Abstract
Negative impact of radicals on humans and animals is responsible for growing research interest in antioxidant properties of substances, which protect living organisms from the damaging influence of these reactive species. Among numerous methods for antioxidant activity estimation, DPPH and ABTS are the most popular and commonly used ones due to their ease, speed, sensitivity and the usage of stabile radicals. The paper shows and discusses the antioxidant properties of a few compounds exhibiting different extinction of the wavelength used to monitor colored radical depletion in spectrophotometric measurements—e.g. 516 and 744 nm in DPPH and ABTS assays, respectively. As results from the presented data, the difference between true and spectrophotometrically estimated IC50 values for colored antioxidants depends not only on extinction coefficient of the monitored wavelength by the antioxidant but also on its remaining concentration in the measuring system. The presented results seems to be helpful for the researches examining coloured antioxidants and plant extracts containing substances having spectra in Vis region.
Graphical Abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Antioxidant activity is defined as the ability of given compounds or mixtures to reduce pro-oxidants or reactive species, including free radicals. Many methods are available for its estimation (Antolovich et al. 2002; Moon and Shibamoto 2009; Alam et al. 2013; Lὀpez-Alarcὀn and Denicola 2013; Apak et al. 2016). Among them, two colorimetric methods, DPPH and ABTS (employing 2,2′-diphenylpicrylhydrazyl and 2,2′-azinobis (3-ethylbenzothiazoline-6-sulfonic acid) diammonium salt), seem to be the most popular and willingly used ones due to their measurement simplicity, short experimental time and the employment of the inexpensive spectrophotometer (Mishra et al. 2012). In both methods the antioxidant activity of an examined antioxidant is determined in terms of absorbance changes of artificial, stable and colored radicals—DPPH· or ABTS·+ (Prior et al. 2005; Floegel et al. 2011; Can and Baltas 2016). The reduction degree of colored radical during its reaction with antioxidant is measured at 515–517 nm in the case of the DPPH method and at 734–744 in the ABTS assay.
As results from the literature (Arnao 2000; Pérez-Jiménez et al. 2008; Paśko et al. 2009; Shalaby and Shanab 2013; Gayosso-García Sancho et al. 2013), the mentioned methods should not be applied for the estimation of antioxidant properties of substances which themselves (or their oxidation products formed during neutralization of radicals) absorb in the range of the monitored wave length. The presence of such compounds in the measuring system falsifies the changes of absorbance resulting from the concentration decrease of the colored radical and makes the estimation of the real antioxidant activity of the examined compounds impossible. In such cases:
-
Another wave length from the spectrum range of the colored radical (if possible) should be monitored in spectrophotometric measurements, or.
-
The concentration changes of the radical should be monitored after its prior chromatographic separation from other components present in the measuring system.
The paper shows and discusses the antioxidant properties estimated by the DPPH and ABTS methods of a few compounds which exhibit different extinction at 516 nm and negligible extinction at 744 nm. The antioxidant activities obtained by these two spectrophotometric methods are compared with those determined from the radicals concentration changes in the measuring system using HPLC. Such comparison seems to be valuable as there are many examples in literature concerning inappropriate application of DPPH and ABTS methods to the estimation of antioxidant properties of colored antioxidants, including colored natural components of food and its colored artificial additives (Sánchez-Moreno et al. 1999; Paixão et al. 2007; Roussis et al. 2008; Ramos-Esudero et al. 2012; Castro-López et al. 2016).
Experimental
Reagents and equipment
2,2′-diphenylpicrylhydrazyl (DPPH), buthylhydroxyanisole (BHA), buthylhydroxytoluene (BHT), 6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid (Trolox), 2,2′-azinobis (3-ethylbenzothiazoline-6-sulfonic acid) diammonium salt (ABTS), potassium persulfate (di-potassium peroxdisulfate), cyanidin 3-glucoside, quercetin, alizarin red S, methanol were purchased from Sigma Aldrich (Poznań, Poland). The pigments: E155 (disodium4-[(2E)-2-[(5Z)-3-(hydroxymethyl)-2,6-dioxo-5-[(4-sulfonatonaphthalen-1-yl) hydrazinylidene]-1-cyclohex-3-enylidene]hydrazinyl]naphthalene-1-sulfonate), thymol blue and E132 (3,3′-dioxo-2,2′-bisindolyden-5,5′-disulfonic acid disodium salt) were purchased from Food Colours (Piotrków Trybunalski, Poland). Water was purified on a Milli-Q system from Millipore (Bedford, MA, USA).
Measurements of antioxidant activity
The antioxidant activities of the examined compounds were estimated on the basis of unreacted DPPH· or ABTS·+ concentration in the measuring system. The changes of both radicals were monitored spectrophotometrically and chromatographically.
Spectrophotometric measurements
DPPH method
The concentration of the unreacted DPPH radical after its reaction with the examined antioxidants was estimated by the slightly modified Brand-Williams method (Brand-Williams et al. 1995). DPPH· solution (c = 6 × 10−5 molL−1 with initial absorbance 0.7 ± 0.05 at 516 nm) (2.94 mL) was mixed in a 4 mL test tube with methanol antioxidants solution (60 µL). The mixture was vigorously shaken for 30 s and then poured into an optical glass cuvette (1 cm × 1 cm × 3.5 cm) and immediately placed in a spectrophotometer. The decrease in absorbance at 516 nm was registered in a continuous manner for 60 min with a UV Probe-1800 spectrophotometer (Shimadzu, Kyoto, Japan). To zero the spectrophotometer, methanol without radical and the antioxidant was used.
ABTS method
Before the estimation of antioxidant properties of examined compounds by ABTS method ABTS cation radical was prepared according to Nenandis et al. (2004). For this reason 5 mL of 7 mmol L−1 aqueous ABTS solution was mixed with 88 μL of 140 mmol L−1 potassium persulfate (K2S2O8). The mixture was incubated in the dark for 16 h and diluted with methanol until the absorbance value at 744 nm equaled 0.7 (final ABTS concentration c = 4.29 × 10−5 mol L−1). 2940 µL of the prepared ABTS cation radical solution was mixed with methanolic solution of chosen antioxidant (60 µL) in a 4 mL test tube. The mixture was vigorously shaken for 30 s, transferred into an optical glass cuvette (1 cm × 1 cm × 3.5 cm) and immediately placed in a spectrophotometer. The unreacted ABTS cation radical was estimated in the same way and with the same equipment as the DPPH radical. In this case the decrease of absorbance at 744 nm was monitored.
The inhibition percent of DPPH· or ABTS·+ was calculated according to the following equation:
where A 0 and A 60 are the values of DPPH· or ABTS·+ absorbance at 0 and 60 min of the radical neutralization reaction, respectively.
HPLC measurements
The Varian Pro Star model 210/215 with the Pro Star 325 UV–Vis detector, Rheonyne 7725 sample injector (20 μL loop) and the ODS column (Microsorb MV 100 C18, 25 cm × 4.6 mm i.d.) was used for the chromatographic estimation of unreacted DPPH· or ABTS·+ in the measuring system. Its components were separated by isocratic elution employing mobile phase composed of methanol/water 80/20 v/v. The measuring systems for HPLC analysis were prepared in the same way as those for spectrophotometry, i.e. 2940 µL of DPPH radical solution or ABTS cation radical solution was mixed with methanolic solution of chosen antioxidant (60 µL) in a 4 mL test tube. The mixture was vigorously shaken for 30 s, incubated for 60 min and subjected to HPLC analysis. The concentrations of unreacted DPPH· or ABTS·+ were calculated from chromatograms recorded at 516 and 744 nm, respectively.
The inhibition percent of DPPH· or ABTS·+ was calculated according to the following equation:
where P S and P R are areas of DPPH· or ABTS·+ peaks from the chromatograms relating to the measuring system containing the examined antioxidant and without it, respectively.
Statistical analysis
The results are presented as the mean values ± SD. In order to determine the measurements reproducibility, each antioxidant activity assay was repeated three times. The RSD values of all measurements were smaller than 10%. P < 0.01 was assumed as statistical difference between the experimental points.
Results and discussion
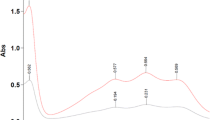
The UV–Vis spectra of the applied antioxidants and UV–Vis spectra of the DPPH and ABTS radicals, the concentration of which was monitored to determine antioxidant properties of the antioxidants are presented in Fig. 1. As results from the figure, none of the applied antioxidants absorbs light near 744 nm wavelength. Considering their absorption ability of 516 nm wavelength, the examined antioxidants can be divided into two groups. The first one consists of four antioxidants (BHA, BHT, Trolox and quercetin) which do not absorb 516 nm wavelength at all. Five other components—cyanidin, alizarin, thymol blue, E155 and E132—constitute the second group of antioxidants absorbing the mentioned wavelength. It is worth noting, however, that the extinction of 516 nm wavelength by these compounds varies (see the last column of Table 1). The greatest molar extinction of 516 nm is shown by cyanidin whereas the smallest by E132.
IC50 parameter is most frequently applied to express the antioxidant activity of compounds or mixtures by the DPPH and ABTS methods (Antolovich et al. 2002; Magalhăes et al. 2008; Dawidowicz et al. 2012). IC50 values for the examined antioxidants, estimated by monitoring absorbance changes of the measuring system at 516 nm (spectrophotometric measurements) and by monitoring DPPH concentration changes in the same system by chromatographic analysis, are collected in the second and third column of Table 1. As results from the data, the IC50 values estimated spectrophotometrically and chromatographically are almost the same only for BHA, BHT, Trolox and quercetin (i.e. for the antioxidants of the first group).
As to the components of the second group, IC50 values from spectrophotometric and chromatographic measurements could be calculated only for cyanidin and alizarin. It should be stressed, however, that spectrophotometrically estimated IC50 values for these two compounds are greater than those calculated from the chromatographic measurements. Thus, judging by the spectrophotometric data, the antioxidant properties of these compounds are worse than those established by monitoring the DPPH radical concentration by chromatography. Such discrepancies are explicable if one takes into account that some amount of the antioxidant absorbing 516 nm wavelength may still remain in the measuring system at the moment of measuring the DPPH radical concentration spectrophotometrically. In consequence, the remaining antioxidant precludes the estimation of the true concentration of the DPPH radicals in the measuring system. The greater difference between the spectrophotometrically and chromatographically estimated IC50 values for alizarin than for cyanidin, despite a significantly lower extinction of 516 nm wavelength by the former (see Table 1), indicate that there is another factor, beside the excitation value, responsible for the observed discrepancies.
It must be remembered that the IC50 values for given antioxidants are calculated from the relation illustrating the influence of antioxidant concentration on DPPH inhibition percent. Such dependencies determined chromatographically (solid lines) and spectrophotometrically (dotted lines), assuming that the absorbance at 516 nm in spectrophotometric measurements corresponds only with the DPPH radical concentration, are presented in Figs. 2 and 3. As results from Fig. 2, the run of the dependencies between inhibition percent and antioxidant concentration, determined spectrophotometrically and chromatographically, are the same for antioxidants of the first examined group, which do not exhibit any extinction at 516 nm. The same run of both dependencies confirms the reliability and utility of spectrophotometric monitoring of the DPPH radical concentration changes for the estimation of the antioxidant ability of these compounds.
For the antioxidants in the second group (see Fig. 3), the run of the dependencies between inhibition percent and antioxidant concentration determined in spectrophotometric and chromatographic measurements is different. The inhibition % values determined spectrophotometrically for these antioxidants are either significantly lower than those calculated from the chromatographic data (cyanidin, alizarin, E132) or even have negative values (E155, thymol blue). The observed discrepancies in the run of the curves obtained from the spectrophotometric and chromatographic measurements and in the shape of the curves obtained from the spectrophotometric measurements can be explained by analyzing the extinction of 516 nm wavelength by these antioxidants (see Table 1) and their residue in the measuring systems after the reactions with the DPPH radicals (see Table 2).
The smallest difference in the run of dependencies in Fig. 3 is observed for cyanidin despite its greatest molar extinction of 516 nm wavelength (see the last column of Table 1). Of all the examined colored antioxidants, this compound exhibits the greatest antioxidant activity and its remaining amount in the measuring system after the reaction with the DPPH radicals is relatively low: between 2.67 and 5.87% (see Table 2) at the concentration equal IC50 = 0.138 mg mL−1 (see HPLC data in Table 1). It was also observed that the run of the dependencies in Fig. 3 for alizarin and E132 showed even greater differences. Admittedly molar extinctions of 516 nm wavelength for these two antioxidants are the lowest in the second group (see the last column of Table 1); however, their residue in the measuring system after the reaction with the DPPH radicals is significantly greater. At concentration equal IC50 = 0.778 mg mL−1 for alizarin and IC50 = 4.746 mg mL−1 for E132 (see HPLC data in Table 1), their residues are between 14.07 and 21.50% and 67.07–69.52%, respectively (see Table 2).
Table 1 does not contain spectrophotometrically estimated IC50 values for three examined compounds of the second group: E132, thymol blue and E155. Although the run of the dependencies between inhibition percent and antioxidant concentration estimated spectrophotometrically for E132 has the classical shape, the curve is situated in the region of low inhibition % (below 30%). The extinction value of 516 nm for this weak antioxidant is the lowest in the second group (see Table 1), however, its residue in the measuring system is considerably larger. The concentration of the remaining E132 at the estimated IC50 = 4.746 mg mL−1 (see HPLC data in Table 1) is between 67.07 and 69.52% (see Table 2). The inflated absorbance of the reaction mixture, reflected by a low run of the dependencies between inhibition percent and antioxidant concentration, simulates small DPPH radical scavenging.
Quite different shape of the dependencies between spectrophotometrically estimated inhibition percent and antioxidant concentration is observed for thymol blue and E155 (see Fig. 3d, e). The negative values of the inhibition % for these antioxidants are due to greater absorption of 516 nm wavelength by the reaction mixtures than by the initial DPPH radical solution. This results from the high extinction of 516 nm by these antioxidants and from their significant concentration in the measuring system at the moment of spectrophotometric reading (see Tables 1, 2). In the case of E155, which exhibits higher extinction of 516 nm wavelength and more of it remains in the measuring system than of thymol blue, the number of spectrophotometrically estimated negative values of inhibition percent is greater.
IC50 values for the examined antioxidants, estimated by monitoring absorbance changes of the measuring system at 744 nm (spectrophotometric measurements) and by monitoring ABTS cation radical concentration changes in the same system by chromatographic analysis, are gathered in Table 3.
As results from the table, the IC50 values established for individual antioxidants using both monitoring ways of ABTS cation radical concentration changes are almost the same. It should be noticed, however, that in these experiments the applied monitored wavelength (744 nm) in spectrophotometric measurements is outside the spectra of examined antioxidants. Hence, the presence of antioxidant residues in the measuring system do not influence the estimation of true ABTS concentration.
Conclusions
Negative influence of the reactive oxygen species on living organisms and on the stability of food products is responsible for a significant interest in substances exhibiting antioxidant properties and in the methods of estimating them. In most such methods, the ability of antioxidants to trap free radicals is measured by the reaction kinetics between the examined antioxidant and the radical. The methods applying chromogen compounds are commonly used due to their ease, speed and sensitivity most popular being those employing the stable DPPH· or ABTS·+. As mentioned in Introduction, these methods should not be applied for the estimation of antioxidant properties of substances which absorb in the range of monitored wave length.
As results from the presented data:
-
ABTS and DPPH is adequate for measuring antioxidant characteristics when the spectrum of examined antioxidant or real biological system does not coincide with the wavelength used to monitor colored radical depletion;
-
if the examined antioxidant absorbs the wavelength used to monitor radical concentration changes, the difference between true and spectrophotometrically estimated IC50 values depends not only on its extinction coefficient of the monitored wavelength but also on its residue concentration in the measuring system;
-
it is possible to apply spectrophotometric measurements for the estimation of the antioxidant properties of compounds absorbing the monitored wavelength provided that the examined antioxidant, reacting with colored radicals, undergoes total depletion and that the reaction products allow to monitor the neutralization reaction of DPPH or ABTS radicals.
References
Alam MN, Bristi NJ, Rafiquzzman M (2013) Review on in vivo and in vitro methods evaluation of antioxidant activity. Saudi Pharm J 21:143–152. doi:10.1016/j.jsps.2012.05.002
Antolovich M, Prenzler PD, Patsalides E, McDonald S, Robards K (2002) Methods for testing antioxidant activity. Analyst 127:183–198. doi:10.1039/B009171P
Apak R, Özyürek M, Güçlü K, Çapanoğlu E (2016) Antioxidant activity/capacity measurement. 1. Classification, physicochemical principles, mechanisms, and electron transfer (ET)-based assays. J Agric Food Chem 64:997–1027. doi:10.1021/acs.jafc.5b04739
Arnao MB (2000) Some methodological problems in the determination of antioxidant activity using chromogen radicals: a practical case. Trends Food Sci Technol 11:419–421. doi:10.1016/S0924-2244(01)00027-9
Brand-Williams W, Cuvelier ME, Berset C (1995) Use of a free radical method to evaluate antioxidant activity. LWT Food Sci Technol 28:25–30. doi:10.1016/S0023-6438(95)80008-5
Can Z, Baltas N (2016) Bioactivity and enzyme inhibition properties of Stevia rebaudiana. Curr Enzym Inhib 12:188–194. doi:10.2174/1573408012666160402001925
Castro-López C, Sánchez-Alejo EJ, Saucedo-Pompa S, Rojas R, Aranda-Ruiz J, Martínez-Avila GCG (2016) Fluctuations in phenolic content, ascorbic acid and total carotenoids and antioxidant activity of fruit beverages during storage. Helyion. doi:10.1016/j.heliyon.2016.e00152
Dawidowicz AL, Wianowska D, Olszowy M (2012) On practical problems in estimation of antioxidant activity of compounds by DPPH method (Problems in estimation of antioxidant activity). Food Chem 131:1037–1043. doi:10.1016/j.foodchem.2011.09.067
Floegel A, Kim D-O, Chung S-J, Koo SI, Chun OK (2011) Comparison of ABTS/DPPH assays to measure antioxidant capacity in popular antioxidant-rich US foods. J Food Compos Anal 24:1043–1048. doi:10.1016/j.jfca.2011.01.008
Gayosso-García Sancho LE, Yahia EM, González-Aguilar GA (2013) Contribution of major hydrophilic and lipophilic antioxidants from papaya fruit to total antioxidant capacity. Food Nutr Sci 4:93–100. doi:10.4236/fns.2013.48A012
Lὀpez-Alarcὀn C, Denicola A (2013) Evaluating the antioxidant capacity of natural products: a review on chemical and cellular-based assays. Anal Chim Acta 763:1–10. doi:10.1016/j.aca.2012.11.051
Magalhăes LM, Segundo MA, Reis S, Lima JLFC (2008) Methodological aspects about in vitro evaluation of antioxidant properties. Anal Chim Acta 613:1–19. doi:10.1016/j.aca.2008.02.047
Mishra K, Ojha H, Chaudhury NK (2012) Estimation of antiradical properties of antioxidants using DPPH· assay: a critical review and results. Food Chem 130:1036–1043. doi:10.1016/j.foodchem.2011.07.127
Moon J-K, Shibamoto T (2009) Antioxidant assays for plant and food components. J Agric Food Chem 57:1655–1666. doi:10.1021/jf803537k
Nenandis N, Wang LF, Tsimidou M, Zhang HY (2004) Estimation of scavenging activity of phenolic compounds using the ABTS·+ assay. J Agric Food Chem 52:4669–4674. doi:10.1021/jf0400056
Paixão N, Perestrelo R, Marques JC, Câmara JS (2007) Relationship between antioxidant capacity and total phenolic content of red, rosé and white wines. Food Chem 105:204–214. doi:10.1016/j.foodchem.2007.04.017
Paśko P, Bartoń H, Zagrodzki P, Gorinstein S, Fołta M, Zachwieja Z (2009) Anthocyanins, total polyphenols and antioxidant activity in amaranth and quinoa seeds and sprouts during their growth. Food Chem 115:994–998. doi:10.1016/j.foodchem.2009.01.037
Pérez-Jiménez J, Arranz S, Tabernero M, Diaz-Rubio E, Serrano J, Goni I, Saura-Calixto F (2008) Updated methodology to determine antioxidant capacity in plant foods, oils and beverages: extraction, measurement and expression of results. Food Res Int 41:274–285. doi:10.1016/j.foodres.2007.12.004
Prior RL, Wu X, Schaich K (2005) Standardized methods for the determination of antioxidant capacity and phenolic in foods and dietary supplements. J Agric Food Chem 53:4290–4302. doi:10.1021/jf0502698
Ramos-Esudero F, Gonzáles-Miret M, Garcia-Asuero A (2012) Effect of various extraction systems on the antioxidant activity, kinetic and color of extracts from purple corn. Vitae (REVISTA DE LA FACULTAD DE QUÍMICA FARMACÉUTICA) 19:41–48
Roussis IG, Lambropoulos I, Tzimas P, Gkoulioti A, Marinos V, Tsoupeis D, Boutaris I (2008) Antioxidant activity of some Greek wines and wine phenolic extracts. J Food Compos Anal 21:614–621. doi:10.1016/j.jfca.2008.02.011
Sánchez –Moreno C, Larrauri JA, Saura-Calixto F (1999) Free radical scavenging capacity and inhibition of lipid oxidation of wines, grape juices and related polyphenolic constituents. Food Res Int 32:407–412. doi:10.1016/S0963-9969(99)00097-6
Shalaby EA, Shanab SMM (2013) Comparison of DPPH and ABTS assays for determining of antioxidant of water and methanol extracts of Spirulina platensis. Indian J Geo-Marine Sci 42:556–564
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Olszowy, M., Dawidowicz, A.L. Is it possible to use the DPPH and ABTS methods for reliable estimation of antioxidant power of colored compounds?. Chem. Pap. 72, 393–400 (2018). https://doi.org/10.1007/s11696-017-0288-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-017-0288-3