Abstract
Some patients experience weight regain (WR) or insufficient weight loss (IWL) after bariatric surgery (BS). We undertook a scoping review of WR and IWL after BS. We searched electronic databases for studies addressing the definitions, prevalence, mechanisms, clinical significance, preoperative predictors, and preventive and treatment approaches including behavioral, pharmacological, and surgical management strategies of WR and IWL. Many definitions exist for WR, less so for IWL, resulting in inconsistencies in the reported prevalence of these two conditions. Mechanisms and preoperative predictors contributing to WR are complex and multifactorial. A range of the current knowledge gaps are identified and questions that need to be addressed are outlined. Therefore, there is an urgent need to address these knowledge gaps for a better evidence base that would guide patient counseling, selection, and lead to improved outcomes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Bariatric surgery (BS) is currently the most efficacious and durable intervention for severe obesity. Unfortunately, some degree of weight regain (WR) is common after patients reach their nadir weight, where about 20–25% of patients struggle with considerable WR after BS [1,2,3,4,5]. Likewise, insufficient weight loss (IWL) (< 50% EWL) was the most common reason to qualify for revisional BS [6].
Some authors have proposed behavioral and biological mechanisms for WR [1], but the preoperative factors that predispose patients to significant WR remain unclear. Identifying these factors could improve the counseling of patients regarding the prevention of WR [2]. It is also important to mention that WR is associated with the deterioration of the quality of life and the reappearance or worsening of obesity-associated comorbidities, e.g., hypertension and type 2 diabetes (T2D) which necessitate close monitoring and appropriate management [7, 8]. Moreover, revisional BS to manage WR/IWL may have higher complication and mortality rates compared to primary BS [9,10,11,12].
The literature reveals gaps. Previous reviews focused mainly on the prevention strategies that improve weight loss [13, 14]. However, in terms of management approaches, one meta-analysis assessed surgical revision for the management of WR following BS [3]. Another scoping review explored various treatment options including behavioral, psychotherapeutic, and pharmacological interventions [15]. To the best of our knowledge, no previous study examined the broader landscape of WR and IWL following BS, to include the definitions, prevalence, causes, predictors, implications, prevention approaches, and management strategies.
Therefore, in order to address these knowledge gaps, the current scoping review explored the definitions, prevalence, causes, preoperative predictors, implications, prevention and management strategies of WR and IWL following BS. Given the obesity epidemic and frequency of BS as a treatment strategy, WR/IWL after BS requires a clear understanding in order to prevent patient dissatisfaction, lower quality of life, and relapse of comorbidities. These considerations inspired the current scoping review.
Methods
The goal of a scoping review is not to locate, recover, and include every single published report on the subject. Rather, the aim is deliberately broader in order to explore the literature, ascertain the essential characteristics of the subject, discover the likely gaps, and demonstrate critical examples. A scoping review can establish the types of prevailing evidence in a given field, the critical features related to a specified topic, or the underlying knowledge gaps. Scoping reviews are beneficial for broad questions, e.g., “What information exist on this topic?” and for assembling and evaluating information before commencing a systematic review. A scoping review is advantageous when the information on a topic has not been comprehensively reviewed or is complicated and diverse, as the broad extent of the information renders applying formal meta-analytic processes unachievable.
Thus, the scoping review was chosen to appraise WR after BS, a compound and diverse topic that demands extensive coverage. We utilized a six-step framework for scoping reviews (emphasized below) and applied its criteria that included identifying the research question/s; identifying relevant studies; study selection; charting the data (data extraction process); collating, summarizing, and reporting the results; and (optional step 6) a consultation exercise.
Research Questions
The present review “scoped” the literature in order to respond to six questions related to WR and IWL following BS: (1) What are the definition/s?; (2) What is the reported prevalence?; (3) What are the possible mechanisms and preoperative predictors?; (4) What are the clinical impacts and implications?; (5) What are the prevention and management strategies?; and, given the answers to the first 5 questions, (6) what are the knowledge gaps and possible ways forward?
Identifying Relevant Studies
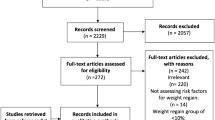
Information sources: We searched electronic databases including PubMed, MEDLINE, Embase, CINAHL, Web of Science, and Scopus, as well as Google scholar for published articles of all types of WR/IWL and their relationships with any type of BS. Keywords and search terms: keywords were “bariatric surgery” [in Title/Abstract]. The medical subject heading (MeSH) terms used were bariatric surgery [All Fields] AND (“weight regain” [MeSH Terms]; bariatric surgery [All Fields] AND (“weight gain” [MeSH Terms]; bariatric surgery [All Fields] AND (“insufficient weight loss” [MeSH Terms]; bariatric surgery [All Fields] AND (“postoperative AND insufficient weight loss”[MeSH Terms]; bariatric surgery [All Fields] AND (“postoperative AND weight regain” [MeSH Terms]; bariatric surgery [All Fields] AND (“postoperative AND weight gain” [MeSH Terms]. As the retrieved literature uncovered more features about post-BS WR/IWL, supplementary searches were formulated and conducted to obtain the literature related to the uncovered features.
Study Selection
Inclusion criteria included original studies; published in English; from 1 January 1994 through 30 September 2020; that assessed BS, WR/IWL; and enrolled patients of any age, gender, and ethnicity. Exclusion criteria comprised studies that did not include BS or WR/IWL.
Charting the Data
The data extracted consisted of items relevant to the five research questions being examined.
Collating, Summarizing, and Reporting the Results
The review team gathered, organized, and summarized the findings and report it below. Based on the emergent findings, we mapped the potential gaps that, if addressed, could present opportunities for advancing the field.
Consultation Exercise
Two experts (senior consultants, bariatric surgery and bariatric medicine) reviewed the findings to advise on and corroborate the findings of the current review.
Definition/s of WR and IWL after BS
In order to answer the first research question, there is a need to have a clear differentiation between two types of weight loss failure: IWL and WR. Insufficient weight loss is defined as excess weight loss percentage (EWL%) of < 50% 18 months post-BS [16]. WR is defined as progressive weight regain that occurs after achievement of an initial successful weight loss (defined as EWL>50%) [16]. The existing inconsistency, multiplicity, and lack of a standardized definition of WR (Table 1) lead to poor reporting and understanding of the clinical significance of WR [8, 16, 37, 38]. For example, the use of 5 continuous and 8 dichotomous measures among 1406 patients reported WR rates that ranged between 44 and 87% five years post Roux-en-Y gastric bypass (RYGB) [8]. Moreover, the percentage of maximum weight lost had the strongest association with the progression of T2D and hypertension, reduced quality of life, and reduced satisfaction with surgery [8]. Others observed a 16–37% WR five years after BS, where the three WR definitions examined were found to be associated with deterioration in quality of life; however, no associations were found with other comorbidities [23]. Therefore, more research is required to assess the extent of clinically significant WR that would indicate when an intervention is required and guide the type of intervention [16]. As for IWL, despite its distinct definition, it has been much less assessed, being only informally referred to in association with WR [16].
What Is the Reported Prevalence of WR and IWL After BS?
In terms of the second research question, WR following BS varies by the type of BS performed, whether restrictive and/or malabsorptive. Table 1 shows the variations in the reported prevalence of the two outcomes that the current review examined. A large prospective multicenter Swedish study found that 10 years after laparoscopic adjustable gastric banding (LAGB), patients regained 38% of the maximal weight they lost at 1 year [32], and WR after laparoscopic sleeve gastrectomy (LSG) was 27.8% (range 14–37%) at long-term follow-up (≥ 7 years) [33]. On the other hand, the Longitudinal Assessment of Bariatric Surgery (LABS) study reported a 3.9% WR 3–7 years after RYGB [34].
In comparison with WR, data on the prevalence of IWL is more limited [36], mostly assessed or stated as a “spin-off” when discussing the indication for revisional surgeries [17, 39]. For instance, among 17 patients who underwent revision after LSG, 40% were indicated for conversion to biliopancreatic diversion/duodenal switch (BPD/DS) and RYGB because of IWL [17], and others found that 32% of patients underwent revisional RYGB because of IWL [35].
Mechanisms of WR
As regards the third research question, several mechanisms contribute to WR following BS (Table 2). These include hormonal mechanisms, nutritional non-adherence, physical inactivity, mental health causes, and maladaptive eating [3, 37]. Surgical mechanisms are also implicated in WR, e.g., enlargement of gastric pouch, stoma dilatation, or gastrogastric fistula. Table 2 provides a summary of the mechanisms of WR and IWL.
Hormonal and Metabolic
Weight loss after BS is due to the anatomical exclusion of the foregut. This leads to a hormonal upregulation of pancreatic peptide YY, glucagon-like peptide-1 (GLP-1), and gastric inhibitory polypeptide hormones which promote satiety and minimize hunger, as well as downregulation of ghrelin with subsequent decrease in food intake [40, 41]. With time, alterations in the levels of ghrelin, leptin, and incretins diminish, resulting in WR [42,43,44].
For ghrelin, one study found that RYGB patients with WR had significantly higher ghrelin before and 2 years after surgery compared to those who maintained or lost weight (722 ± 29 vs 540 ± 156 pg/ml) [45]. Similar findings were observed among patients with WR 5 years after LSG [42]. Likewise, after RYGB, gastric inhibitory polypeptide and GLP-1 were lower among WR patients compared with patients who successfully maintained WL [43]. As for PYY, rodent studies showed that postsurgical WR was associated with failure to maintain elevated plasma PYY concentrations [46]. However, the role of leptin in WR is not completely understood, with suggestions that the weight plateau after BS could be due to a decline in leptin level, despite that the administration of leptin did not result in significant weight reduction among women with WR after RYGB [47, 48]. Nevertheless, leptin administration may diminish sweet cravings [48].
Post-bariatric hypoglycemia is also associated with WR. This is a reactive hypoglycemia that occurs after carbohydrate intake, caused by the change in intestinal anatomy that leads to an exaggerated insulin surge. Glucose fluctuation causes hunger a few hours after a meal leading to frequent snacking. After RYGB, 54% of patients with WR experienced blood glucose levels consistent with hypoglycemia [24]. After LSG and RYGB, 79.2% of the patients exhibited 10.8% WR at 40 months following surgery, and the odds of ≥ 10% WR was found to be significantly more among those with post-BS hypoglycemia (OR = 1.66) [49]. Collectively such findings highlight the need for more research to explore the long-term effects of BS on the metabolic and gut hormonal regulation of WR.
Dietary Non-adherence
BS reduces the caloric intake in the immediate postoperative period due to the reduced gastric capacity, decreased hunger, and increased satiety. Nevertheless, for some patients, the caloric intake gradually increases, hence contributing to WR. In a Swedish study, the mean daily caloric intakes increased from1500 kcal/day at 6 months to 2000 kcal/day at 4–10 years post-BS, contributing to long-term WR [32]. Among RYGB patients, 23% demonstrated dietary non-adherence and continuation of their pre-surgical eating patterns, leading to suboptimal weight loss, WR, or both [50]. After BS, food indiscretion such as intake of excessive calories, snacks, sweets, oils, and fatty foods was statistically higher among patients with WR [51]. As for IWL, after RYGB, more than 50% of patients had IWL due to excessive consumption of high-calorie liquids [52]. Moreover, the incidence of intake of high-calorie liquids was higher among LAGB patients than among RYGB/LSG patients [52], probably because the intake of such liquids is more tolerated post-LAGB than after RYGB/LSG due to the anatomical restriction and dumping syndrome associated with each of these two procedures [13].
Grazing is defined as repeated episodes of consumption of smaller quantities of food over a long period of time, accompanied with feelings of loss of control [53, 54]. Due to the restrictive effect of BS, grazing is physiologically more possible than large binges. Grazing has been shown to be associated with poor weight loss outcomes post-BS. A meta-analysis of 994 post-BS subjects, mostly women, found that the prevalence of grazing was 16.6–46.6%, while the prevalence of WR was 47% [55]. Likewise, the lack of post-BS nutritional follow-up was significantly associated with WR [51], where 60% of patients with WR never maintained nutritional follow-up [56]. This highlights the importance of appropriate nutritional counseling after BS in order to prevent WR, and to ensure a long-term weight maintenance.
Physical Inactivity
Physical activity (PA) is associated with weight loss after BS [57, 58]. Although many patients increase their PA, most remain insufficiently active. Studies have shown that only 10–24% of post-BS patients met the PA guidelines for health promotion (i.e., ≥ 150 min/week or moderate-to-vigorous PA in bouts of ≥ 10 min) [59]. Moreover, the method used to measure PA could lead to conflicting results. For instance, a prospective study found that although PA significantly increased post-surgery based on participants’ subjective self-reported exercise diary, objective measurement of the PA showed that 24–29% of participants were less active postoperatively than preoperatively [60]. Inadequate PA and sedentary lifestyle also contributed to WR post-RYGB, where the incidence of WR was higher among patients who remained relatively inactive compared with patients who performed PA [51]. Addressing the barriers to exercise, e.g., health concerns, pain, lack of proximity to a gym or park, and feeling self-conscious, might have a positive effect on post-BS PA [60, 61].
Mental Health Conditions
Psychological factors might undermine WL by impeding motivation or hindering the compliance with diet, PA, and other behaviors that are critical to maintain WL [62]. For example, a study found that one year after RYGB/LAGB, psychiatric disease was associated with IWL (47.5%) and WR (29.5%) [63]. Moreover, patients with ≥ 2 psychiatric conditions were ≈ 6 times more likely to lose no further weight or exhibit WR relative to those with ≤ 1 psychiatric diagnosis [63]. Likewise, depressive disorders are associated with poorer WL, but the directionality remains uncertain [64]. For instance, depression was identified in 45%, 12%, and 13% of patients prior to RYGB, and at 6 and 12 months following surgery, and while preoperative depression did not predict postoperative weight outcomes, depressive symptoms after surgery predicted poor weight loss outcomes [65]. Similarly, 50% of patients had depressive symptoms at 22–132 months post-BS [65]. These symptoms were significantly related to WR, loss of control over eating (LOC), and concerns with body image [66]. Equally, binge eating may persist or remerge after BS despite the physical limitations of BS on the capacity of the stomach [54, 67]. Researchers found that binge eaters had a 5.3 kg/m2 increase in BMI compared to a 2.4 kg/m2 among non-binge eaters 2–7 years after RYGB [68]. In addition, LOC predicted poorer WL, where, at 12 and 24 months after RYGB, patients with LOC lost significantly less weight compared to those with no LOC [65].
Anatomic Surgical Failure
The mechanism by which surgical failure leads to WR varies by the type of BS. After LAGB, distension of the pouch hinders WL [69]. After LSG, dilation of gastric pouch is correlated with postoperative BMI [70, 71], where dilatation leads to the loss of restriction resulting in reduced satiety, increased food intake, and subsequent WR. Research has shown that the mean gastric volume in patients with WR increased from 120 ml early post-surgery to 524 ml at 5 years [72]. After RYGB, the dilatation of the gastric pouch or gastrojejunostomy (GJ) stoma outlet was associated with increased food intake and WR [73, 74]. Among RYGB patients who underwent upper endoscopy as workup for WR, researchers identified the dilation of the GJ in 58.9%, enlarged gastric pouch in 28.8%, and both abnormalities in 12.3% of the patients [73]. Stoma diameter (> 2 cm) was also independently associated with WR [74]. Gastrogastric fistula may also diminish the restrictive and malabsorptive components of RYGB leading to WR [75].
Preoperative Predictors of WR and IWL Post-BS
In terms of the fourth research question, very few studies have addressed the preoperative predictors of IWL after BS. Table 2 provides a summary of the predictors of post-BS WR and IWL.
For instance, higher preoperative BMI was associated with WR and a worse weight trajectory, where patients with baseline BMI ≥ 50 kg/m2 were more likely to have significant WR, while those with BMI < 50 kg/m2 were more likely to continue losing weight at 12 months post-surgery [76]. Similarly, among LSG patients with BMI > 40 kg/m2 prior to surgery, 80–100% of them reported WR two years after surgery [77]. Greater preoperative BMI was also significantly associated with suboptimal WL [78, 79].
Age seems to be another preoperative predictor of WR and IWL, but findings are inconsistent. Some studies found that older age (> 60 years) was associated with WR [80, 81]. Conversely, other research observed younger age to be more likely associated with WR [82]. As for IWL, older age predicted IWL one year after BS [36]. Likewise, research suggested that male gender was associated with suboptimal or worse WL after RYGB [34, 78].
Mental health conditions represent another preoperative predictor of WR and IWL. The diagnosis of binge eating disorder prior to surgery predicted higher BMI at 5 years [83]. In agreement, the lack of control of food urges and low self-reported well-being scores independently predicted WR at 28 months post-RYGB [84]. Interestingly, preoperative psychiatric disorder was a weaker predictor of WR than the postoperative psychiatric disorder, likely due to the impact of the latter on eating behavior after BS [85]. The association of depression and anxiety with IWL has been less examined; however, recent research found that both conditions significantly predicted IWL 1 year after BS [36].
The presence of comorbidities predicts WR and IWL. For instance, T2D predicted WR and IWL [34, 79,80,81, 86]; while hypertension and low HDL cholesterol were both associated with poor weight trajectory [34]. Similarly, the number of comorbidities and previous history of hypertension predicted IWL [36]. Likewise, patients with obstructive sleep apnea achieved significantly lower EWL% at 1 year after surgery than patients with no sleep apnea [86].
Prevention and Management Strategies of WR and IWL After BS
Due to the complex etiology of WR and IWL, a multidisciplinary approach to treatment ensures effective long-term success. Table 2 provides a summary of the prevention and management strategies of WR and IWL.
The management starts with a comprehensive assessment that includes dietary patterns, PA level, psychological disorders, and motivation [3, 15]. Education sessions provide patients with practical knowledge, skills, and support in order to make the necessary changes required for weight optimization. Dietary and PA counseling by a team of health professionals ensures that patients receive specialist-tailored advice. Mental health issues such as mood, anxiety, addiction, and personality disorders should be addressed and managed effectively [3, 14]. Upper gastrointestinal contrast studies and esophagogastroduodenoscopy (EGD) should be undertaken, where appropriate, to evaluate the gastrointestinal tract and provide essential data about the gastric remnant, gastrojejunal anastomosis size, presence of gastrogastric fistula, as well as the location and integrity of the gastric band [101].
The prevention and management strategies are essentially similar for WR and IWL, with the exception that prevention does not include a surgical component. The management options of WR include behavior interventions, pharmacotherapy, endoscopic interventions, and surgical revision.
Behavioral Therapy
The aim of behavioral therapy is to assist patients in making long-term changes through monitoring and modification of their eating and PA behaviors. This can be achieved by controlling the environmental cues and stimuli that trigger eating or sedentary behaviors.
Management: A 10-week behavioral intervention emphasizing psychological skills to stop WR was feasible, acceptable, with high retention and satisfaction rates among completers [87]. More importantly, WR was stopped and even reversed with this intervention [87]. Similarly, among WR patients post-RYGB, cognitive and dialectical behavior therapies for 6 weeks resulted in a significant decrease in weight (mean 1.6 ± 2.38 kg), as well as improvements in the depressive symptoms, grazing patterns, and binge eating episodes [88]. Likewise, an online and phone behavioral intervention showed feasibility, acceptability, efficacy, and high satisfaction (70% retention) [89]. Moreover, WR was reversed, as participants achieved a significant 5.1% WL that was maintained at 3 months [89]. Given such promising results, more research is required to assess the effectiveness of behavioral therapy for IWL after BS as there are almost no studies on the subject.
Prevention: A systemic review of 15 behavioral management studies after RYGB/LAGB (8 studies provided cognitive behavioral therapy, 7 studies provided group support) found that patients achieved greater WL than controls across both interventions [90].
Dietary Therapy
Management: Very few studies have addressed the dietary management of IWL after BS. As for dietary management of WR, a randomized controlled trial (RCT) among 144 post-RYGB patients found that a nutritional intervention that comprised education sessions with a dietitian every other week for 6 weeks resulted in significantly greater EWL% (80% vs 64%) and BMI reduction (6.48 ± 4.37 vs 3.63 ± 3.41) at 12 months compared with usual care [91]. Likewise, another RCT one year post RYGB used a structured dietary intervention that incorporated portion-controlled foods compared to usual care with both groups receiving behavioral WL instructions [92]. This RCT found that the intervention group had significantly reduced calorie intake at 4 months (− 108 vs − 116) and increased WL at 4 and 6 months (− 4.56% vs − 0.13%, − 4.07% vs − 0.14%, respectively) compared with the usual care group [92]. Similarly, a 16-week RCT among women who regained ≥ 5% of their lowest post-RYGB weight found that whey protein supplementation promoted WL (− 1.86 kg) and fat mass loss, with preservation of the muscle mass, compared to controls who gained weight (0.42 kg) [93]. Protein intake after BS preserves lean muscle mass, increases satiety, and improves and promotes WL [93].
Prevention: A study randomized 84 patients to dietary counseling (15 min, every other week, in person dietary counseling by a dietitian for the first 4 postoperative months) versus standard care (no formal nutrition counseling sessions) [94]. The dietary counseling patients lost more total WL percentage (TWL%) than those who received the standard care (20.7 ± 1.1% vs 18.5 ± 1.1%) [94].
Pharmacotherapy
In terms of management, several anti-obesity medications have been utilized in conjunction with lifestyle modifications in order to decrease hunger, promote satiety, and halt the WR after BS (Table 2). Research found that, among 319 patients with WR or inadequate WL post-RYGB or LSG, 54%, 30.3%, and 15% of the sample lost ≥ 5%, ≥ 10%, and ≥ 15% of their total body weight (TBW) respectively using medications [95]. Furthermore, patients who received topiramate were 1.9 times more likely to lose ≥ 10% of their TBW% compared to those who received other medications [95]. In the same study, regardless of the postoperative BMI, RYGB patients were significantly more likely to lose ≥ 5% of their weight with medications [95].
Other research that examined the use of topiramate, phentermine, and/or metformin among young adults with WR found that 54.1%, 34.3%, and 22.9% of the sample lost ≥ 5%, ≥ 10%, and ≥ 15% of their postsurgical weight, respectively, and those who used metformin demonstrated the highest percent weight change compared to the other medications [96]. This study also observed that patients who underwent RYGB achieved significantly higher TWL% compared to those who underwent LSG (− 8.1% vs − 3.3%) [96].
Likewise, a study of phentermine vs phentermine–topiramate extended release among post-RYGB/LAGB patients with WR and weight plateau found that phentermine and phentermine–topiramate patients lost 6.35 kg (12.8% EWL%) and 3.81 kg (12.9% EWL%) respectively, at 90 days [97]. This study also observed that patients receiving phentermine weighed significantly less than those on phentermine–topiramate throughout the study, with no reported serious side effects for both medications [97].
An evaluation of liraglutide (a GLP-1 analog that promotes WL) among 117 patients who undertook RYGB, LAGB, or LSG observed that patients achieved statistically significant WL (− 6.3 ± 7.7 kg) 7 months after receiving the drug, regardless of the type of surgery [98]. Moreover, the decrease in weight remained significant after 1 year of liraglutide 3 mg, and nausea was the most prevalent side effect (29.1% of patients) [98].
Generally, research on the use of prescription weight loss medications to treat WR or IWL is scarce and is primarily retrospective, and no studies were powered to determine the best medication/s or timing of introduction of the medication, during either the weight loss plateau or initial WR [102].
Surgical Revision
Revision of BS is indicated to address surgical complications, WR or IWL [103]. For instance, IWL was the cause of 68.03% of the conversion to LSG or RYGB after LAGB [99]. Both conversions were associated with comparable WL at 6 and 12 months, although %EWL and BMI reduction after 24 months were greater among the patients who underwent RYGB [99]. Similarly, others found that IWL and WR were the indications for revision to BPD/DS or RYGB after LSG [17]. The EWL% was significantly greater at 34 months for BPD/DS compared to RYGB (59% vs 23%), however the short-term complications and vitamin deficiencies were higher among the patients who underwent revisional BPD/DS [17]. A systematic review (799 studies) assessed the revisions of RYGB for WR, including two conversions (to distal RYGB or BPD/DS) and three revisions (revision of gastric pouch and anastomosis, revision with gastric band or with endoluminal procedures) [100]. The review found that the mean percentage excess body mass index loss (%EBMIL) at 3 years for these five revisional procedures was 52.2%, 76%, 14%, 47.3%, and 32.1%, respectively [100].
Now What? Knowledge Gaps and Possible Way Forward
Table 3 depicts the current knowledge gaps. Although WR/IWL is encountered after BS, their definitions are far from being standardized. Equally, our current understanding of the associations between the different definitions of WR/IWL and their subsequent clinical impact is limited, and practically, no data exists on the clinical significance of IWL. In addition, the extent and significance of the variety of mechanisms implicated in WR/IWL still require more certainty, and within this domain, the association of such mechanisms with IWL seems completely deficient. Likewise, the prevailing knowledge about the range of preoperative predictors of WR could be strengthened. Moreover, RCTs are needed to assess the current prevention and management strategies of WR/IWL. Table 4 outlines a possible way forward in terms of a range of research questions that need to be urgently addressed in order to progress the evidence base.
Conclusion
BS remains effective and durable for treating obesity and ameliorating or resolving the obesity-related comorbidities. Understanding and tackling the WR/IWL after BS will likely require multi-pronged approaches. Standardized definitions could lead to more accurate and realistic estimates of the prevalence of these two conditions. The grouping together of WR and IWL as a common outcome or not depends on the context of the study; however, the current scoping review suggests that it would be better if WR and IWL were considered independently. Enhanced knowledge of the hormonal, psychological, behavioral, and surgical mechanisms contributing to WR/IWL could provide a more refined evidence base upon which appropriate prevention and management strategies could be premised. Better appreciation of the sequence and extents of the clinical impacts of WR/IWL will assist in determining the ideal timing and suitable intervention strategy/ies. Informed knowledge of the preoperative predictors of WR/IWL could aid to identify patients who are potentially at risk for both these conditions in order to offer them the necessary resources and counseling. Better understanding of the effectiveness and safety of the dietary, behavioral, pharmacological, and surgical prevention and management strategies will assist in the selection of intervention/s to mitigate WR/IWL. As the combination of such range of factors that contribute to WR/IWL are likely to be unique for each patient, applying such newly gained knowledge within a multidisciplinary team could be a step forward to individualized management plans tailored to patients in order to achieve resolution of WR/IWL.
References
Heinberg LJ, Bond DS, Carroll I, et al. Identifying mechanisms that predict weight trajectory after bariatric surgery: rationale and design of the biobehavioral trial. Surg Obes Relat Dis Off J Am Soc Bariatr Surg. 2020;16:1816–26.
Aliakbarian H, Bhutta HY, Heshmati K, et al. Pre-operative predictors of weight loss and weight regain following Roux-en-Y gastric bypass surgery: a prospective human study. Obes Surg. 2020;30:4852–9.
Karmali S, Brar B, Shi X, et al. Weight recidivism post-bariatric surgery: a systematic review. Obes Surg. 2013;23:1922–33.
Baig SJ, Priya P, Mahawar KK, et al. Weight regain after bariatric surgery-a multicentre study of 9617 patients from Indian bariatric surgery outcome reporting group. Obes Surg. 2019;29:1583–92.
Adams TD, Davidson LE, Litwin SE, et al. Weight and metabolic outcomes 12 years after gastric bypass. N Engl J Med. 2017;377:1143–55.
Andalib A, Alamri H, Almuhanna Y, et al. Short-term outcomes of revisional surgery after sleeve gastrectomy: a comparative analysis of re-sleeve, Roux en-Y gastric bypass, duodenal switch (Roux en-Y and single-anastomosis). Surg Endosc. 2020;
Debédat J, Sokolovska N, Coupaye M, et al. Long-term relapse of type 2 diabetes after Roux-en-Y gastric bypass: prediction and clinical relevance. Diabetes Care. 2018;41:2086–95.
King WC, Hinerman AS, Belle SH, et al. Comparison of the performance of common measures of weight regain after bariatric surgery for association with clinical outcomes. JAMA. 2018;320:1560–9.
Roth AE, Thornley CJ, Blackstone RP. Outcomes in bariatric and metabolic surgery: an updated 5-year review. Curr Obes Rep. 2020;9:380–9.
Yeung L, Durkan B, Barrett A, et al. Single-stage revision from gastric band to gastric bypass or sleeve gastrectomy: 6- and 12-month outcomes. Surg Endosc. 2016;30:2244–50.
Radtka JF, Puleo FJ, Wang L, et al. Revisional bariatric surgery: who, what, where, and when? Surg Obes Relat Dis Off J Am Soc Bariatr Surg. 2010;6:635–42.
Hamdi A, Julien C, Brown P, et al. Midterm outcomes of revisional surgery for gastric pouch and gastrojejunal anastomotic enlargement in patients with weight regain after gastric bypass for morbid obesity. Obes Surg. 2014;24:1386–90.
McGrice M, Don PK. Interventions to improve long-term weight loss in patients following bariatric surgery: challenges and solutions. Diabetes Metab Syndr Obes Targets Ther. 2015;8:263–74.
Kushner RF, Sorensen KW. Prevention of weight regain following bariatric surgery. Curr Obes Rep. 2015;4:198–206.
Shukla AP, He D, Saunders KH, et al. Current concepts in management of weight regain following bariatric surgery. Expert Rev Endocrinol Metab. 2018;13:67–76.
Nedelcu M, Khwaja HA, Rogula TG. Weight regain after bariatric surgery-how should it be defined? Surg Obes Relat Dis Off J Am Soc Bariatr Surg. 2016;12:1129–30.
Homan J, Betzel B, Aarts EO, et al. Secondary surgery after sleeve gastrectomy: Roux-en-Y gastric bypass or biliopancreatic diversion with duodenal switch. Surg Obes Relat Dis Off J Am Soc Bariatr Surg. 2015;11:771–7.
Liu SY-W, Wong SK-H, Lam CC-H, et al. Long-term results on weight loss and diabetes remission after laparoscopic sleeve gastrectomy for a morbidly obese Chinese population. Obes Surg. 2015;25:1901–8.
Colquitt JL, Pickett K, Loveman E, Frampton GK. Surgery for weight loss in adults. Cochrane Database Syst Rev. 2014;CD003641.
da Silva FBL, Gomes DL, de Carvalho KMB. Poor diet quality and postoperative time are independent risk factors for weight regain after Roux-en-Y gastric bypass. Nutr Burbank Los Angel Cty Calif. 2016;32:1250–3.
Amundsen T, Strømmen M, Martins C. Suboptimal weight loss and weight regain after gastric bypass surgery-postoperative status of energy intake, eating behavior, physical activity, and psychometrics. Obes Surg. 2017;27:1316–23.
Bastos ECL, Barbosa EMWG, Soriano GMS, et al. Determinants of weight regain after bariatric surgery. Arq Bras Cir Dig ABCD Braz Arch Dig Surg. 2013;26(Suppl 1):26–32.
Voorwinde V, Steenhuis IHM, Janssen IMC, et al. Definitions of long-term weight regain and their associations with clinical outcomes. Obes Surg. 2020;30:527–36.
Roslin M, Damani T, Oren J, et al. Abnormal glucose tolerance testing following gastric bypass demonstrates reactive hypoglycemia. Surg Endosc. 2011;25:1926–32.
Yanos BR, Saules KK, Schuh LM, et al. Predictors of lowest weight and long-term weight regain among Roux-en-Y gastric bypass patients. Obes Surg. 2015;25:1364–70.
Cooper TC, Simmons EB, Webb K, et al. Trends in weight regain following Roux-en-Y gastric bypass (RYGB) bariatric surgery. Obes Surg. 2015;25:1474–81.
Field AE, Wing RR, Manson JE, et al. Relationship of a large weight loss to long-term weight change among young and middle-aged US women. Int J Obes Relat Metab Disord J Int Assoc Study Obes. 2001;25:1113–21.
Angrisani L, Santonicola A, Iovino P, et al. Bariatric surgery worldwide 2013. Obes Surg. 2015;25:1822–32.
Jiménez A, Casamitjana R, Flores L, et al. Long-term effects of sleeve gastrectomy and Roux-en-Y gastric bypass surgery on type 2 diabetes mellitus in morbidly obese subjects. Ann Surg. 2012;256:1023–9.
Brethauer SA, Aminian A, Romero-Talamás H, et al. Can diabetes be surgically cured? Long-term metabolic effects of bariatric surgery in obese patients with type 2 diabetes mellitus. Ann Surg. 2013;258:628–36. discussion 636-637
Carmeli I, Golomb I, Sadot E, et al. Laparoscopic conversion of sleeve gastrectomy to a biliopancreatic diversion with duodenal switch or a Roux-en-Y gastric bypass due to weight loss failure: our algorithm. Surg Obes Relat Dis Off J Am Soc Bariatr Surg. 2015;11:79–85.
Sjöström L, Lindroos A-K, Peltonen M, et al. Lifestyle, diabetes, and cardiovascular risk factors 10 years after bariatric surgery. N Engl J Med. 2004;351:2683–93.
Clapp B, Wynn M, Martyn C, et al. Long term (7 or more years) outcomes of the sleeve gastrectomy: a meta-analysis. Surg Obes Relat Dis Off J Am Soc Bariatr Surg. 2018;14:741–7.
Courcoulas AP, King WC, Belle SH, et al. Seven-year weight trajectories and health outcomes in the longitudinal assessment of bariatric surgery (LABS) study. JAMA Surg. 2018;153:427–34.
Abdulrazzaq S, Elhag W, El Ansari W, et al. Is revisional gastric bypass as effective as primary gastric bypass for weight loss and improvement of comorbidities? Obes Surg. 2020;30:1219–29.
Cadena-Obando D, Ramírez-Rentería C, Ferreira-Hermosillo A, et al. Are there really any predictive factors for a successful weight loss after bariatric surgery? BMC Endocr Disord. 2020;20:20.
Lauti M, Kularatna M, Hill AG, et al. Weight regain following sleeve gastrectomy-a systematic review. Obes Surg. 2016;26:1326–34.
Lauti M, Lemanu D, Zeng ISL, et al. Definition determines weight regain outcomes after sleeve gastrectomy. Surg Obes Relat Dis Off J Am Soc Bariatr Surg. 2017;13:1123–9.
Amiki M, Seki Y, Kasama K, et al. Revisional bariatric surgery for insufficient weight loss and gastroesophageal reflux disease: our 12-year experience. Obes Surg. 2020;30:1671–8.
Tsouristakis AI, Febres G, McMahon DJ, et al. Long-term modulation of appetitive hormones and sweet cravings after adjustable gastric banding and Roux-en-Y gastric bypass. Obes Surg. 2019;29:3698–705.
Jirapinyo P, Jin DX, Qazi T, et al. A meta-analysis of GLP-1 after Roux-en-Y gastric bypass: impact of surgical technique and measurement strategy. Obes Surg. 2018;28:615–26.
Bohdjalian A, Langer FB, Shakeri-Leidenmühler S, et al. Sleeve gastrectomy as sole and definitive bariatric procedure: 5-year results for weight loss and ghrelin. Obes Surg. 2010;20:535–40.
Santo MA, Riccioppo D, Pajecki D, et al. Weight regain after gastric bypass: influence of gut hormones. Obes Surg. 2016;26:919–25.
Zalesin KC, Franklin BA, Miller WM, et al. Preventing weight regain after bariatric surgery: an overview of lifestyle and psychosocial modulators. Am J Lifestyle Med SAGE Publications. 2010;4:113–20.
Tamboli RA, Breitman I, Marks-Shulman PA, et al. Early weight regain after gastric bypass does not affect insulin sensitivity but is associated with higher ghrelin levels. Obes Silver Spring Md. 2014;22:1617–22.
Meguid MM, Glade MJ, Middleton FA. Weight regain after Roux-en-Y: a significant 20% complication related to PYY. Nutr Burbank Los Angel Cty Calif. 2008;24:832–42.
Korner J, Conroy R, Febres G, et al. Randomized double-blind placebo-controlled study of leptin administration after gastric bypass. Obes Silver Spring Md. 2013;21:951–6.
Conroy R, Febres G, McMahon DJ, et al. Recombinant human leptin does not alter gut hormone levels after gastric bypass but may attenuate sweet cravings. Int J Endocrinol. 2014;2014:120286.
Varma S, Clark JM, Schweitzer M, et al. Weight regain in patients with symptoms of post-bariatric surgery hypoglycemia. Surg Obes Relat Dis Off J Am Soc Bariatr Surg. 2017;13:1728–34.
Rusch MD, Andris D. Maladaptive eating patterns after weight-loss surgery. Nutr Clin Pract Off Publ Am Soc Parenter Enter Nutr. 2007;22:41–9.
Freire RH, Borges MC, Alvarez-Leite JI, et al. Food quality, physical activity, and nutritional follow-up as determinant of weight regain after Roux-en-Y gastric bypass. Nutr Burbank Los Angel Cty Calif. 2012;28:53–8.
Brolin RE, Robertson LB, Kenler HA, et al. Weight loss and dietary intake after vertical banded gastroplasty and Roux-en-Y gastric bypass. Ann Surg. 1994;220:782–90.
Saunders R. “Grazing”: a high-risk behavior. Obes Surg. 2004;14:98–102.
Colles SL, Dixon JB, O’Brien PE. Grazing and loss of control related to eating: two high-risk factors following bariatric surgery. Obes Silver Spring Md. 2008;16:615–22.
Pizato N, Botelho PB, Gonçalves VSS, et al. Effect of grazing behavior on weight regain post-bariatric surgery: a systematic review. Nutrients. 2017;9
Magro DO, Geloneze B, Delfini R, et al. Long-term weight regain after gastric bypass: a 5-year prospective study. Obes Surg. 2008;18:648–51.
Livhits M, Mercado C, Yermilov I, et al. Exercise following bariatric surgery: systematic review. Obes Surg. 2010;20:657–65.
Jacobi D, Ciangura C, Couet C, et al. Physical activity and weight loss following bariatric surgery. Obes Rev Off J Int Assoc Study Obes. 2011;12:366–77.
Rosenberger PH, Henderson KE, White MA, et al. Physical activity in gastric bypass patients: associations with weight loss and psychosocial functioning at 12-month follow-up. Obes Surg. 2011;21:1564–9.
King WC, Hsu JY, Belle SH, et al. Pre- to postoperative changes in physical activity: report from the longitudinal assessment of bariatric surgery-2 (LABS-2). Surg Obes Relat Dis Off J Am Soc Bariatr Surg. 2012;8:522–32.
Livhits M, Mercado C, Yermilov I, et al. Preoperative predictors of weight loss following bariatric surgery: systematic review. Obes Surg. 2012;22:70–89.
Sarwer DB, Allison KC, Wadden TA, et al. Psychopathology, disordered eating, and impulsivity as predictors of outcomes of bariatric surgery. Surg Obes Relat Dis Off J Am Soc Bariatr Surg. 2019;15:650–5.
Rutledge T, Groesz LM, Savu M. Psychiatric factors and weight loss patterns following gastric bypass surgery in a veteran population. Obes Surg. 2011;21:29–35.
Sheets CS, Peat CM, Berg KC, et al. Post-operative psychosocial predictors of outcome in bariatric surgery. Obes Surg. 2015;25:330–45.
White MA, Kalarchian MA, Masheb RM, et al. Loss of control over eating predicts outcomes in bariatric surgery patients: a prospective, 24-month follow-up study. J Clin Psychiatry. 2010;71:175–84.
Sousa P, Bastos AP, Venâncio C, et al. Understanding depressive symptoms after bariatric surgery: the role of weight, eating and body image. Acta Medica Port. 2014;27:450–7.
Niego SH, Kofman MD, Weiss JJ, et al. Binge eating in the bariatric surgery population: a review of the literature. Int J Eat Disord. 2007;40:349–59.
Kalarchian MA, Marcus MD, Wilson GT, et al. Binge eating among gastric bypass patients at long-term follow-up. Obes Surg. 2002;12:270–5.
Eid I, Birch DW, Sharma AM, et al. Complications associated with adjustable gastric banding for morbid obesity: a surgeon’s guides. Can J Surg J Can Chir. 2011;54:61–6.
Weiner RA, Weiner S, Pomhoff I, et al. Laparoscopic sleeve gastrectomy--influence of sleeve size and resected gastric volume. Obes Surg. 2007;17:1297–305.
Braghetto I, Cortes C, Herquiñigo D, et al. Evaluation of the radiological gastric capacity and evolution of the BMI 2-3 years after sleeve gastrectomy. Obes Surg. 2009;19:1262–9.
Braghetto I, Csendes A, Lanzarini E, et al. Is laparoscopic sleeve gastrectomy an acceptable primary bariatric procedure in obese patients? Early and 5-year postoperative results. Surg Laparosc Endosc Percutan Tech. 2012;22:479–86.
Yimcharoen P, Heneghan HM, Singh M, et al. Endoscopic findings and outcomes of revisional procedures for patients with weight recidivism after gastric bypass. Surg Endosc. 2011;25:3345–52.
Heneghan HM, Yimcharoen P, Brethauer SA, et al. Influence of pouch and stoma size on weight loss after gastric bypass. Surg Obes Relat Dis Off J Am Soc Bariatr Surg. 2012;8:408–15.
Filho AJB, Kondo W, Nassif LS, et al. Gastrogastric fistula: a possible complication of Roux-en-Y gastric bypass. JSLS. 2006;10:326–31.
Ochner CN, Jochner MCE, Caruso EA, et al. Effect of preoperative body mass index on weight loss after obesity surgery. Surg Obes Relat Dis Off J Am Soc Bariatr Surg. 2013;9:423–7.
Csendes A, Burgos AM, Martinez G, et al. Loss and regain of weight after laparoscopic sleeve gastrectomy according to preoperative BMI : late results of a prospective study (78-138 months) with 93% of follow-up. Obes Surg. 2018;28:3424–30.
Melton GB, Steele KE, Schweitzer MA, et al. Suboptimal weight loss after gastric bypass surgery: correlation of demographics, comorbidities, and insurance status with outcomes. J Gastrointest Surg Off J Soc Surg Aliment Tract. 2008;12:250–5.
Campos GM, Rabl C, Mulligan K, et al. Factors associated with weight loss after gastric bypass. Arch Surg Chic Ill 1960. 2008;143:877–84.
Al-Khyatt W, Ryall R, Leeder P, et al. Predictors of inadequate weight loss after laparoscopic gastric bypass for morbid obesity. Obes Surg. 2017;27:1446–52.
Paul L, van der Heiden C, Hoek HW. Cognitive behavioral therapy and predictors of weight loss in bariatric surgery patients. Curr Opin Psychiatry. 2017;30:474–9.
Shantavasinkul PC, Omotosho P, Corsino L, et al. Predictors of weight regain in patients who underwent Roux-en-Y gastric bypass surgery. Surg Obes Relat Dis Off J Am Soc Bariatr Surg. 2016;12:1640–5.
Marek RJ, Ben-Porath YS, van Dulmen MHM, et al. Using the presurgical psychological evaluation to predict 5-year weight loss outcomes in bariatric surgery patients. Surg Obes Relat Dis Off J Am Soc Bariatr Surg. 2017;13:514–21.
Odom J, Zalesin KC, Washington TL, et al. Behavioral predictors of weight regain after bariatric surgery. Obes Surg. 2010;20:349–56.
Wimmelmann CL, Dela F, Mortensen EL. Psychological predictors of weight loss after bariatric surgery: a review of the recent research. Obes Res Clin Pract. 2014;8:e299–313.
de Raaff CAL, Coblijn UK, de Vries N, et al. Predictive factors for insufficient weight loss after bariatric surgery: does obstructive sleep apnea influence weight loss? Obes Surg. 2016;26:1048–56.
Bradley LE, Forman EM, Kerrigan SG, et al. A pilot study of an acceptance-based behavioral intervention for weight regain after bariatric surgery. Obes Surg. 2016;26:2433–41.
Himes SM, Grothe KB, Clark MM, et al. Stop regain: a pilot psychological intervention for bariatric patients experiencing weight regain. Obes Surg. 2015;25:922–7.
Bradley LE, Forman EM, Kerrigan SG, et al. Project HELP: a remotely delivered behavioral intervention for weight regain after bariatric surgery. Obes Surg. 2017;27:586–98.
Rudolph A, Hilbert A. Post-operative behavioural management in bariatric surgery: a systematic review and meta-analysis of randomized controlled trials. Obes Rev Off J Int Assoc Study Obes. 2013;14:292–302.
Nijamkin MP, Campa A, Sosa J, et al. Comprehensive nutrition and lifestyle education improves weight loss and physical activity in Hispanic Americans following gastric bypass surgery: a randomized controlled trial. J Acad Nutr Diet. 2012;112:382–90.
Kalarchian MA, Marcus MD, Courcoulas AP, et al. Structured dietary intervention to facilitate weight loss after bariatric surgery: a randomized, controlled pilot study. Obes Silver Spring Md. 2016;24:1906–12.
Lopes Gomes D, Moehlecke M, Lopes da Silva FB, et al. Whey protein supplementation enhances body fat and weight loss in women long after bariatric surgery: a randomized controlled trial. Obes Surg. 2017;27:424–31.
Sarwer DB, Moore RH, Spitzer JC, et al. A pilot study investigating the efficacy of postoperative dietary counseling to improve outcomes after bariatric surgery. Surg Obes Relat Dis Off J Am Soc Bariatr Surg. 2012;8:561–8.
Stanford FC, Alfaris N, Gomez G, et al. The utility of weight loss medications after bariatric surgery for weight regain or inadequate weight loss: a multi-center study. Surg Obes Relat Dis Off J Am Soc Bariatr Surg. 2017;13:491–500.
Toth AT, Gomez G, Shukla AP, et al. Weight loss medications in young adults after bariatric surgery for weight regain or inadequate weight loss: a multi-center study. Child Basel Switz. 2018;5
Schwartz J, Chaudhry UI, Suzo A, et al. Pharmacotherapy in conjunction with a diet and exercise program for the treatment of weight recidivism or weight loss plateau post-bariatric surgery: a retrospective review. Obes Surg. 2016;26:452–8.
Wharton S, Kuk JL, Luszczynski M, et al. Liraglutide 3.0 mg for the management of insufficient weight loss or excessive weight regain post-bariatric surgery. Clin Obes. 2019;9:e12323.
Magouliotis DE, Tasiopoulou VS, Svokos AA, et al. One-anastomosis gastric bypass versus sleeve gastrectomy for morbid obesity: a systematic review and meta-analysis. Obes Surg. 2017;27:2479–87.
Tran DD, Nwokeabia ID, Purnell S, et al. Revision of Roux-en-Y gastric bypass for weight regain: a systematic review of techniques and outcomes. Obes Surg. 2016;26:1627–34.
Velapati SR, Shah M, Kuchkuntla AR, et al. Weight regain after bariatric surgery: prevalence, etiology, and treatment. Curr Nutr Rep. 2018;7:329–34.
Cohen RV, Cummings DE. Weight regain after bariatric/metabolic surgery: a wake-up call. Obesity. 2020;28:1004–4.
Patel S, Szomstein S, Rosenthal RJ. Reasons and outcomes of reoperative bariatric surgery for and complicated procedures (excluding adjustable gastric banding). Obes Surg. 2011;21:1209–19.
Funding
Open access funding provided by Qatar National Library.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflicts of interest.
Informed Consent
Informed consent does not apply.
Ethical Approval
This review analyzed data from existing published and unpublished studies. These studies are available in the public domain; ethics approval is not required.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
El Ansari, W., Elhag, W. Weight Regain and Insufficient Weight Loss After Bariatric Surgery: Definitions, Prevalence, Mechanisms, Predictors, Prevention and Management Strategies, and Knowledge Gaps—a Scoping Review. OBES SURG 31, 1755–1766 (2021). https://doi.org/10.1007/s11695-020-05160-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-020-05160-5