–TiO2 composite nanoparticles modified with PVP. J Coll Int Sci 542:296–307. https://doi.org/10.1016/j.jcis.2019.02.026
Wang Y, Li X, Lu G et al (2008) Synthesis and photo-catalytic degradation property of nanostructured ZnO with different morphology. Mater Lett 62:2359–2362. https://doi.org/10.1016/j.matlet.2007.12.019
Wang Z, Guo ZHuO et al (2020) Facile constructing ZnO/ZnCO3 heterojunction for high performance photocatalytic NO oxidation and reaction pathway study. J Mater Sci Mater Electron 31:4527–4534. https://doi.org/10.1007/s10854-020-03002-9
Wei SF, Lian JS, Jiang Q (2009) Controlling growth of ZnO rods by polyvinylpyrrolidone (PVP) and their optical properties. Appl Surf Sci 255:6978–6984. https://doi.org/10.1016/j.apsusc.2009.03.023
Woo Chae B, Amna T et al (2017) CeO2 – Cu2O composite nanofibers: synthesis, characterization photocatalytic and electrochemical application. Adv Pow Tech 28(1):230–235. https://doi.org/10.1016/j.apt.2016.09.010
Wu F, Wang X, Hu S et al (2017) Solid state preparation of CuO/ZnO nanocomposites for functional supercapacitor electrodes and photocatalysts with enhanced photocatalytic properties. Inter Jour Hydr Ene 42(51):30098–30108. https://doi.org/10.1016/j.ijhydene.2017.10.064
Yu H, Ru X, Tan H et al (2013) Cu doped V2O5 flowers as cathode material for high-performance lithium ion batteries. Nanoscale 5:4937–4943. https://doi.org/10.1039/C3NR00548H
Zhai YJ, Li JH, Fang X, Chen XY, Fang F et al (2014) Preparation of cadmium-doped zinc oxide nanoflowers with enhanced photocatalytic activity. Mater Sci Semicond Process 26:225–230. https://doi.org/10.1016/j.mssp.2014.04.023
Zhang Yu, Ji Y, Li J, Liu H et al (2017) Hierarchical zinc–copper oxide hollow microspheres as active rochow reaction catalysts: the formation and effect of charge transferable interfaces. J Catal 348:233–245. https://doi.org/10.1016/j.jcat.2017.02.030
Zhao R, Li K, Wang Z et al (2018) Gas-sensing performances of Cd-doped ZnO nanoparticles synthesized by a surfactant-mediated method for n-butanol gas. J Phys Chem Solids 112:43–49. https://doi.org/10.1016/j.jpcs.2017.08.039
Zhu C, Li Y et al (2013) Electrospinning direct preparation of SnO2/Fe2O3 heterojunction nanotubes as an efficient visible–light photocatalyst. J Alloy Comp 575:333–338. https://doi.org/10.1016/j.jallcom.2013.05.172
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sabarinathan, A., Jayaprakash, R. & Robert, R. The shape transformation from nanoflakes into nanorods of coupled nanophase CuO–ZnO on higher-level accretion of Cu in ZnO. Chem. Pap. 76, 4481–4496 (2022). https://doi.org/10.1007/s11696-022-02177-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-022-02177-1