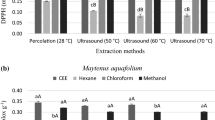
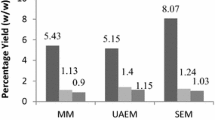
Abstract
In this study, the effects of extraction solvents on the yield and total phenolic content of Vernonia amygdalina leaf were examined. The considered solvents were water, 20% v/v ethanol, 40% v/v ethanol, 60% v/v ethanol, 80% v/v ethanol, and 100% v/v ethanol using Soxhlet extraction technique. Highest extraction yield (19.45 ± 0.22% g/g) and total phenolic content (96.25 ± 2.52 mg GAE/g d.w.) were achieved using 60% v/v aqueous ethanol solution. In addition, there exist a significant correlation between total phenolic content and antioxidant activities using DPPH (R2 = 0.9397, p < 0.05) and ABTS scavenging activity (R2 = 0.7700, p < 0.05). More so, the FTIR analysis confirmed the presence of functional groups attributed to its antioxidant property. There was a significant damage in the cell wall of V. amygdalina leaf after extracted with 60% v/v ethanol solution. Therefore, V. amygdalina leaf can be a good source of antioxidants.




Similar content being viewed by others
References
O.R. Alara, J.A. Alara, O.A. Olalere, Review on Phaleria macrocarpa pharmacological and phytochemical properties. Drug Des 5, 1–5 (2016)
O.R. Alara, S.K. Abdul Mudalip, O.A. Olalere, Optimization of mangiferin extrated from Phaleria macrocarpa fruits using response surface methodology. J. Appl. Res. Med. Aromat. Plants 5, 82–87 (2017)
Y. Xiong, L. Rao, L. Xiong, Q. Ai, X. Wu, Effects of extraction solvent on polyphenolic contents and antioxidant activities of Osmanthus fragrans’ seed. Proc.-2012 Int. Conf. Biomed. Eng. Biotechnol. 217–220 (2012)
K.M. Ramkumar, P. Rajaguru, R. Ananthan, Antimicrobial properties and phytochemical constituents of an antidiabetic plant Gymnema montanum. Food Chem 1, 67–71 (2007)
S.K. Yeap, W.Y. Ho, B.K. Beh, W.S. Liang, H. Ky, A. Hadi, N. Yousr, N.B. Alitheen, Vernonia amygdalina, an ethnomedical used green vegetable with multiple bio-activities. J. Med. Plants Res 4, 2787–2812 (2010)
B. Adaramola, A. Onigbinde, Effect of extraction solvent on the phenolic content, flavonoid content and antioxidant capacity of clove bud. IOSR J. Pharm. Biol. Sci 11, 33–38 (2016)
N.J. Toyang, R. Verpoorte, A review of the medicinal potentials of plants of the genus Vernonia (Asteraceae). J. Ethnopharmacol. 146, 681–723 (2013)
G.O. Igile, W. Oleszek, M. Jurzysta, S. Burda, M. Fafunso, A.A. Fasanmade, Flavonoids from Vernonia amygdalina and their antioxidant activities. J. Agric. Food Chem. 42, 2445–2448 (1994)
F. Dahmounea, G. Spignob, K. Moussia, H. Reminia, A. Cherbalc, K. Madania, Pistacia lentiscus leaves as a source of phenolic compounds: microwave-assisted extraction optimized and compared with ultrasound-assisted and conventional solvent extraction. Ind. Crops Prod. 61, 31–40 (2014)
X. Luo, Y. Jiang, F.R. Fronczek, C. Lin, E.B. Izevbigie, K.S. Lee, Isolation and structure determination of a sesquiterpene lactone (vernodalinol) from Vernonia amygdalina extracts. Pharm. Biol. 49, 464–470 (2011)
K. Oriakhi, E.I. Oikeh, N. Ezeugwu, O. Anoliefo, O. Aguebor, E.S. Omoregie, Comparative antioxidant activities of extracts of Vernonia amygdalina and Ocimum gratissimum leaves. J. Agric. Sci. 6, 13–20 (2013)
E.O. Farombi, O. Owoeye, Antioxidative and chemopreventive properties of Vernonia amygdalina and Garcinia biflavonoid. Int. J. Environ. Res. Public Health 8, 2533–2555 (2011)
U. Zlotek, S. Mikulska, M. Nagajek, M. Swieca, The effect of different solvents and number of extraction steps on the polyphenol content and antioxidant capacity of basil leaves (Ocimum basilicum L.) extracts. Saudi. J. Biol. Sci. 23, 628–633 (2016)
O.R. Alara, N.H. Abdurahman, O.A. Olalere, Ethanolic extraction of flavonoids, phenolics and antioxidants from Vernonia amygdalina leaf using two-level factorial design. J. King Saud Univ. Sci. (2017). doi.10.1016/j.jksus.2017.08.001
G.A. Ayoola, H.A.B. Coker, S.A. Adesegun, A.A. Adepoju-Bello, K. Obaweya, E.C. Ezennia, T.O. Atangbayila, Phytochemical screening and antioxidant activities of some selected medicinal plants used for malaria therapy in Southwestern Nigeria. Trop. J. Pharm. Res. 7, 1019–1024 (2008)
M.N. Alam, N.J. Bristi, M. Rafiquzzaman, Review on in vivo and in vitro methods evaluation of antioxidant activity. Saudi Pharm. J. 21, 143–152 (2013)
K. Thaipong, U. Boonprakob, K. Crosby, L. Cisneros-zevallos, D. Hawkins, Comparison of ABTS, DPPH, FRAP, and ORAC assays for estimating antioxidant activity from guava fruit extracts. J. Food Compos. Anal. 19, 669–675 (2006)
R. Ashokkumar, M. Ramaswamy, Phytochemical screening by FTIR spectroscopic analysis of leaf extracts of selected Indian medicinal plants. Int. J. Curr. Microbiol. Appl. Sci. 3, 395–406 (2014)
Q.D. Doa, A.E. Angkawijaya, P.L. Tran-Nguyena, L.H. Huynhb, F.E. Soetaredjo, S. Ismadji, Y.-H. Ju, Effect of extraction solvent on total phenol content, total flavonoid content, and antioxidant activity of Limnophila aromatica. J. Food Drug Anal. 22, 296–302 (2014)
M. Dhobi, V. Mandal, S. Hemalatha, Optimization of microwave assisted extraction of bioactive flavonolignan - silybinin. J. Chem. Metrol. 3, 13–23 (2009)
L. Tomsone, Z. Kruma, R. Galoburda, Comparison of different solvents and extraction methods for isolation of phenolic compounds from horseradish roots. Int. Sch. Sci. Res. Innov. 6, 1164–1169 (2012)
G. Hithamani, K. Ramalakshmi, Microwave assisted extraction of phenolics from Origanum vulgare. Int. Invent. J. Agric. Soil Sci. 1, 7–12 (2013)
F. Dahmoune, B. Nayak, K. Moussi, H. Remini, K. Madani, Optimization of microwave-assisted extraction of polyphenols from Myrtus communis L. leaves. Food Chem. 166, 585–595 (2015)
Acknowledgements
The authors wish to acknowledge the financial support of Universiti Malaysia Pahang through the soft grant RDU160325.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest on this study.
Rights and permissions
About this article
Cite this article
Alara, O.R., Abdurahman, N.H., Mudalip, S.K.A. et al. Characterization and effect of extraction solvents on the yield and total phenolic content from Vernonia amygdalina leaves. Food Measure 12, 311–316 (2018). https://doi.org/10.1007/s11694-017-9642-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11694-017-9642-y