Abstract
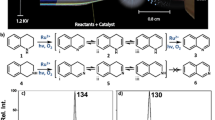
We had developed a visible light-catalyzed green synthesis method of 3-position substituted 3,4-dihydroisoquinoline-1(2H)-one compounds without metal participation, and initially proposed the reaction mechanism of visible light-catalyzed free radical involvement. In order to further verify the reliability of the proposed mechanism, density functional theory was used to analyze the structures involved in the reaction process. The theoretical results rationalized the experimental observations well and provided deeper insight into the mechanism. In addition, the photocatalytic reaction was further recognized by measuring the emission spectrum and absorption spectrum of the reaction system.




Similar content being viewed by others
Abbreviations
- SET:
-
Single-electron transfer
- DFT:
-
Density functional theory
- ZPT:
-
Zero point energy
- IRC:
-
Intrinsic reaction coordinate
- TS:
-
Transition state
- r.t. :
-
Room temperature, K
- t :
-
Time, h
References
Biczysko M, Panek P, Scalmani G, Bloino J, Barone V (2010) Harmonic and anharmonic vibrational frequency calculations with the double-hybrid B2PLYP method: analytic second derivatives and benchmark studies. J Chem Theory Comput 6:2115–2125. https://doi.org/10.1021/ct100212p
Chen JR, Hu XQ, Lu LQ, Xiao WJ (2016a) Exploration of visible-light photocatalysis in heterocycle synthesis and functionalization: reaction design and beyond. Acc Chem Res 49:1911–1923. https://doi.org/10.1021/acs.accounts.6b00254
Chen JR, Hu XQ, Lu LQ, Xiao WJ (2016b) Visible light photoredox-controlled reactions of N-radicals and radical ions. Chem Soc Rev 45:2044–2056. https://doi.org/10.1039/c5cs00655d
Choi GJ, Knowles RR (2015) Catalytic alkene carboaminations enabled by oxidative proton-coupled electron transfer. J Am Chem Soc 137:9226–9229. https://doi.org/10.1021/jacs.5b05377
Fukui K (1981) The path of chemical reactions - the IRC approach. Acc Chem Res 14:363–368. https://doi.org/10.1021/ar00072a001
Glushkov VA, Shklyaev YV (2001) Synthesis of 1(2H)-isoquinolones. khimiya geterotsiklicheskikh soedinenii. Chem Heteroc Compound. https://doi.org/10.1023/A:1011958810129
Gonzalez C, Schlegel HB (1989) An improved algorithm for reaction path following. J Chem Phys 90:2154–2161. https://doi.org/10.1063/1.456010
Grunewald GL, Caldwell TM, Li QF, Criscione KR (1999) Synthesis and evaluation of 3-trifluoromethyl-7-substituted-1,2,3,4-tetrahydroisoquinolines as selective inhibitors of phenylethanolamine N-methyltransferase versus the alpha(2)-adrenoceptor. J Med Chem 42:3315–3323. https://doi.org/10.1021/jm980734a
Guo XY, Cao QY, Tang YM, Liang QL (2018) Simple synthesis and anti-inflammatory activities of spanrstolonin B derivatives. Phytochem Lett 24:158–162. https://doi.org/10.1016/j.phytol.2018.02.011
Hari DP, Koenig B (2013) The photocatalyzed meerwein arylation: classic reaction of aryl diazonium salts in a new light. Angew Chem-Int Ed 52:4734–4743. https://doi.org/10.1002/anie.201210276
Hu XQ, Chen JR, Wei Q, Liu FL, Deng QH, Beauchemin AM, Xiao WJ (2014) Photocatalytic generation of N-centered hydrazonyl radicals: a strategy for hydroamination of beta, gamma-unsaturated hydrazones. Angew Chem-Int Ed 53:12163–12167. https://doi.org/10.1002/anie.201406491
Hu XQ, Qi X, Chen JR, Zhao QQ, Wei Q, Lan Y, Xiao WJ (2016) Catalytic N-radical cascade reaction of hydrazones by oxidative deprotonation electron transfer and TEMPO mediation. Nat Commun 7:11188. https://doi.org/10.1038/ncomms11188
Kung P-P et al (2018) Optimization of orally bioavailable enhancer of zeste homolog 2 (EZH2) inhibitors using ligand and property-based design strategies: identification of development candidate (R)-5,8-dichloro-7-(methoxy(oxetan-3-yl)methyl)-2-((4-methoxy-6-methyl-2 -oxo-1,2-dihydropyridin-3-yl)methyl)-3,4-dihydroisoquinolin-1(2H)-one. J Med Chem 61:650–665. https://doi.org/10.1021/acs.jmedchem.7b01375
Li X, Duan R, Wang Y, Qu LB, Li Z, Wei D (2019a) Insights into N-heterocyclic carbene-catalyzed oxidative alpha-C(sp(3))-H activation of aliphatic aldehydes and cascade 2+2 cycloaddition with ketimines. J Org Chem 84:6117–6125. https://doi.org/10.1021/acs.joc.9b00295
Li X, Li SJ, Wang Y, Wang Y, Qu LB, Li Z, Wei D (2019b) Insights into NHC-catalyzed oxidative -C(sp(3))-H activation of aliphatic aldehydes and cascade 2+3 cycloaddition with azomethine imines. Catal Sci Technol 9:2514–2522. https://doi.org/10.1039/c9cy00526a
Michal M, Fabiana F, Axel J (2014) On the mechanism of photocatalytic reactions with eosin Y. Beilstein J Org Chem 10:981–989. https://doi.org/10.3762/bjoc.10.97
Miller DC, Choi GJ, Orbe HS, Knowles RR (2015) Catalytic olefin hydroamidation enabled by proton-coupled electron transfer. J Am Chem Soc 137:13492–13495. https://doi.org/10.1021/jacs.5b09671
Narayanam JMR, Stephenson CRJ (2011) Visible light photoredox catalysis: applications in organic synthesis. Chem Soc Rev 40:102–113. https://doi.org/10.1039/b913880n
Natsugari H et al (1995) Novel, potent, and orally-active substance-p antagonists - synthesis and antagonist activity of n-benzylcarboxamide derivatives of pyrido 3,4-b pyridine. J Med Chem 38:3106–3120. https://doi.org/10.1021/jm00016a014
Ortega R et al (2009) Synthesis, binding affinity and SAR of new benzolactam derivatives as dopamine D-3 receptor ligands. Bioorg Med Chem Lett 19:1773–1778. https://doi.org/10.1016/j.bmcl.2009.01.067
Paine HA et al (2015) Exploration of the nicotinamide-binding site of the tankyrases, identifying 3-arylisoquinolin-1-ones as potent and selective inhibitors in vitro. Bioorg Med Chem 23:5891–5908. https://doi.org/10.1016/j.bmc.2015.06.061
Prier CK, Rankic DA, MacMillan DWC (2013) Visible light photoredox catalysis with transition metal complexes: applications in organic synthesis. Chem Rev 113:5322–5363. https://doi.org/10.1021/cr300503r
Qiao Y, Zhao J, Chang J, Wei D (2019) Insights into the oxidative palladium-catalyzed regioselective synthesis of 3-arylindoles from N-Ts-anilines and styrenes: a computational study. ChemCatChem 11:780–789. https://doi.org/10.1002/cctc.201801531
Rassolov VA, Pople JA, Ratner MA, Windus TL (1998) 6-31G* basis set for atoms K through Zn. J Chem Phys 109:1223–1229
Rassolov VA, Ratner MA, Pople JA, Redfern PC, Curtiss LA (2001) 6-31G* basis set for third-row atoms. J Comp Chem 22:976–984
Shen K, Wang Q (2016) Copper-catalyzed aminotrifluoromethylation of alkenes: a facile synthesis nnnof CF3-containing lactams. Org Chem Front 3:222–226. https://doi.org/10.1039/c5qo00353a
Shi L, Xia W (2012) Photoredox functionalization of C-H bonds adjacent to a nitrogen atom. Chem Soc Rev 41:7687–7697. https://doi.org/10.1039/c2cs35203f
Tazawa A, Ando J, Ishizawa K, Azumaya I, Hikawa H, Tanaka M (2018) An efficient route to N-alkylated 3,4-dihydroisoquinolinones with substituents at the 3-position. RSC Adv 8:6146–6151. https://doi.org/10.1039/c7ra13627g
Tucker JW, Stephenson CRJ (2012) Shining light on photoredox catalysis: theory and synthetic applications. J Org Chem 77:1617–1622. https://doi.org/10.1021/jo202538x
Vishal S, Singh PP (2017) Eosin Y catalysed photoredox synthesis: a review. RSC Adv 7:31377–31392. https://doi.org/10.1039/c7ra05444k
Wang Y, Qu L-B, Wei D (2019a) Prediction on the origin of selectivities in base-controlled switchable NHC-catalyzed transformations. Chem-an Asian J 14:293–300. https://doi.org/10.1002/asia.201801583
Wang Y, Wu Q-Y, Lai T-H, Zheng K-J, Qu L-B, Wei D (2019b) Prediction on the origin of selectivities of NHC-catalyzed asymmetric dearomatization (CADA) reactions. Catal Sci Technol 9:465–476. https://doi.org/10.1039/c8cy02238k
Xie J, Yuan X, Abdukader A, Zhu C, Ma J (2014) Visible-light-promoted radical C-H trifluoromethylation of free anilines. Org Lett 16:1768–1771. https://doi.org/10.1021/ol500469a
Xuan J, Zhang Z-G, Xiao WJ (2015) Visible-light-induced decarboxylative functionalization of carboxylic acids and their derivatives. Angew Chem-Int Ed 54:15632–15641. https://doi.org/10.1002/anie.201505731
Yoon TP, Ischay MA, Du J (2010) Visible light photocatalysis as a greener approach to photochemical synthesis. Nat Chem 2:527–532. https://doi.org/10.1038/nchem.687
Yu X, Zhou F, Chen J, Xiao W (2017) Visible light photocatalytic N-radical-based intramolecular hydroamination of benzamides. Acta Chim Sinica 75:86–91. https://doi.org/10.6023/a16070367
Yuan YC, Liu HL, Hu XB, Wei Y, Shi M (2016) Visible-light-induced trifluoromethylation of isonitrile-substituted methylenecyclopropanes: facile access to 6-(trifluoromethyl)-7,8-dihydrobenzo k phenanthridine derivatives. Chem-a Eur J 22:13059–13063. https://doi.org/10.1002/chem.201602920
Zhang QC, Li X, Wang X, Li SJ, Qu LB, Lan Y, Wei D (2019) Insights into highly selective ring expansion of oxaziridines under Lewis base catalysis: a DFT study. Org Chem Front 6:679–687. https://doi.org/10.1039/c8qo01370e
Zhao Y, and D. G. Truhlar, “The M06 suite of density functionals for main group thermochemistry, thermochemical kinetics, noncovalent interactions, excited states, and transition elements: two new functionals and systematic testing of four M06-class functionals and 12 other functionals,” Theor. Chem. Acc., 120 (2008) 215–41.
Zhao Y, Xia W (2018) Recent advances in radical-based C-N bond formation via photo-/electrochemistry. Chem Soc Rev 47:2591–2608. https://doi.org/10.1039/c7cs00572e
Zhao QQ, Hu XQ, Yang MN, Chen JR, Xiao W-J (2016a) A visible-light photocatalytic N-radical cascade of hydrazones for the synthesis of dihydropyrazole-fused benzosultams. Chem Commun 52:12749–12752. https://doi.org/10.1039/c6cc05897c
Zhao XZ et al (2016b) 6,7-Dihydroxyisoindolin-1-one and 7,8-dihydroxy-3,4-dihydroisoquinolin-1(2H)-one based HIV-1 integrase inhibitors. Curr Top Med Chem 16:435–440. https://doi.org/10.2174/1568026615666150813150058
Zhou D-G, Wang P (2019) Mechanisms of the reaction between benzonitrile and 4-octyne catalyzed by Ni(PMe3)2: A theoretical investigation. J Phys Org Chem 32:3932. https://doi.org/10.1002/poc.3932
Zou G, Wang X (2017) Visible-light induced di- and trifluoromethylation of N-benzamides with fluorinated sulfones for the synthesis of CF2H/CF3-containing isoquinolinediones. Org Biomol Chem 15:8748–8754. https://doi.org/10.1039/c7ob02226c
Zou S, Geng S, Chen L, Wang H, Huang F (2019) Visible light driven metal-free intramolecular cyclization: a facile synthesis of 3-position substituted 3,4-dihydroisoquinolin-1(2H)-one.17:380–387. Organic & Biomolecular Chemistry doi:https://doi.org/10.1039/C8OB02560F
Acknowledgements
We gratefully acknowledge the funding of the research group and the help provided by Dr. Pan Li in theoretical calculations.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zou, S., Pan, L., Xue, T. et al. Metal-free visible-light-catalyzed synthesis of 3-methyl-3,4-dihydroisoquinolin-1(2H)-one: mechanism, DFT calculation and optical properties. Chem. Pap. 75, 4069–4074 (2021). https://doi.org/10.1007/s11696-021-01639-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-021-01639-2