Abstract
Background
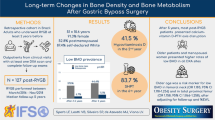
The aim of the present study was to study longitudinal changes in bone mineral density (BMD), vitamin D, and parathyroid hormone (PTH) levels in females over a 10-year period after laparoscopic Roux-en-Y gastric bypass (LRYGB).
Methods
Twenty-three women, mean age 43.4 ± 8.7 years and mean body mass index (BMI) 44.6 ± 5.17 kg/m2 at baseline, were included. BMD, BMI, S-calcium, S-25(OH)-vitamin D, and fP-PTH were measured preoperatively and 2, 5, and 10 years postoperatively.
Results
Ten years after surgery, BMD of the spine and femoral neck decreased by 20% and 25%, respectively. Changes in serum levels of vitamin D, PTH, and calcium over the same period were small.
Conclusion
After LRYGB with subsequent massive weight loss, a large decrease in BMD of the spine and femoral neck was seen over a 10-year postoperative period. The fall in BMD largely occurred over the first 5 years after surgery.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Obesity continues to increase in developed countries and even more so in developing countries. In Europe, the estimated prevalence of obesity among adults is 25%, and in Sweden the prevalence among women and men has reached 20% and 24% respectively [1]. Obesity is strongly associated with morbidity and mortality [2, 3]. Surgery is the most effective treatment for obesity, resulting in sustained weight loss [4, 5]. Gastric bypass is a commonly performed procedure worldwide, including Sweden [6, 7]. Though obesity constitutes a considerable health threat, these patients have increased bone mineral density compared with persons with normal weight status [8]. Bariatric procedures, in particular those based on malabsorption, lead to a fall in bone mineral density (BMD) [9,10,11,12], an increase in bone resorption and bone remodeling, and changes in bone histomorphometry parameters [10, 13, 14]. The risk for fracture increases at several skeletal sites after bariatric surgery [15,16,17]. These changes in bone metabolism seem not to be the result of weight loss alone, since BMD continues to decline even when the patient’s weight has stabilized [18]. Vitamin D deficiency [19,20,21,22,23] and secondary hyperparathyroidism [24, 25] could be important in this context; however, the long-term effects of gastric bypass surgery on the skeleton remain unclear.
The aim of this prospective study was to follow a group of female patients longitudinally over a 10-year period after laparoscopic Roux-en-Y gastric bypass (LRYGB) in order to detect changes in BMD and associated changes in serum levels of calcium, vitamin D, and PTH.
Methods
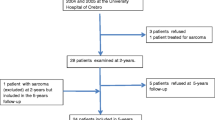
From January 2004 to December 2005, thirty-two consecutive female patients undergoing LRYGB at the Department of Surgery of the University Hospital of Örebro were recruited for this prospective longitudinal study. This cohort, as well as inclusion and exclusion criteria, has been described previously [18, 26]. Nine patients declined participation in the 10-year follow-up (details in the flow chart, Fig. 1) leaving the study group of 23 women.
Flow chart of the present and previous study [18]
BMD and total body fat (% of body weight) were measured using dual-energy X-ray absorptiometry (DXA) (DPX-L, Lunar Corp. Madison, WI, USA) preoperatively and at 2, 5, and 10 years postoperatively. Total body, lumbar spine, and femoral neck BMD were recorded. Osteopenia was defined as a BMD between 1.0 SD above and 2.5 SD below the young adult reference mean for women (t score), and osteoporosis as a BMD (t score) lower than − 2.5 SD.
S-calcium, S-albumin, S-creatinine, fP-alkaline phosphatase, fasting-P-PTH, and S-25-(OH)-vitamin D were measured at baseline, 5, and 10 years postoperatively. The blood samples were analyzed at the local laboratory in accordance with the stipulated routine for each method. Fasting-P-PTH was measured by immunochemistry. Initially, this was calibrated for the normal range of 10–73 ng/L, but during the study period units were changed from ng/L to pmol/L. To convert the results of fP-PTH in pmol/L to ng/L units, we used the equation (X pmol/L)/0.106 [27]. Vitamin D status was based on the level of S-25-OH-vitamin D using HPLC–APCI-MS. Vitamin D deficiency was defined as a S-25-OH-vitamin D < 50 nmol/L [28]. Lab tests and DXA measurements were not done on the same day and a few patients did not turn up the second day, as indicated in Table 3.
Statistical Analysis
Statistical analyses were performed with IBM SPSS Statistics 23 (IBM, Armonk, NY USA). Unless stated otherwise, continuous variables were presented as mean ± standard deviation. Normality of the continuous variables was evaluated using Shapiro–Wilk test. Standard mean difference and t test were used to compare independent groups. Differences of continuous variables between two dependent groups were determined using paired sample t test. All tests were two-sided and a p value less than 0.05 was considered statistically significant.
Results
Mean age at baseline was 43.4 ± 8.7 years. The mean preoperative weight was 122.8 ± 14.8 kg, and BMI 44.6 ± 5.17 kg/m2. Three patients were treated for diabetes mellitus and one patient had CPAP treatment for sleep apnea syndrome. At 10 years, four patients had treatment for hypertension but no patient was taking medication for diabetes mellitus and no patient required CPAP. The number of menopausal patients increased during the study (Table 1).
As expected, a significant decrease in BMI seen at 5 years (12.6 ± 6.14 BMI-units) and at 10 years (11.6 ± 5.75 BMI-units) were noted compared with baseline. The total body fat percentage decreased by 14% (Table 2).
Twenty-two of the 23 patients had initially normal BMD values in both spine and hip (exceeding 1 S. D above reference level in 18), but one woman had a spine DXA measurement showing osteopenia. All measurements showed a fall in bone mineral density over time with statistically significant differences at 5 and 10 years compared with baseline. The fall was greatest during the first 5 years with no significant difference in BMD between 5 and 10 years (Table 2 and Fig. 2). This corresponded to an overall decrease in spine BMD of 20% (0.27 g/cm2) and 25% in the femoral neck BMD (0.31 g/cm2). One patient had osteopenia prior to surgery and eight patients had developed osteopenia and one osteoporosis in the femur and/or spine at 5 years. DXA measurements at 10 years were basically the same apart from one additional patient who had developed osteopenia, giving nine patients with osteopenia and one with osteoporosis. Three patients with osteopenia and one with osteoporosis were affected in both the spine and the femoral neck.
The mean height of the patients decreased over the 10 years by 1.9 ± 1.05 cm (95% CI: 1.45–2.37, p = 0.000).
No patient was admitted or treated for a fracture during the first 5-year period. Between 5 and 10 years, three patients suffered a fracture of the wrist after minor trauma and one of the knee in a bicycle accident. Two of these had osteopenia.
Preoperative vitamin D levels were below 50 nmol/L (deficiency) in 50%, in 12 of 19 at 5 years and in 7 of 21 at 10 years. When patients without (n = 15) and those with (n = 8) vitamin D/calcium supplementation were compared, no statistically significant differences between preoperative and 10 year variables were observed (Table 3).
Patients not prescribed vitamin D/calcium had a mean vitamin D level that was higher at 10 years than preoperatively, but less pronounced than for the group with supplementation (Table 3).
Patients who declined participation in the 10-year follow-up did not differ significantly in age, comorbidity, or in preoperative measurements from those who did. For ethical reasons, we could not collect information from the medical charts of those patients, making further comparisons impossible.
Discussion
The main result of this study was the large decrease in bone mineral density seen over 10 years after gastric bypass. This has previously been reported in several studies with follow-up over a few years [29,30,31] and by us in a longitudinal study over 5 years [18]. We now report on a longitudinal follow-up of the same group of patients (less one) over 10 years. BMD fell throughout the study period albeit at a much slower rate over the last 5 years (no significant difference). Some of the BMD decrease, especially during the first few years during weight loss, could be a result from adaptation to the decreased stress of a heavy body. However, BMD continued to fall even after body weight nadir. Our results are in agreement with a recent cross-sectional study from Norway [32] of 124 patients 10 years after gastric bypass. There is also a small study from Denmark with longitudinal data over 7 years reporting similar results [33]. The fall in BMD in these studies was far more pronounced than natural loss of roughly 1% per year in the normal population [12, 34]. In the present study, BMD fell by 20% in the spine and 25% in the femoral neck over the 10-year follow-up. A similar disparity in fall in BMD between spine and femur/hip has been reported by others [35]. However, the fall in spine BMD could be an underestimation since a mean decrease in height of almost 2 cm was observed. This suggests a reduction in vertebral volume (i.e., height) due to compression secondary to a fall in BMD, but could also be caused by soft tissue (discs) reductions.
These findings indicate that following gastric bypass, BMD decreases from almost supernormal to levels approaching osteopenia and osteoporosis, with an increase in fracture risk compared with the general population [16], obese controls, and patients having a restrictive procedure. Two studies compared the gastric bypass procedure with a restrictive procedure such as gastric banding. In these, bypass seemed to carry a greater risk for osteoporotic fractures such as hip fractures, as well as wrist fractures [15, 17]. Reduction in bone mineral density does not fully explain the occurrence of wrist fractures; other risk factors could be involved including lower grip strength, increased walking speed, and increased risk for a fall outdoors [36,37,38]. Furthermore, decrease in weight is not only loss of fat mass but also lean mass and muscle mass [39]. However, our study was not sized to use fracture as the primary end-point.
At the time our patients were operated (2004–2005), vitamin D/calcium supplementation was not routinely prescribed postoperatively. European recommendations for such supplementation came several years later [40] and formal guidelines for the Nordic countries were published as late as 2017 [41, 42]. Awareness of these recommendations in Swedish primary healthcare has slowly grown over the last years and this is reflected in the number of patients prescribed vitamin D/calcium outside our study. This study was not designed to compare groups with or without vitamin D supplementation, such a study would need larger groups of patients, probably larger doses of supplementation [43], and well-controlled compliance.
Even though low vitamin D and high PTH levels have been reported after gastric bypass [23], it is also known that obesity itself is associated with increased PTH levels as well as low levels of vitamin D. In the present study, half of the patients had vitamin D deficiency preoperatively and a similar number was seen at 10 years in the non-supplemented group. Elevated PTH levels were seen in approximately 25% of patients preoperatively and this remained at 10 years. Considering the great SD for PTH values, we cannot draw certain conclusions from the differences in mean values at the different time points. The changes in PTH do not seem to explain the considerable changes in BMD. Present guidelines have focused on vitamin D/calcium supplementation, but there could well be physiological reasons for the fall in BMD other than lack of vitamin D and secondary hyperparathyroidism, such as changes in gastrointestinal peptides and hormone levels and uptake of protein and other nutrients [44]. In fact, current guidelines are not evidence-based but rather the opinion of experts. We could only find one randomized controlled study addressing this subject in which vitamin D, calcium, and protein supplementation together with physical exercise modified the fall in BMD but did not prevent it [45].
Estrogen deficiency is a well-known cause of low BMD. The mean age at the beginning of this study was 43 years and all but six patients were premenopausal. Obesity is commonly associated with menstrual irregularity leading to relative estrogen deficiency with androgen excess [46], and BMD is usually above normal preoperatively [47]. Bariatric surgery reverses the situation and can lead to resumption of a normal menstrual cycle [48]. It seems unlikely, therefore, that estrogen deficiency explains the decrease in BMD, but measured hormone levels during our study could have been of value.
IGF-1 is the main mediator of the anabolic effects of growth hormone, promoting cell proliferation and growth in several organs, including bone, which correlates with serum insulin levels. The synthesis of IGF-1 is dependent on adequate nutrition, but is also found to be inversely correlated to fat mass [49], more specifically to abdominal fat [50]. The GH/IGF-1-axis is more or less restored after gastric bypass surgery [50, 51] implying that weight loss potentially improves the impaired GH/IGF-1 axis seen in obesity. If, however, this is not accompanied by adequate nutrition, then the positive effect of improved IGF-1 levels may be hampered.
This longitudinal study following gastric bypass is the first to reporting on long-term changes in BMD over a 10-year period. The main limitations are the small cohort size, the spontaneous and uncontrolled consumption of vitamin D during follow-up and lack of an obese control group to compare with. DXA measurement of other parts of the skeleton, as well as more sophisticated laboratory tests measuring bone metabolism, could have provided a broader picture. The two sites we chose, however, are those recommended when diagnosing osteopenia and osteoporosis, and those closely associated with fracture risk. Considering the large changes in BMD observed, it seems unlikely that use of a more advanced DXA-technique measuring volume-BMD instead of area-BMD used here would have had an impact on the main result.
The marked and continued fall in BMD seen in this longitudinal study is probably the consequence an array of factors the nature of which remains unknown. Well-designed interventional studies are required if we are to develop strategies aimed at preventing a fall in BMD after gastric bypass.
References
Krzysztoszek J, Laudanska-Krzeminska I, Bronikowski M. Assessment of epidemiological obesity among adults in EU countries. Ann Agric Environ Med. 2019;26(2):341–9.
Guh DP, Zhang W, Bansback N, et al. The incidence of co-morbidities related to obesity and overweight: a systematic review and meta-analysis. BMC Public Health. 2009;9:88.
Ng M, Fleming T, Robinson M, et al. Global, regional, and national prevalence of overweight and obesity in children and adults during 1980-2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet. 2014;384(9945):766–81.
Buchwald H, Oien DM. Metabolic/bariatric surgery worldwide 2011. Obes Surg. 2013;23(4):427–36.
Sudlow A, le Roux CW, Pournaras DJ. The metabolic benefits of different bariatric operations: what procedure to choose? Endocrine connections. 2020;9(2):R28–r35.
(SOReg) SOSR. http://www.ucr.uu.se/soreg/. 2019 [Available from: http://www.ucr.uu.se/soreg/.
Angrisani L, Santonicola A, Iovino P, et al. Bariatric surgery and endoluminal procedures: IFSO Worldwide Survey 2014. Obes Surg. 2017;27(9):2279–89.
Qiao D, Li Y, Liu X, et al. Association of obesity with bone mineral density and osteoporosis in adults: a systematic review and meta-analysis. Public Health. 2019;180:22–8.
Shah M, Simha V, Garg A. Review: long-term impact of bariatric surgery on body weight, comorbidities, and nutritional status. J Clin Endocrinol Metab. 2006;91(11):4223–31.
Liu C, Wu D, Zhang JF, et al. Changes in bone metabolism in morbidly obese patients after bariatric surgery: a meta-analysis. Obes Surg. 2016;26(1):91–7.
Yu EW, Bouxsein ML, Putman MS, et al. Two-year changes in bone density after Roux-en-Y gastric bypass surgery. J Clin Endocrinol Metab. 2015;100(4):1452–9.
Hansen S, Shanbhogue V, Folkestad L, et al. Bone microarchitecture and estimated strength in 499 adult Danish women and men: a cross-sectional, population-based high-resolution peripheral quantitative computed tomographic study on peak bone structure. Calcif Tissue Int. 2014;94(3):269–81.
Parfitt AM, Podenphant J, Villanueva AR, et al. Metabolic bone disease with and without osteomalacia after intestinal bypass surgery: a bone histomorphometric study. Bone. 1985;6(4):211–20.
Cornejo-Pareja I, Clemente-Postigo M, Tinahones FJ. Metabolic and endocrine consequences of bariatric surgery. Front Endocrinol (Lausanne). 2019;10:626.
Ahlin S PM, Sjöholm K, et al. Fracture risk after three different bariatric surgery procedures in Swedish obese subjects: up to 26 years follow-up of a controlled intervention study. . J Int Med 2020; accepted.
Axelsson KF, Werling M, Eliasson B, et al. Fracture risk after gastric bypass surgery: a retrospective cohort study. J Bone Miner Res. 2018;33(12):2122–31.
Yu EW, Kim SC, Sturgeon DJ, Lindeman KG, Weissman JS. Fracture risk after Roux-en-Y gastric bypass vs adjustable gastric banding among Medicare beneficiaries. JAMA surgery. 2019.
Raoof M, Naslund I, Rask E, et al. Effect of gastric bypass on bone mineral density, parathyroid hormone and vitamin D: 5 years follow-up. Obes Surg. 2016;26(5):1141–5.
Compher CW, Badellino KO, Boullata JI. Vitamin D and the bariatric surgical patient: a review. Obes Surg. 2008;18(2):220–4.
Goldner WS, Stoner JA, Thompson J, et al. Prevalence of vitamin D insufficiency and deficiency in morbidly obese patients: a comparison with non-obese controls. Obes Surg. 2008;18(2):145–50.
Costa TM, Paganoto M, Radominski RB, et al. Impact of deficient nutrition in bone mass after bariatric surgery. Arq Bras Cir Dig. 2016;29(1):38–42.
Al-Shoha A, Qiu S, Palnitkar S, et al. Osteomalacia with bone marrow fibrosis due to severe vitamin D deficiency after a gastrointestinal bypass operation for severe obesity. Endocr Pract. 2009;15(6):528–33.
Karefylakis C, Naslund I, Edholm D, et al. Vitamin D status 10 years after primary gastric bypass: gravely high prevalence of hypovitaminosis D and raised PTH levels. Obes Surg. 2014;24(3):343–8.
Hultin H, Edfeldt K, Sundbom M, et al. Left-shifted relation between calcium and parathyroid hormone in obesity. J Clin Endocrinol Metab. 2010;95(8):3973–81.
Pugnale N, Giusti V, Suter M, et al. Bone metabolism and risk of secondary hyperparathyroidism 12 months after gastric banding in obese pre-menopausal women. Int J Obes Relat Metab Disord. 2003;27(1):110–6.
Simonyte K, Rask E, Naslund I, et al. Obesity is accompanied by disturbances in peripheral glucocorticoid metabolism and changes in FA recycling. Obesity (Silver Spring, Md). 2009;17(11):1982–7.
Institution K. PTH measurment: Karolinska Institution; 2013-01-23 [fPTH measurment]. Available from: http://karolinska.se/contentassets/28d05e254688414a9301c236c515d65c/2013-01-24_kundinfo-enhetsbyte-prolaktin-insulin-och-pth-130123.pdf+&cd=1&hl=sv&ct=clnk&gl=se.
Holick MF, Binkley NC, Bischoff-Ferrari HA, et al. Evaluation, treatment, and prevention of vitamin D deficiency: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(7):1911–30.
Casagrande DS, Repetto G, Mottin CC, et al. Changes in bone mineral density in women following 1-year gastric bypass surgery. Obes Surg. 2012;22(8):1287–92.
Vilarrasa N, San Jose P, Garcia I, et al. Evaluation of bone mineral density loss in morbidly obese women after gastric bypass: 3-year follow-up. Obes Surg. 2011;21(4):465–72.
Yu EW. Bone metabolism after bariatric surgery. J Bone Miner Res. 2014;29(7):1507–18.
Blom-Hogestol IK, Hewitt S, Chahal-Kummen M, et al. Bone metabolism, bone mineral density and low-energy fractures 10years after Roux-en-Y gastric bypass. Bone. 2019;127:436–45.
Hansen S, Jorgensen NR, Hermann AP, et al. Continuous decline in bone mineral density and deterioration of bone microarchitecture 7 years after Roux-en-Y gastric bypass surgery. Eur J Endocrinol. 2020;182(3):303–11.
Burt LA, Hanley DA, Boyd SK. Cross-sectional versus longitudinal change in a prospective HR-pQCT study. J Bone Miner Res. 2017;32(7):1505–13.
Gagnon C, Schafer AL. Bone health after bariatric surgery. JBMR plus. 2018;2(3):121–33.
Arnold CM, Dal Bello-Haas VP, Farthing JP, et al. Falls and wrist fracture: relationship to women’s functional status after age 50. Can J Aging. 2016;35(3):361–71.
Crandall CJ, Hovey KM, Andrews CA, et al. Bone mineral density as a predictor of subsequent wrist fractures: findings from the women’s health Initiative study. J Clin Endocrinol Metab. 2015;100(11):4315–24.
Crandall CJ, Hovey KM, Cauley JA, et al. Wrist fracture and risk of subsequent fracture: findings from the women’s health initiative study. J Bone Miner Res. 2015;30(11):2086–95.
Simonyte K, Olsson T, Naslund I, et al. Weight loss after gastric bypass surgery in women is followed by a metabolically favorable decrease in 11beta-hydroxysteroid dehydrogenase 1 expression in subcutaneous adipose tissue. J Clin Endocrinol Metab. 2010;95(7):3527–31.
Yumuk V, Tsigos C, Fried M, et al. European guidelines for obesity management in adults. Obesity facts. 2015;8(6):402–24.
Laurenius A, Naslund I, Sandvik J, et al. Nordiska riktlinjer för kosttillskott och uppföljning efter obesitaskirurgi. Lakartidningen. 2018;115
Sandvik J, Laurenius A, Naslund I, Videhult P, Wiren M, Aasheim ET. [Nordic guidelines for follow-up after obesity surgery]. Tidsskr Nor Laegeforen. 2018;138(4).
Lotito A, Teramoto M, Cheung M, Becker K, Sukumar D. Serum parathyroid hormone responses to vitamin D supplementation in overweight/obese adults: a systematic review and meta-analysis of randomized clinical trials. Nutrients. 2017;9(3).
Ben-Porat T, Elazary R, Sherf-Dagan S, et al. Bone health following bariatric surgery: implications for management strategies to attenuate bone loss. Adv Nutr. 2018;9(2):114–27.
Muschitz C, Kocijan R, Haschka J, et al. The impact of vitamin D, calcium, protein supplementation, and physical exercise on bone metabolism after bariatric surgery: the BABS study. J Bone Miner Res. 2016;31(3):672–82.
Butterworth J, Deguara J, Borg CM. Bariatric surgery, polycystic ovary syndrome, and infertility. J Obes. 2016;2016:1871594.
Jaruvongvanich V, Vantanasiri K, Upala S, et al. Changes in bone mineral density and bone metabolism after sleeve gastrectomy: a systematic review and meta-analysis. Surg Obes Relat Dis. 2019;15(8):1252–60.
Kjaer MM, Madsbad S, Hougaard DM, et al. The impact of gastric bypass surgery on sex hormones and menstrual cycles in premenopausal women. Gynecol Endocrinol. 2017;33(2):160–3.
Fornari R, Marocco C, Francomano D, et al. Insulin growth factor-1 correlates with higher bone mineral density and lower inflammation status in obese adult subjects. Eating and weight disorders : EWD. 2018;23(3):375–81.
Mittempergher F, Pata G, Crea N, et al. Preoperative prediction of growth hormone (GH)/insulin-like growth factor-1 (IGF-1) axis modification and postoperative changes in candidates for bariatric surgery. Obes Surg. 2013;23(5):594–601.
Eden Engstrom B, Burman P, Holdstock C, et al. Effects of gastric bypass on the GH/IGF-I axis in severe obesity--and a comparison with GH deficiency. Eur J Endocrinol. 2006;154(1):53–9.
Funding
Open access funding provided by Örebro University. DXA measurements were funded by Lisa & Johan Grönbergs stiftelse, Stockholm. The funders had no influence on any part of the study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Dr. Näslund has received private fees for consulting and lectures from Baricol Bariatrics AB Sweden, AstraZeneca A/S Denmark and Ethicon, and Johnson & Johnson. These payments were not connected with this study. The remaining authors have nothing to declare.
Ethical Approval Statement
The study was approved by the Ethics Committee of the Uppsala-Örebro Region in Sweden.
Informed Consent Statement
All patients completed a health declaration form including medication, possible menopause and previous history of fracture. All study participants signed a written informed consent form.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Raoof, M., Näslund, I., Rask, E. et al. Bone Mineral Density, Parathyroid Hormone, and Vitamin D After Gastric Bypass Surgery: a 10-Year Longitudinal Follow-Up. OBES SURG 30, 4995–5000 (2020). https://doi.org/10.1007/s11695-020-04912-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-020-04912-7