Abstract
Background
The benefit of beta-blockade on postoperative outcome remains controversial, though recent studies have suggested a role during major non-cardiac surgery. The benefit of beta-blockade during minimally invasive gastric bypass surgery remains unclear. The aim of the present study was to evaluate the possible association between preoperative beta-blocker therapy and postoperative outcome after laparoscopic gastric bypass surgery.
Methods
Patients operated with primary laparoscopic gastric bypass surgery in Sweden between 2007 and 2017 were identified through the Scandinavian Obesity Surgery Registry. The dataset was linked to the Swedish National Patient Registry, the Swedish Prescribed Drug Registry, and Statistics Sweden. The main outcome was serious postoperative complication within 30 days of surgery; with postoperative complication, 90-day and 1-year mortality, and weight loss at 2 years after surgery as secondary endpoints. The Poisson regression model was used to evaluate primary and secondary categorical outcomes. A general mixed model was performed to evaluate 2-year weight loss.
Results
In all, 50281 patients were included in the study. No difference was seen between patients on beta-blockade and the control group regarding postoperative complications (adjusted incidence rate ratio 1.04 (95%CI 0.93–1.15), p = 0.506), serious postoperative complication (adjusted IRR 1.06 95%CI 0.89–1.27), p = 0.515), 90-day mortality (adjusted IRR 0.71 (95%CI 0.24–2.10), p = 0.537), and 1-year mortality (adjusted IRR 1.26 (95%CI 0.67–2.36), p = 0.467). Weight loss 2 years after surgery was slightly greater in patients on beta-blockade (adjusted coefficient 0.53 (95%CI 0.19–0.87), p = 0.002).
Conclusions
Beta-blockade has limited impact on postoperative outcome after laparoscopic gastric bypass surgery.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Only two decades ago, bariatric surgery was considered a high-risk surgical procedure with mortality rates reaching 1% or higher [1]. The introduction and refinement of laparoscopic bariatric surgery in conjunction with better pre-, peri-, and postoperative management offered by programs such as enhanced recovery after surgery (ERAS) has led to low complication and mortality rates in these cases [2, 3]. Despite these advances, the rate of adverse outcomes remains high due to the high volume of procedures performed annually worldwide [4].
Surgical and anesthesiological trauma during bariatric surgery causes a physiological stress response resulting in adrenergic hyperactivity, inflammation activity, and hypercoagulability, ultimately increasing the risk for perioperative complications [5]. While a reduction in stress calls for a multimodal approach [6], several details in perioperative care play an important role in risk reduction. Previous studies have suggested an association between beta-blocker (beta-adrenergic blocking agents) exposure and lower adverse outcome rates in patients subjected to elective and emergency major abdominal surgery [7,8,9,10]. Whether the same benefits are to be gained in elective, minimally invasive bariatric surgery remains unclear.
The aim of the present study was to evaluate the effect of beta-blocker therapy on postoperative outcome after laparoscopic gastric bypass surgery.
Methods
Study Design
In order to retain a homogenous study population, all adult patients (≥ 18 years) subjected to a primary laparoscopic gastric bypass procedure in Sweden between Jan 1, 2007, and Sept 21, 2017, were recruited from the Scandinavian Obesity Surgery Registry (SOReg) and included in the present study. SOReg is the Swedish national quality and research register for all bariatric surgical procedures with an overall coverage approaching 100% [11]. Using the Swedish personal identity number, unique to each citizen, the SOReg file was matched with individual data from the Swedish population registry, the Swedish National Patient Registry (for hospital admission data and outpatient data), the Swedish prescribed drug registry, and Statistics Sweden.
Baseline data, perioperative data and follow-up data on short-term complications and weight loss were extracted from the SOReg database. Mortality data were based on information from the Swedish population registry. Data on income were extracted from Statistics Sweden and sub-classified into percentiles based on the disposable income (total taxable income minus taxes and other negative transfers) of all adults in Sweden the year of surgery. Comorbidity was based on data from the SOReg and defined as an obesity-related comorbidity requiring ongoing pharmacologic treatment or continuous positive airway pressure treatment. Cardiovascular comorbidity was based on data from the Swedish National Patient Registry, and defined as arrhythmia, ischemic heart disease, or heart failure. Preoperative weight reduction was defined as a weight reduction > 5% of the total weight on a very low calorie diet during the weeks prior to surgery [12].
Use of beta-blocker medication was based on information from the Swedish Prescribed Drug Registry including all physician-issued prescriptions in Sweden. Patients were divided into a beta-blocker-negative (control group) and a beta-blocker-positive group. Beta-blocker-positive patients were defined as those with a prescription issued (ATC-codes C07AA, C07AB, C07AG, C07FB) within 12-months prior to their surgery.
Postoperative complications were classified according to the Clavien-Dindo classification of postoperative complications [13]. A complication graded as ≥ 3b (a complication requiring intervention under general anesthesia, or resulting in organ failure or death) was considered a serious postoperative complication. The main outcomes of interest were serious postoperative complication, specific postoperative complication, and 90-day and 1-year mortality rates. A secondary outcome of interest was weight loss 2 years after surgery.
Procedure
The surgical procedure for laparoscopic gastric bypass is highly standardized in Sweden, with 99% using the antecolic, antegastric technique where a small gastric pouch (approximately 25 mL) is constructed by linear stapling of the gastroenterostomy combined with hand-sewn closure of the remaining defect [14].
Statistical Analyses
Baseline sociodemographic and clinical characteristics of the patients are presented using mean ± standard deviation (SD) for continuous variables and percentage for categorical variables. Logistic regression was used to compare categorical baseline characteristics, and Student’s t test for continuous variables.
Because of the time-to-event feature of the data, risk for early postoperative complication, serious postoperative complication, and mortality rates were evaluated using the Poisson regression model. Risks for 90-day and 1-year mortality were also examined using the proportional hazards Cox-regression model in a sensitivity analysis. The generalized linear mixed model was used for evaluating associated factors for weight loss 2 years after surgery. Year of surgery, sex, age, body mass index (BMI), disposable income, obesity-related comorbidity, previous deep venous thrombosis/pulmonary embolism (DVT/PE), and preoperative weight reduction were included as covariates in the multivariable regression models for postoperative complication, 1-year mortality, and total weight loss 2 years after surgery. Due to low numbers of cases, preoperative weight reduction was excluded from the 90-day mortality analysis.
In the multivariable models, multiple imputations were used to handle missing values of disposable income and preoperative weight reduction by applying the iterative Markov chain Monte Carlo method [15]. The estimates of 5 imputed data sets were combined according to Rubin’s rules [16]. Risks for complication and mortality were reported as incidence rate ratios (IRR) with corresponding 95% confidence intervals (CI). The effect of a factor on weight loss was reported as a coefficient with 95%CI for the percentage of total weight loss. A two-sided p value < 0.05 was considered statistically significant. The SPSS Statistics version 25 (IBM, Armonk, NY, USA) and Stata 15.1 (Stata Corp, College Station, TX, USA) were used for the statistical analyses.
Ethics
The study was approved by the regional ethics committee in Stockholm and followed the ethical standards of the 1964 Helsinki Declaration and its later amendments.
Results
In all, 50281 patients were included in the study. Follow-up rate at the day 30 clinic visit was 95.8% (n = 48168), and since registry classification of postoperative complications was introduced in 2010, detailed information was available for 41726 patients. Mortality registered in the Swedish population registry is 100% complete, so that all patients in the study population were included in the 90-day mortality analysis. Only patients with a postoperative follow-up longer than 1 year were included in the analysis of 1-year mortality (n = 48450). Registrations of weight at both baseline and 2 years after surgery were available for 27650 patients.
Baseline characteristics for patients on beta-blocker therapy and the control group are presented in Table 1. A preoperative weight reduction of > 5% TWL was achieved in 3721 patients (61.0%) in the beta-blocker-positive group and 24562 patients (62.3%) in the control group (p = 0.055).
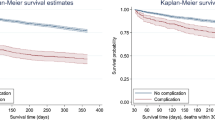
A postoperative complication occurred in 4010 patients (8.3%) with no difference between beta-blocker-positive (9.9%) and beta-blocker-negative (8.1%) patients (Table 2). A serious postoperative complication occurred in 1356 cases (3.2%). In the Poisson regression model, statistically significant risk for the development of a serious postoperative complication was associated with increased age, while higher BMI and higher disposable income (> 50th percentile) were associated with lower risk (Table 3). There was no statistically significant difference in the risk for a postoperative complication associated with beta-blocker use.
The 90-day mortality rate was 0.06% (n = 29). After adjustment for potential confounders, no statistically significant association with beta-blocker use could be detected (adjusted IRR 0.71 (CI 0.24–2.10), p = 0.537). The 1-year mortality rate was 0.2% (n = 77), and no statistically significant association with beta-blocker therapy was found after adjustment for potential confounders (adjusted IRR 1.26 (CI 0.67–2.36), p = 0.467). The same results were obtained in the sensitivity analysis using proportional hazard Cox-regression model.
Two years after surgery, the mean fall in BMI was 13.7 ± 4.45 kg/m2 or 32.3 ± 8.75 %TWL. After adjustment for potential confounders, patients on beta-blockade medication experienced a slightly better weight reduction (Table 4).
Discussion
In contrast to the positive effects of beta-blockers in patients subjected to a major complex abdominal surgical procedure, the same convincing benefits are not seen in morbidly obese patients undergoing laparoscopic gastric bypass who are on beta-blocker therapy.
Surgery and general anesthesia are associated with physiological stress causing inflammatory responses, hypercoagulability, and hypoxic states, with increased risk for perioperative complications [5]. A central part of stress activation appears to be caused by adrenergic hyperactivity leading to an increase in blood pressure, heart rate, coronary artery sheer stress, relative insulin deficiency, and free fatty acid levels [5, 17]. It is well established that reducing the physiological stress caused by surgical trauma improves postoperative outcome [6]. It has been postulated that the lower mortality seen in trauma patients on a beta-blocker is due to downregulation of the toxic effects of the trauma-induced hyperadrenergic state [18]. This association is even more pronounced in patients suffering a severe isolated traumatic brain injury who are on beta-blocker therapy prior to their injury [19]. A similar finding has been observed in patients undergoing major non-cardiac surgery [7,8,9,10]. Lindenauer and colleagues demonstrated a significant reduction in hospital deaths in a cohort of more than 780,000 patients with preoperative beta-blocker exposure [9]. Similar findings were reproduced by Kwon et al [10]. Furthermore, a strong association with adverse events up to 90 days following non-cardiac surgery has been demonstrated when regular beta-blocker therapy is discontinued preoperatively [10]. However, it is far from established that beta-blockade offers a general risk reduction in all non-cardiac surgical procedures. In the POISE trial, patients undergoing a wide range of surgical procedures did not show any benefit from preoperative beta-blockade on the postoperative outcomes measured [20]. One explanation for these contradicting results could be different patient populations and surgical procedures. Furthermore, in the POISE-trial, a very high dose of beta-blocker (not commonly used in clinical practice) was used and some patients did not discontinue the use of beta-blockers in spite of trial recommendations, which could explain the marginally increased risk for stroke in their study population [21]. Beta-blockade may offer most benefit in major abdominal surgical procedures, with pronounced surgical trauma, on older, frailer patients with higher incidence of comorbidities [7,8,9,10]. This is not the case, however, with relatively younger patients subjected to a minimally invasive surgical procedure such as laparoscopic gastric bypass under elective conditions and multimodal optimization. The present study supports the continuation of beta-blockade therapy during the perioperative phase, as previously described in a combined colorectal and bariatric surgical group [10].
Although no association between beta-blockade and serious postoperative complications was detected in the present study, increasing age, lower BMI, and lower disposable income were all associated with increased risk for such adverse events. While age is a well-known risk factor for postoperative complications after bariatric surgery [3, 22,23,24], BMI has been reported to be a poor predictor [3, 22]. However, previous studies have reported a tendency towards higher risk for patients with extreme BMI values, both low and high [3, 25, 26]. Since there are very few patients with an extremely high BMI in Sweden [3], this may well be the explanation for the lack of association between high BMI and complications in the present study. An association between lower socioeconomic status and postoperative adverse outcome has recently been reported [27], and this is supported by the present study. However, the association is complex and multifactorial, and related to a multitude of factors from health-promoting activities and behavior and good nutritional status, to the ability to understand and utilize healthcare information more often seen in patients with higher socioeconomic status [28,29,30].
Other than the small influence of beta-blockade on postoperative weight loss, higher disposable income and cardiovascular disease both had a small positive effect on postoperative weight loss. Sleep apnea, hypertension, and dyslipidemia, however, were associated with a small negative effect. Higher age, male gender, depression, and diabetes were all associated with a somewhat greater negative influence on postoperative weight loss, while year of surgery, preoperative BMI, and preoperative weight reduction had a greater positive influence. Although the mechanism of action to these findings is still to be fully elucidated, the downregulation of the stress response which could affect the metabolic system is a theory that needs further investigation. Also, patients on beta-blocker therapy might have higher compliance to recommended diet prescription and more regularly attend follow-up visits due to other comorbidities. Patients with a high BMI have previously been reported to lose more weight after bariatric surgery compared with patients with a low BMI [31], although these patients have difficulties in reaching higher success rates when defined as excess weight loss [32]. Our results also support previous reports of lower weight loss among patients with type 2 diabetes [32, 33], males, and older patients [34, 35]. While the lower weight loss among patients with diabetes and higher age may be explained by increased insulin resistance, mobility limitations, and associated comorbidity, the gender difference is more difficult to explain. Furthermore, the negative influence of socioeconomic factors and depression are somewhat controversial and also warrant future studies designed to look at these specific variables. Finally, time and experience generally leads to improved outcome after bariatric surgery [26]. As evident from the results of the present study, this improvement appears to apply to postoperative weight loss as well.
Another potential risk of beta-blocker therapy is the interference with early clinical signs of stress due to complications, i.e., tachycardia, that may lead to late diagnosis and treatment. In the present study, no distinction between complications, signs of stress, and time-line of recognition of such events and treatment could be made.
Despite the obvious strength of the large material based on data of very high quality in this study, there are limitations that must be acknowledged. The main limitation lies in the study design. An observational study can never fully compensate for all potential factors that may influence postoperative outcome. Despite combining high-quality sources of information, the variables used are limited to those available in the registries used. There may thus be factors of importance that we have not evaluated or adjusted for in the present study. Furthermore, due to the retrospective nature of the study, we had to assume that patients taking a beta-blocker were continued on such treatment throughout the perioperative period. Current Swedish guidelines recommend that beta-blocker therapy should always be continued prior to and after surgery as long as there is no absolute contraindication. Finally, follow-up of weight loss after surgery was limited to 2 years, a time when most patients have maximum weight loss after surgery [36]. Longer follow-up would capture weight regain, an important issue in bariatric surgery. However, a large observational study beyond this time framework is difficult due to high loss to follow-up [37].
In this large nationwide observational study, beta-blockade had limited influence on postoperative outcomes after laparoscopic gastric bypass surgery. The safety benefits of beta-blockade hitherto described may be limited to abdominal surgery with greater surgical trauma and the hyperadrenergic state thus induced.
References
Fernandez Jr AZ, Demaria EJ, Tichansky DS, et al. Multivariate analysis of risk factors for death following gastric bypass for treatment of morbid obesity. Ann Surg. 2004;239(5):698–702.
Thorell A, MacCormick AD, Awad S, et al. Guidelines for perioperative care in bariatric surgery: enhanced recovery after surgery (ERAS) society recommendations. World J Surg. 2016;40(9):2065–83.
Stenberg E, Szabo E, Agren G, et al. Early complications after laparoscopic gastric bypass surgery: results from the Scandinavian obesity surgery registry. Ann Surg. 2014;260(6):1040–7.
Angrisani L, Santonicola A, Iovino P, et al. IFSO worldwide survey 2016: primary, endoluminal, and revisional procedures. Obes Surg. 2018;28(12):3783–94.
Devereaux PJ, Goldman L, Cook DJ, et al. Perioperative cardiac events in patients undergoing noncardiac surgery: a review of the magnitude of the problem, the pathophysiology of the events and methods to estimate and communicate risk. CMAJ. 2005;173(6):627–34.
Kehlet H, Wilmore DW. Evidence-based surgical care and the evolution of fast-track surgery. Ann Surg. 2008;248(2):189–98.
Ahl R, Matthiessen P, Fang X, et al. Effect of beta-blocker therapy on early mortality after emergency colonic cancer surgery. Br J Surg. 2019;106(4):477–83.
Ahl R, Matthiessen P, Fang X, et al. beta-Blockade in rectal cancer surgery: a simple measure of improving outcomes. Ann Surg. 2018. E-pub Jul 24
Lindenauer PK, Pekow P, Wang K, et al. Perioperative beta-blocker therapy and mortality after major noncardiac surgery. N Engl J Med. 2005;353(4):349–61.
Kwon S, Thompson R, Florence M, et al. beta-blocker continuation after noncardiac surgery: a report from the surgical care and outcomes assessment program. Arch Surg. 2012;147(5):467–73.
Hedenbro JL, Naslund E, Boman L, et al. Formation of the Scandinavian obesity surgery registry, SOReg. Obes Surg. 2015;25(10):1893–900.
Anderin C, Gustafsson UO, Heijbel N, et al. Weight loss before bariatric surgery and postoperative complications: data from the Scandinavian obesity registry (SOReg). Ann Surg. 2015;261(5):909–13.
Clavien PA, Barkun J, de Oliveira ML, et al. The Clavien-Dindo classification of surgical complications: five-year experience. Ann Surg. 2009;250(2):187–96.
Olbers T, Lonroth H, Fagevik-Olsen M, et al. Laparoscopic gastric bypass: development of technique, respiratory function, and long-term outcome. Obes Surg. 2003;13(3):364–70.
White IR, Royston P, Wood AM. Multiple imputation using chained equations: issues and guidance for practice. Stat Med. 2011;30(4):377–99.
Rubin D. Multiple imputation for nonresponse in surveys, vol. 81. Hoboken: Wiley; 2004.
Desborough JP. The stress response to trauma and surgery. Br J Anaesth. 2000;85(1):109–17.
Alali AS, Mukherjee K, McCredie VA, et al. Beta-blockers and traumatic brain injury: a systematic review, meta-analysis, and eastern association for the surgery of trauma guideline. Ann Surg. 2017;266(6):952–61.
Mohseni S, Talving P, Wallin G, et al. Preinjury beta-blockade is protective in isolated severe traumatic brain injury. J Trauma Acute Care Surg. 2014;76(3):804–8.
POISE study group, Devereaux PJ, Yang H, et al. Effects of extended-release metoprolol succinate in patients undergoing non-cardiac surgery (POISE trial): a randomised controlled trial. Lancet. 2008;371(9627):1839–47.
Caramelli B, Gualandro DM, Freitas S, et al. Beta-blocker therapy in non-cardiac surgery. Lancet. 2008;372(9644):1145.
Geubbels N, de Brauw LM, Acherman YI, et al. Risk stratification models: how well do they predict adverse outcomes in a large Dutch bariatric cohort? Obes Surg. 2015;25(12):2290–301.
Gerber P, Anderin C, Szabo E, et al. Impact of age on risk of complications after gastric bypass: a cohort study from the Scandinavian obesity surgery registry (SOReg). Surg Obes Relat Dis. 2018;14(4):437–42.
Finks JF, Kole KL, Yenumula PR, et al. Predicting risk for serious complications with bariatric surgery: results from the Michigan Bariatric Surgery Collaborative. Ann Surg. 2011;254(4):633–40.
Gupta PK, Franck C, Miller WJ, et al. Development and validation of a bariatric surgery morbidity risk calculator using the prospective, multicenter NSQIP dataset. J Am Coll Surg. 2011;212(3):301–9.
Stenberg E, Cao Y, Szabo E, et al. Risk prediction model for severe postoperative complication in bariatric surgery. Obes Surg. 2018;28(7):1869–75.
Stenberg E, Persson C, Näslund E, et al. The impact of socioeconomic factors on the early postoperative complication rate after laparoscopic gastric bypass surgery. Surg Obes Relat Dis. 2019;15(4):575–81.
Wright JP, Edwards GC, Goggins K, et al. Association of health literacy with postoperative outcomes in patients undergoing major abdominal surgery. JAMA Surg. 2018;153(2):137–42.
Venkataramani AS, Brigell R, O'Brien R, et al. Economic opportunity, health behaviours, and health outcomes in the USA: a population-based cross-sectional study. Lancet Public Health. 2016;1(1):e18–25.
Murray TC, Rodgers WM, Fraser SN. Exploring the relationship between socioeconomic status, control beliefs and exercise behavior: a multiple mediator model. J Behav Med. 2012;35(1):63–73.
Seyssel K, Suter M, Pattou F, et al. A predictive model of weight loss after roux-en-Y gastric bypass up to 5 years after surgery: a useful tool to select and manage candidates to bariatric surgery. Obes Surg. 2018;28(11):3393–9.
Ortega E, Morinigo R, Flores L, et al. Predictive factors of excess body weight loss 1 year after laparoscopic bariatric surgery. Surg Endosc. 2012;26(6):1744–50.
Al-Khyatt W, Ryall R, Leeder P, et al. Predictors of inadequate weight loss after laparoscopic gastric bypass for morbid obesity. Obes Surg. 2017;27(6):1446–52.
Adams ST, Salhab M, Hussain ZI, et al. Roux-en-Y gastric bypass for morbid obesity: what are the preoperative predictors of weight loss? Postgrad Med J. 2013;89(1053):411–6.
Alfa Wali M, Ashrafian H, Schofield KL, et al. Is social deprivation associated with weight loss outcomes following bariatric surgery? A 10-year single institutional experience. Obes Surg. 2014;24(12):2126–32.
Courcoulas AP, King WC, Belle SH, et al. Seven-year weight trajectories and health outcomes in the longitudinal assessment of bariatric surgery (LABS) study. JAMA Surg. 2018;153(5):427–34.
Higa K, Ho T, Tercero F, et al. Laparoscopic Roux-en-Y gastric bypass: 10-year follow-up. Surg Obes Relat Dis. 2011;7(4):516–25.
Acknowledgements
Open access funding provided by Örebro University.
Funding
The study was supported by grants from Örebro County Council, Örebro University, Stockholm County Council, SRP Diabetes and the NovoNordisk Foundation. None of the supporting agents had any influence on the contents of this article.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Erik Näslund received grants from Stockholm County Council, SRP Diabetes and NovoNordisk Foundation during the conduct of the study. None of the other authors declare any conflict of interest.
Ethical Approval
The study was conducted in accordance with the ethical standards of the 1964 Helsinki Declaration and its later amendments and with the approval of the regional ethics committee in Uppsala, Sweden.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Stenberg, E., Mohseni, S., Cao, Y. et al. Limited Effect of Beta-blockade on Postoperative Outcome After Laparoscopic Gastric Bypass Surgery. OBES SURG 30, 139–145 (2020). https://doi.org/10.1007/s11695-019-04108-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-019-04108-8