Abstract
Background
Visceral fat (VF) plays a major role in the development of metabolic syndrome associated with obesity. The aim of our study is to compare VF and subcutaneous fat (SCF) reduction measured by ultrasonography (US) after laparoscopic adjustable gastric banding (LAGB), laparoscopic sleeve gastrectomy (LSG), and laparoscopic Roux-En-Y gastric bypass (LRYGB).
Methods
Thirty-nine morbidly obese patients were prospectively evaluated by US before surgery and 3, 6, and 12 months following surgery to determine VF and SCF thickness.
Results
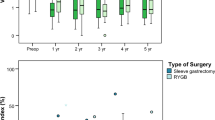
Three statistically comparable groups of morbidly obese patients underwent LRYGB (n = 13), LSG (n = 15), and LAGB (n = 11). The three groups did not differ in initial age, gender, body mass index (BMI), VF, or SCF. Final excess weight loss (EWL%) was highest after LSG and LRYGB followed by LAGB (81 ± 5.8 vs. 69.5 ± 4.5 vs. 43.4 ± 5.2, p < 0.001). LSG and LRYGB were significantly more efficient in VF reduction (ΔVF) compared with LAGB (7.1 ± 0.5 vs. 5.6 ± 0.6 vs. 3.6 ± 0.8, p = 0.004). SCF reduction (ΔSCF) was also highest after LSG followed by LRYGB and LAGB (3 ± 0.2 vs. 2.2 ± 0.4 vs. 1.9 ± 0.4, p = 0.08). The change in fat distribution, determined as Δ(VF/SCF), showed a preferential VF reduction in the LSG and LRYGB patients compared with patients that underwent LAGB (0.59 ± 0.1 vs. 0.52 ± 0.2 vs. 0.19 ± 0.2, p = 0.42). In a subgroup analysis comparing only LSG to LRYGB, no statistically significant difference was seen in EWL%, ΔVF, ΔSCF, or in fat distribution Δ(VF/SCF).
Conclusion
LSG and LRYGB show better preferential and overall VF reduction than LAGB. US may serve as a simple tool of evaluating postoperative fat distribution.



Similar content being viewed by others
Abbreviations
- US:
-
Ultrasonography
- VF:
-
Visceral fat
- SCF:
-
Subcutaneous fat
- LAGB:
-
Laparoscopic adjustable gastric banding
- LSG:
-
Laparoscopic sleeve gastrectomy
- LRYGB:
-
Laparoscopic Roux-en-Y gastric bypass
- BMI:
-
Body mass index
References
Allison DB, Fontaine KR, Manson JE, Stevens J, VanItallie TB. Annual deaths attributable to obesity in the United States. JAMA. 1999;282:1530–8.
Haslam DW, James WP. Obesity. Lancet. 2005;366:1197–209.
Bjorntorp P. “Portal” adipose tissue as a generator of risk factors for cardiovascular disease and diabetes. Arteriosclerosis. 1990;10:493–6.
Ahima RS, Flier JS. Adipose tissue as an endocrine organ. Trends Endocrinol Metab. 2000;11:327–32.
Bjorntorp P. Metabolic implications of body fat distribution. Diabetes Care. 1991;14:1132–43.
Frayn KN. Visceral fat and insulin resistance—causative or correlative? Br J Nutr. 2000;83(1):S71–7.
Wajchenberg BL. Subcutaneous and visceral adipose tissue: their relation to the metabolic syndrome. Endocr Rev. 2000;21:697–738.
Schneider HJ, Glaesmer H, Klotsche J, Bohler S, Lehnert H, et al. Accuracy of anthropometric indicators of obesity to predict cardiovascular risk. J Clin Endocrinol Metab. 2007;92:589–94.
Lemieux I, Drapeau V, Richard D, Bergeron J, Marceau P, et al. Waist girth does not predict metabolic complications in severely obese men. Diabetes Care. 2006;29:1417–9.
Stolk RP, Meijer R, Mali WP, Grobbee DE, van der Graaf Y. Ultrasound measurements of intraabdominal fat estimate the metabolic syndrome better than do measurements of waist circumference. Am J Clin Nutr. 2003;77:857–60.
Kim SK, Park SW, Kim SH, Cha BS, Lee HC, et al. Visceral fat amount is associated with carotid atherosclerosis even in type 2 diabetic men with a normal waist circumference. Int J Obes. 2009;33:131–5.
Rossner S, Bo WJ, Hiltbrandt E, Hinson W, Karstaedt N, et al. Adipose tissue determinations in cadavers—a comparison between cross-sectional planimetry and computed tomography. Int J Obes. 1990;14:893–902.
van der Kooy K, Seidell JC. Techniques for the measurement of visceral fat: a practical guide. Int J Obes Relat Metab Disord. 1993;17:187–96.
Abate N, Garg A, Coleman R, Grundy SM, Peshock RM. Prediction of total subcutaneous abdominal, intraperitoneal, and retroperitoneal adipose tissue masses in men by a single axial magnetic resonance imaging slice. Am J Clin Nutr. 1997;65:403–8.
Liu KH, Chan YL, Chan WB, Kong WL, Kong MO, et al. Sonographic measurement of mesenteric fat thickness is a good correlate with cardiovascular risk factors: comparison with subcutaneous and preperitoneal fat thickness, magnetic resonance imaging and anthropometric indexes. Int J Obes Relat Metab Disord. 2003;27:1267–73.
Heath ML, Kow L, Slavotinek JP, Valentine R, Toouli J, et al. Abdominal adiposity and liver fat content 3 and 12 months after gastric banding surgery. Metab Clin Exp. 2009;58:753–8.
Armellini F, Zamboni M, Rigo L, Todesco T, Bergamo-Andreis IA, et al. The contribution of sonography to the measurement of intra-abdominal fat. J Clin Ultrasound. 1990;18:563–7.
Ribeiro-Filho FF, Faria AN, Azjen S, Zanella MT, Ferreira SR. Methods of estimation of visceral fat: advantages of ultrasonography. Obes Res. 2003;11:1488–94.
Pontiroli AE, Pizzocri P, Giacomelli M, Marchi M, Vedani P, et al. Ultrasound measurement of visceral and subcutaneous fat in morbidly obese patients before and after laparoscopic adjustable gastric banding: comparison with computerized tomography and with anthropometric measurements. Obes Surg. 2002;12:648–51.
De Lucia RE, Sleigh A, Finucane FM, Brage S, Stolk RP, et al. Ultrasound measurements of visceral and subcutaneous abdominal thickness to predict abdominal adiposity among older men and women. Obesity (Silver Spring). 2010;18:625–31.
Del Genio F, Del Genio G, De Sio I, Marra M, Alfonsi L, et al. Noninvasive evaluation of abdominal fat and liver changes following progressive weight loss in severely obese patients treated with laparoscopic gastric bypass. Obes Surg. 2009;19:1664–71.
Pontiroli AE, Pizzocri P, Librenti MC, Vedani P, Marchi M, et al. Laparoscopic adjustable gastric banding for the treatment of morbid (grade 3) obesity and its metabolic complications: a three-year study. J Clin Endocrinol Metab. 2002;87:3555–61.
Busetto L, Perini P, Giantin V, Valente P, Segato G, et al. Relationship between energy expenditure and visceral fat accumulation in obese women submitted to adjustable silicone gastric banding (ASGB). Int J Obes Relat Metab Disord. 1995;19:227–33.
Busetto L, Tregnaghi A, De Marchi F, Segato G, Foletto M, et al. Liver volume and visceral obesity in women with hepatic steatosis undergoing gastric banding. Obes Res. 2002;10:408–11.
Busetto L, Tregnaghi A, Bussolotto M, Sergi G, Beninca P, et al. Visceral fat loss evaluated by total body magnetic resonance imaging in obese women operated with laparascopic adjustable silicone gastric banding. Int J Obes Relat Metab Disord. 2000;24:60–9.
Djuric-Stefanovic A, Vasin D, Jovanovic S, Lazic L, Kovac J, et al. Ultrasonographic evaluation of visceral and subcutaneous abdominal fat tissue before and after bariatric surgery. Acta Chir Lugosl. 2013;60:25–30.
Despres JP, Lemieux I. Abdominal obesity and metabolic syndrome. Nature. 2006;444:881–7.
Vidal J, Ibarzabal A, Romero F, Delgado S, Momblan D, et al. Type 2 diabetes mellitus and the metabolic syndrome following sleeve gastrectomy in severely obese subjects. Obes Surg. 2008;18:1077–82.
Schauer PR, Kashyap SR, Wolski K, Brethauer SA, Kirwan JP, et al. Bariatric surgery versus intensive medical therapy in obese patients with diabetes. N Engl J Med. 2012;366:1567–76.
Jimenez A, Casamitjana R, Flores L, Viaplana J, Corcelles R, et al. Long-term effects of sleeve gastrectomy and Roux-en-Y gastric bypass surgery on type 2 diabetes mellitus in morbidly obese subjects. Ann Surg. 2012;256:1023–9.
Abbatini F, Rizzello M, Casella G, Alessandri G, Capoccia D, et al. Long-term effects of laparoscopic sleeve gastrectomy, gastric bypass, and adjustable gastric banding on type 2 diabetes. Surg Endosc. 2010;24:1005–10.
Ernst B, Wilms B, Thurnheer M, Schultes B. Reduced circulating androgen levels after gastric bypass surgery in severely obese women. Obes Surg. 2013;23:602–7.
Pellitero S, Olaizola I, Alastrue A, Martinez E, Granada ML, et al. Hypogonadotropic hypogonadism in morbidly obese males is reversed after bariatric surgery. Obes Surg. 2012;22:1835–42.
Botella-Carretero JI, Balsa JA, Gomez-Martin JM, Peromingo R, Huerta L, et al. Circulating free testosterone in obese men after bariatric surgery increases in parallel with insulin sensitivity. J Endocrinol Investig. 2013;36:227–32.
Conflicts of Interest
All contributing authors declare that they have no conflicts of interest to report.
All procedures performed in the study involving human participants were in accordance with the ethical standards of the institutional and national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent was obtained from all individual participants included in the study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mizrahi, I., Beglaibter, N., Simanovsky, N. et al. Ultrasound Evaluation of Visceral and Subcutaneous Fat Reduction in Morbidly Obese Subjects Undergoing Laparoscopic Gastric Banding, Sleeve Gastrectomy, and Roux-en-Y Gastric Bypass: A Prospective Comparison Study. OBES SURG 25, 959–966 (2015). https://doi.org/10.1007/s11695-014-1495-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-014-1495-4