Abstract
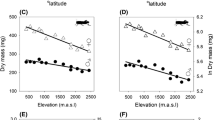
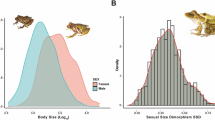
Two macroecological and evolutionary rules are strongly related to the body size of organisms: Bergmann’s and Rensch’s rules. Bergmann’s rule states that organisms are larger in colder regions (high latitudes). Rensch’s rule states that sexual size dimorphism increases when males are larger. Organisms with widespread distribution and resource-mediated growth—such as hermit crabs and their gastropod shells—become excellent models for investigating these patterns. This study is the first to address macroecological and evolutionary patterns in body size among populations and also among sex of the three species of hermit crabs of the genus Clibanarius: C. antillensis, C. sclopetarius and C. symmetricus throughout their distribution. This research included systematic review of data from specialized literature along with primary data by traditional morphometrics of intersexual and populational average body size of the three different species. Regression models were designed to assess the rules separately and altogether. We have observed that the three species of Clibanarius showed interpopulational clines consistent with Bergmann’s rule. Surprisingly, our findings showed a gender-specific divergence from females as a response to latitudinal gradients, suggesting that latitude (as a proxy) increased the interpopulational body effect only in females. We suggest that phenotypic plasticity due to decreased selective pressure with higher latitude (decreased temperature) and greater productivity may affect the bias of these rules. Our data also suggest that female body size variation is modulated by the selection of fecundity in body size.





Similar content being viewed by others
Data Availability
All data analyzed and generated during this study are included in this publish.
References
Alencar, C. E. R. D., Vale, V. F., Moraes, S. A. S. N., Araújo, P. V. N., & Freire, F. A. M. (2017). New record of the six-holed keyhole urchin, Leodia sexiesperforata (Leske, 1778) (Clypeasteroida, Mellitidae), from the Brazilian coast, with an updated distribution map. Check List, 13, 597. https://doi.org/10.15560/13.5.597
Andersson, M., & Iwasa, Y. (1996). Sexual selection. Trends in Ecology & Evolution, 11(2), 53–58. https://doi.org/10.1016/0169-5347(96)81042-1
Asakura, A. (1995). Sexual differences in life history and resource utilization by the hermit crab. Ecology, 76(7), 2295–2313. https://doi.org/10.2307/1941703
Ashton, K. G. (2002). Do amphibians follow Bergmann’s rule? Canadian Journal of Zoology, 80(4), 708–716. https://doi.org/10.1139/z02-049
Atkinson, D. (1994). Temperature and organism size: A biological law for ectotherms? Advances in Ecological Research, 25, 1–58.
Bach, C., Hazlett, B., & Rittschof, D. (1976). Effects of interspecific competition on fitness of the hermit crab Clibanarius tricolor. Ecology, 57(3), 579–586. https://doi.org/10.2307/1936442
Băncilǎ, R. V., Gelder, I., Rotteveel, E., Loman, J., & Arntzen, J. W. (2010). Fluctuating asymmetry is a function of population isolation in island lizards. Journal of Zoology, 282(4), 266–275. https://doi.org/10.1111/j.1469-7998.2010.00736.x
Belk, M. C., & Houston, D. D. (2002). Bergmann’s rule in ectotherms: A test using freshwater fishes. The American Naturalist, 160(6), 803–808.
Bergmann, C. (1847). Über die Verhältnisse der Wärmeökonomie der Thiere zu ihrer Grösse. Vandenhoeck und Ruprecht.
Berrigan, D., & Charnov, E. L. (1994). Reaction norms for age and size at maturity in response to temperature: a puzzle for life historians. Oikos. https://doi.org/10.2307/3545787
Bertness, M. D. (1981). Interference, exploitation, and sexual components of competition in a tropical hermit crab assemblage. Journal of Experimental Marine Biology and Ecology, 49(2–3), 189–202. https://doi.org/10.1016/0022-0981(81)90070-8
Bidau, C. J., & Martí, D. A. (2007). Clinal variation of body size in Dichroplus pratensis (Orthoptera, Acrididae), inversion of Bergmann’s and Rensch’s rules. Annals of the Entomological Society of America, 100(6), 850–860. https://doi.org/10.1603/0013-8746(2007)100[850:CVOBSI]2.0.CO;2
Bidau, C. J., Martí, D. A., & Baldo, D. (2011). Inter-and intraspecific geographic variation of body size in South American redbelly toads of the genus Melanophryniscus Gallardo, 1961 (Anura: Bufonidae). Journal of Herpetology, 45(1), 66–74. https://doi.org/10.1670/09-202.1
Blackburn, T. M., Gaston, K. J., & Loder, N. (1999). Geographic gradients in body size: A clarification of Bergmann’s rule. Diversity and Distributions, 5(4), 165–174. https://doi.org/10.1046/j.1472-4642.1999.00046.x
Blackstone, N. W. (1985). The effects of shell size and shape on growth and form in the hermit crab Pagurus longicarpus. The Biological Bulletin, 168(1), 75–90.
Blanckenhorn, W. U. (2000). The evolution of body size: What keeps organisms small? The Quarterly Review of Biology, 75(4), 385–407. https://doi.org/10.1086/393620
Blanckenhorn, W. U. (2005). Behavioral causes and consequences of sexual size dimorphism. Ethology, 111(11), 977–1016. https://doi.org/10.1111/j.1439-0310.2005.01147.x
Blanckenhorn, W. U., & Demont, M. (2004). Bergmann and converse Bergmann latitudinal clines in arthropods: Two ends of a continuum? Integrative and Comparative Biology, 44(6), 413–424. https://doi.org/10.1093/icb/44.6.413
Blanckenhorn, W. U., Stillwell, R. C., Young, K. A., Fox, C. W., & Ashton, K. G. (2006). When Rensch meets Bergmann, does sexual size dimorphism change with latitude? Evolution, 60, 2004–2011. https://doi.org/10.1111/j.0014-3820.2006.tb01838.x
Brahim, A., Mustapha, N., & Marshall, D. J. (2019). Non-reversible and reversible heat tolerance plasticity in tropical intertidal animals: Responding to habitat temperature heterogeneity. Frontiers in Physiology, 9, 1909. https://doi.org/10.3389/fphys.2018.01909
Carlon, D. B., & Ebersole, J. P. (1995). Life-history variation among three temperate hermit crabs: The importance of size in reproductive strategies. The Biological Bulletin, 188(3), 329–337. https://doi.org/10.2307/1542309
Castiglioni, D. D. S., De Lima, C., Da Silva, J. V., & Azevedo, D. D. S. (2011). Relative growth and its use to determine the morphological sexual maturity of Ucides cordatus (Linnaeus, 1763) (Decapoda, Brachyura, Ucididae) from two mangrove areas on the northeastern Brazilian coast. Crustaceana, 84(10), 1221. https://doi.org/10.1163/001121611X590139
Castilho, A. L., Gavio, M. A., Costa, R. C., Boschi, E. E., Bauer, R. T., & Fransozo, A. (2007). Latitudinal variation in population structure and reproductive pattern of the endemic South American shrimp Artemesia longinaris (Decapoda: Penaeoidea). Journal of Crustacean Biology, 27(4), 548–552. https://doi.org/10.1651/S-2788.1
Castillo, R., Charmantier, G., & Thuet, P. (1988). Osmotic regulation in the hermit crab Clibanarius erythropus. Biochemical Systematics and Ecology, 16(3), 325–328. https://doi.org/10.1016/0305-1978(88)90018-X
Clarke, M. R. B. (1980). The reduced major axis of a bivariate sample. Biometrika, 67(2), 441–446.
Colpo, K. D., & López-Greco, L. S. (2017). Temperature influences the reproduction of fiddler crabs at the southern edge of their distribution. Invertebrate Biology, 136(2), 171–183. https://doi.org/10.1111/ivb.12168
Contreras-Garduño, J., & Córdoba-Aguilar, A. (2006). Sexual selection in hermit crabs: A review and outlines of future research. Journal of Zoology, 270(4), 595–605. https://doi.org/10.1111/j.1469-7998.2006.00182.x
Da Costa, R. C., Branco, J. O., Machado, I. F., Campos, B. R., & Avila, M. G. (2010). Population biology of shrimp Artemesia longinaris (Crustacea: Decapoda: Penaeidae) from the southern coast of Brazil. Journal of the Marine Biological Association of the United Kingdom, 90(4), 663–669. https://doi.org/10.1017/S002531540999124X
Da Costa, R. C. D., & Fransozo, A. (2004). Reproductive biology of the shrimp Rimapenaeus constrictus (Decapoda, Penaeidae) in the Ubatuba region of Brazil. Journal of Crustacean Biology, 24(2), 274. https://doi.org/10.1651/C-2437
Danin, A. P. F., Pombo, M., Martinelli-Lemos, J. M., dos Santos, C. R. M., Aviz, D., & Petracco, M. (2020). Population ecology of the hermit crab Clibanarius symmetricus (Anomura: Diogenidae) on an exposed beach of the Brazilian Amazon coast. Regional Studies in Marine Science, 33, 100944. https://doi.org/10.1016/j.rsma.2019.100944
Darwin, C. (1871). The descent of man and selection in relation to sex. Murray.
Duffy, J. E., & Thiel, M. (2007). Evolutionary ecology of social and sexual systems: crustaceans as model organisms. Oxford University Press.
Fairbairn, D. J. (1997). Allometry for sexual size dimorphism: Pattern and process in the coevolution of body size in males and females. Annual Review of Ecology and Systematics, 28(1), 659–687.
Fairbairn, D. J. (2005). Allometry for sexual size dimorphism: Testing two hypotheses for Rensch’s rule in the water strider Aquarius remigis. The American Naturalist, 166(S4), S69–S84. https://doi.org/10.1086/444600
Flachshaar, D., & Piel, J. (2008). Symbiosis, chemical biology of. Wiley Encyclopedia of Chemical Biology. https://doi.org/10.1002/9780470048672.wecb571
Floeter, S. R., Nalesso, R. C., Rodrigues, M. M. P., & Turra, A. (2000). Patterns of shell utilization and selection in two sympatric hermit crabs (Anomura: Diogenidae) in south-eastern Brazil. Journal of the Marine Biological Association of the United Kingdom, 80(6), 1053–1059. https://doi.org/10.1017/S0025315400003118
Forster, J., Hirst, A. G., & Atkinson, D. (2011). How do organisms change size with changing temperature? The importance of reproductive method and ontogenetic timing. Functional Ecology, 25(5), 1024–1031. https://doi.org/10.1111/j.1365-2435.2011.01852.x
Forster, J., Hirst, A. G., & Atkinson, D. (2012). Warming-induced reductions in body size are greater in aquatic than terrestrial species. Proceedings of the National Academy of Sciences, 109(47), 19310–19314. https://doi.org/10.1073/pnas.1210460109
Fraaije, R. H. (2014). Diverse Late Jurassic anomuran assemblages from the Swabian Alb and evolutionary history of paguroids based on carapace morphology. Neues Jahrbuch Für Geologie Und Paläontologie-Abhandlungen, 273(2), 121–145. https://doi.org/10.1127/0077-7749/2014/0419
Frameschi, I. F., Andrade, L. S. D., Alencar, C. E. R. D., Teixeira, G. M., Fransozo, V., Fernandes-Góes, L. C., & Fransozo, A. (2014b). Life-history traits of the red brocade hermit crab Dardanus insignis on the subtropical Brazilian coast. Marine Biology Research, 11(3), 283–293. https://doi.org/10.1080/17451000.2014.923100
Frameschi, I. F., Paula, B. C., Fernandes-Góes, L. C., Alencar, C. E. R. D., Fransozo, A., & Freire, F. A. M. (2014a). Gastropod shells used by Dardanus insignis (Decapoda, Anomura) near Islands in the Ubatuba region, São Paulo, Brazil. Bioscience, 30(5), 1563–1572.
Fransozo, A., & Mantelatto, F. L. (1998). Population structure and reproductive period of the tropical hermit crab Calcinus tibicen (Decapoda: Diogenidae) in the region of Ubatuba, São Paulo, Brazil. Journal of Crustacean Biology, 18(4), 738–745. https://doi.org/10.1163/193724098X00610
García-Navas, V., Noguerales, V., Cordero, P. J., & Ortego, J. (2017). Ecological drivers of body size evolution and sexual size dimorphism in short-horned grasshoppers (Orthoptera: Acrididae). Journal of Evolutionary Biology. https://doi.org/10.1111/jeb.13131
Gherardi, F. (1990). Competition and coexistence in two Mediterranean hermit crabs, Calcinus ornatus (Roux) and Clibanarius erythropus (Latreille) (Decapoda, Anomura). Journal of Experimental Marine Biology and Ecology, 143(3), 221–238. https://doi.org/10.1016/0022-0981(90)90072-K
Hartnoll, R. G. (1982). Growth. In D. E. Bliss (Ed.), The biology of crustacea, embryology, morphology, and genetics (pp. 11–196). Academic Press Inc.
Hartnoll, R. G. (2001). Growth in Crustacea—twenty years on. Advances in decapod Crustacean research (pp. 111–122). Springer.
Harvey, A. W. (1990). Sexual differences in contemporary selection acting on size in the hermit crab Clibanarius digueti. The American Naturalist, 136(3), 292–304. https://doi.org/10.1086/285099
Hazlett, B. A. (1981). The behavioral ecology of hermit crabs. Annual Review of Ecology and Systematics, 12(1), 1–22.
Hazlett, B. A. (1989). Mating success of male hermit crabs in shell generalist and shell specialist species. Behavioral Ecology and Sociobiology, 25(2), 119–128. https://doi.org/10.1007/BF00302928
Hazlett, B. A., & Baron, L. C. (1989). Influence of shells on mating behavior in the hermit crab Calcinus tibicen. Behavioral Ecology and Sociobiology, 24(6), 369–376. https://doi.org/10.1007/BF00293264
Hines, A. H. (1989). Geographic variation in size at maturity in brachyuran crabs. Bulletin of Marine Science, 45(2), 356–368.
Hirose, G. L., Fransozo, V., Tropea, C., López-Greco, L. S., & Negreiros-Fransozo, M. L. (2013). Comparison of body size, relative growth and size at onset sexual maturity of Uca uruguayensis (Crustacea: Decapoda: Ocypodidae) from different latitudes in the south-western Atlantic. JOurnal of the Marine Biological Association of the United Kingdom, 93(3), 781–788. https://doi.org/10.1017/S0025315412001038
Ho, C. K., Pennings, S. C., & Carefoot, T. H. (2010). Is diet quality an overlooked mechanism for Bergmann’s rule? The American Naturalist, 175(2), 269–276. https://doi.org/10.1086/649583
Huston, M. A., & Wolverton, S. (2009). The global distribution of net primary production: Resolving the paradox. Ecological Monographs, 79(3), 343–377. https://doi.org/10.1890/08-0588.1
Huston, M. A., & Wolverton, S. (2011). Regulation of animal size by eNPP, Bergmann’s rule and related phenomena. Ecological Monographs, 81(3), 349–405. https://doi.org/10.1890/10-1523.1
Imazu, M., & Asakura, A. (1994). Distribution, reproduction and shell utilization patterns in three species of intertidal hermit crabs on a rocky shore on the Pacific coast of Japan. Journal of Experimental Marine Biology and Ecology, 184(1), 41–65. https://doi.org/10.1016/0022-0981(94)90165-1
Jones, A. G., & Ratterman, N. L. (2009). Mate choice and sexual selection: What have we learned since Darwin? Proceedings of the National Academy of Sciences, 106(1), 10001–10008. https://doi.org/10.1073/pnas.0901129106
Jones, M. B., & Simons, M. J. (1983). Latitudinal variation in reproductive characteristics of a mud crab, Helice crassa (Grapsidae). Bulletin of Marine Science, 33(3), 656–670.
Keiler, J., Wirkner, C. S., & Richter, S. (2017). One hundred years of carcinization–the evolution of the crab-like habitus in Anomura (Arthropoda: Crustacea). Biological Journal of the Linnean Society, 121(1), 200–222. https://doi.org/10.1093/biolinnean/blw031
Kendall, M. G., & Stuart, A. (1973). The advanced theory of statistics. Charles Green.
Kingsolver, J. G., & Huey, R. B. (2008). Size, temperature, and fitness: Three rules. Evolutionary Ecology Research, 10(2), 251–268.
Lagerspetz, K. Y., & Vainio, L. A. (2006). Thermal behaviour of crustaceans. Biological Reviews, 81(2), 237–258. https://doi.org/10.1017/S1464793105006998
Lardies, M., & Castilla, J. (2001). Latitudinal variation in the reproductive biology of the commensal crab Pinnaxodes chilensis (Decapoda: Pinnotheridae) along the Chilean coast. Marine Biology, 139(6), 1125–1133. https://doi.org/10.1007/s002270100661
Lardies, M. A., Medina, M. H., & Correa, J. A. (2008). Intraspecific biogeographic pattern breakage in the snapping shrimp Betaeus emarginatus caused by coastal copper mine tailings. Marine Ecology Progress Series, 358, 203–210. https://doi.org/10.3354/meps07347
Lardies, M. A., & Wehrtmann, I. S. (2001). Latitudinal variation in the reproductive biology of Betaeus truncatus (Decapoda: Alpheidae) along the Chilean coast. Ophelia, 55(1), 55–67. https://doi.org/10.1080/00785236.2001.10409473
Lazić, M. M., Carretero, M. A., Crnobrnja-Isailović, J., & Kaliontzopoulou, A. (2015). Effects of environmental disturbance on phenotypic variation: An integrated assessment of canalization, developmental stability, modularity, and allometry in lizard head shape. The American Naturalist, 185(1), 44–58. https://doi.org/10.1086/679011
Levene, H. (1960). Robust tests for equality of variances. In I. Olkin (Ed.), Contributions to probability and statistics (pp. 278–292). Stanford University.
Liao, W. B., Zeng, Y., Zhou, C. Q., & Jehle, R. (2013). Sexual size dimorphism in anurans fails to obey Rensch’s rule. Frontiers in Zoology, 10(1), 10. https://doi.org/10.1186/1742-9994-10-10
Lima-Filho, P. A., Bidau, C. J., Alencar, C. E. R. D., & Molina, W. F. (2017). Latitudinal influence on the sexual dimorphism of the marine fish Bathygobius soporator (Gobiidae: Teleostei). Evolutionary Biology, 44(3), 374–385. https://doi.org/10.1007/s11692-017-9416-9
Madeira, C., Leal, M. C., Diniz, M. S., Cabral, H. N., & Vinagre, C. (2018). Thermal stress and energy metabolism in two circumtropical decapod crustaceans: Responses to acute temperature events. Marine Environmental Research, 141, 148–158. https://doi.org/10.1016/j.marenvres.2018.08.015
Madeira, D., Mendonça, V., Dias, M., Roma, J., Costa, P. M., Larguinho, M., Vinagre, C., & Diniz, M. S. (2015). Physiological, cellular and biochemical thermal stress response of intertidal shrimps with different vertical distributions: Palaemon elegans and Palaemon serratus. Comparative Biochemistry and Physiology Part A: Molecular & Integrative Physiology, 183, 107–115. https://doi.org/10.1016/j.cbpa.2014.12.039
Mantelatto, F. L., Fernandes-Góes, L. C., Fantucci, M. Z., Biagi, R., Pardo, L. M., & de Góes, J. M. (2010). A comparative study of population traits between two South American populations of the striped-legged hermit crab Clibanarius vittatus. Acta Oecologica, 36(1), 10–15. https://doi.org/10.1016/j.actao.2009.09.003
Mantelatto, F. L. M., & Garcia, R. B. (1999). Reproductive potential of the hermit crab Calcinus tibicen (Anomura) from Ubatuba, São Paulo, Brazil. Journal of Crustacean Biology, 19(2), 268–275. https://doi.org/10.1163/193724099X00079
Mantelatto, F. L., & Sousa, L. M. (2000). Population biology of the hermit crab Paguristes tortugae Schmitt, 1933 (Anomura, Diogenidae) from Anchieta island, Ubatuba, Brazil. Nauplius, 8(2), 185–193.
Markham, J. C. (1968). Notes on growth patterns and shell-utilization of the hermit crab Pagurus bernhardus (L.). Ophelia, 5, 189–205. https://doi.org/10.1080/00785326.6812.10407609
Marshall, D. J., Rezende, E. L., Baharuddin, N., Choi, F., & Helmuth, B. (2015). Thermal tolerance and climate warming sensitivity in tropical snails. Ecology and Evolution, 5(24), 5905–5919. https://doi.org/10.1002/ece3.1785
Martinez, P. A., Marti, D. A., Molina, W. F., & Bidau, C. J. (2013). Bergmann’s rule across the equator: A case study in Cerdocyon thous (Canidae). Journal of Animal Ecology, 82(5), 997–1008. https://doi.org/10.1111/1365-2656.12076
McLaughlin, P. A., Komai, T., Lemaitre, R., & Rahayu, D. L. (2010). Annotated checklist of anomuran decapod crustaceans of the world (exclusive of the Kiwaoidea and families Chirostylidae and Galatheidae of the Galatheoidea) Part I Lithodoidea, Lomisoidea and Paguroidea. The Raffles Bulletin of Zoology, 23, 131–137.
Miloslavich, P., Klein, E., Díaz, J. M., Hernandez, C. E., Bigatti, G., Campos, L., Artigas, F., Castillo, J., Penchaszadeh, P. E., Neill, P. E., Carranza, A., Retana, M. V., Astarloa, J. M. D., Lewis, M., Yorio, P., Piriz, M. L., Rodríguez, D., Yoneshigue-Valentin, Y., Gamboa, L., & Martín, A. (2011). Marine biodiversity in the Atlantic and Pacific coasts of South America: Knowledge and gaps. PLoS ONE, 6(1), e14631. https://doi.org/10.1371/journal.pone.0014631
Mousseau, T. A. (1997). Ectotherms follow the converse to Bergmann’s rule. Evolution, 51(2), 630–632. https://doi.org/10.2307/2411138
NASA Ocean Biology (OB.DAAC). (2014). Mean annual sea surface temperature for the period 2009–2013 (composite dataset created by UNEP-WCMC). Data obtained from the Moderate Resolution Imaging Spectroradiometer (MODIS) Aqua Ocean Colour website (NASA OB.DAAC, Greenbelt, MD, USA). Accessed 28/11/2019. URL: http://oceancolor.gsfc.nasa.gov/cgi/l3. Cambridge (UK): UNEP World Conservation Monitoring Centre. URL: http://data.unep-wcmc.org/datasets/36
Negreiros-Fransozo, M. L., Fransozo, A., & Hebling, N. J. (1991). Estrutura populacional e determinação do tamanho da concha em 4 espécies de ermitões (Crustacea, Decapoda, Anomura) do litoral paulista. Biotemas, 4(2), 135–148.
Negri, M., Lemaitre, R., & Mantelatto, F. L. (2014). Molecular and morphological resurrection of Clibanarius symmetricus, a cryptic species hiding under the name for the “thinstripe” hermit crab C. vittatus (Decapoda: Anomura: Diogenidae). Journal of Crustacean Biology, 34(6), 848–861. https://doi.org/10.1163/1937240X-00002277
Nucci, P. R., & Melo, G. D. (2015). Hermit crabs from Brazil: Family Diogenidae (Crustacea: Decapoda: Paguroidea), except Paguristes. Zootaxa, 3947(3), 327–346. https://doi.org/10.11646/zootaxa.3947.3.2
O’Hara, R. B. (2005). The anarchist’s guide to ecological theory. Or, we don’t need no stinkin’laws. Oikos, 110(2), 390–393. https://doi.org/10.1111/j.0030-1299.2005.13959.x
Ohlberger, J. (2013). Climate warming and ectotherm body size–from individual physiology to community ecology. Functional Ecology, 27(4), 991–1001. https://doi.org/10.1111/1365-2435.12098
Olalla-Tárraga, M. Á. (2011). “Nullius in Bergmann” or the pluralistic approach to ecogeographical rules: a reply to Watt et al. (2010). Oikos, 120(10), 1441–1444. https://doi.org/10.1111/j.1600-0706.2011.19319.x
Orensanz, J. M., Ernst, B., & Armstrong, D. A. (2007). Variation of female size and stage at maturity in snow crab (Chionoecetes opilio) (Brachyura: Majidae) from the eastern Bering Sea. Journal of Crustacean Biology, 27(4), 576–591. https://doi.org/10.1651/S-2790.1
R Development Core Team. (2012). R: a Language and environment for statistical computing. R Foundation for statistical computing.
Ray, C. (1960). The application of Bergmann’s and Allen’s rules to the poikilotherms. Journal of Morphology, 106(1), 85–108. https://doi.org/10.1002/jmor.1051060104
Reese, E. S. (1963). The behavioral mechanisms underlying shell selection by hermit crabs. Behaviour, 21(1), 78–124. https://doi.org/10.1163/156853963X00121
Reese, E. S. (1969). Behavioral adaptation of intertidal hermit crabs. American Zoologist, 9(2), 343–355. https://doi.org/10.1093/icb/9.2.343
Regier, J. C., Shultz, J. W., Zwick, A., Hussey, A., Ball, B., Wetzer, R., Martin, J. W., & Cunningham, C. W. (2010). Arthropod relationships revealed by phylogenomic analysis of nuclear protein-coding sequences. Nature, 463(7284), 1079. https://doi.org/10.1038/nature08742
Rensch, B. (1950). Die abhängigkeit der relativen sexualdifferenz von der körpergrösse. Bonner Zoologische Beiträge, 1, 58–69.
Rios, E. C. (2009). Compendium of Brazilian sea shells. Evangraf.
Rodrigues, A. C. M., & Martinelli-Lemos, J. M. (2019). Spatiotemporal distribution and population structure of Clibanarius symmetricus (Randall, 1840) (Crustacea, Diogenidae) in an Amazon estuary. Turkish Journal of Zoology, 43(5), 490–501. https://doi.org/10.3906/zoo-1809-7
Sampaio, S. R., Masunari, S., & Haseyama, K. L. (2009). Distribuição temporal do ermitão Clibanarius vittatus (Anomura, Diogenidae) no litoral do Paraná. Iheringia. Série Zoologia, 99(3), 276–280. https://doi.org/10.1590/S0073-47212009000300007
Sant’Anna, B. S., Christofoletti, R. A., Zangrande, C. M., & Reigada, A. L. D. (2008). Growth of the hermit crab Clibanarius vittatus (Bosc, 1802) (Crustacea, Anomura, Diogenidae) at São Vicente, São Paulo, Brazil. Brazilian Archives of Biology and Technology, 51(3), 547–550. https://doi.org/10.1590/S1516-89132008000300014
Sant’Anna, B. S., Reigada, A. L. D., & Pinheiro, M. A. A. (2009). Population biology and reproduction of the hermit crab Clibanarius vittatus (Decapoda: Anomura) in an estuarine region of southern Brazil. Journal of the Marine Biological Association of the United Kingdom, 89(4), 761–767. https://doi.org/10.1017/S0025315409003075
Seuront, L., Ng, T. P., & Lathlean, J. A. (2018). A review of the thermal biology and ecology of molluscs, and of the use of infrared thermography in Molluscan research. Journal of Molluscan Studies, 84(3), 203–232. https://doi.org/10.1093/mollus/eyy023
Shapiro, S. S., & Wilk, M. B. (1965). An analysis of variance test for Normality (complete samples). Biometrika, 52, 591–611. https://doi.org/10.2307/2333709
Shine, R. (1988). The evolution of large body size in females: A critique of Darwin’s “fecundity advantage” model. The American Naturalist, 131(1), 124–131. https://doi.org/10.1086/284778
Shine, R. (1989). Ecological causes for the evolution of sexual dimorphism: A review of the evidence. The Quarterly Review of Biology, 64(4), 419–461. https://doi.org/10.1086/416458
Silveira, A. R., Magri, F. S., Fransozo, V., Silva, T. E., & Negreiros-Fransozo, M. L. (2017). Does shells type restraining influence relative growth rates of Clibanarius antillensis (Crustacea: Decapoda: Anomura: Diogenidae). Journal of Marine Biology & Oceanography, 6(2), 2. https://doi.org/10.4172/2324-8661.1000175
Simpson, L. A., Ambrosio, L. J., & Baeza, J. A. (2016). Sexual dimorphism and allometric growth in the enigmatic pygmy crab Petramithrax pygmaeus (Decapoda: Brachyura: Mithracidae), with a formal test of Rensch’s rule in spider crabs (Superfamily Majoidea). Journal of Crustacean Biology, 36(6), 792–803. https://doi.org/10.1163/1937240X-00002486
Smith, R. J. (1999). Statistics of sexual size dimorphism. Journal of Human Evolution, 36, 423–459. https://doi.org/10.1006/jhev.1998.0281
Sokal, R. R., & Rohlf, F. J. (1985). Biometry. W.H. Freeman and Company.
Stanski, G., Hippólito, A. G., Costa, R. C., & Castilho, A. L. (2019). Geographical variation in fecundity and reproductive investment of Loxopagurus loxochelis (Decapoda: Anomura: Diogenidae) along the south-eastern coast of Brazil. Invertebrate Reproduction & Development, 63(1), 67–75. https://doi.org/10.1080/07924259.2018.1547225
Subramoniam, T. (2016). Sexual biology and reproduction in crustaceans. Academic Press.
Takahashi, K. H. (2019). Multiple modes of canalization: links between genetic, environmental canalizations and developmental stability, and their trait-specificity. Seminars in Cell & Developmental Biology, 88, 14–20. https://doi.org/10.1016/j.semcdb.2018.05.018
Teder, T., & Tammaru, T. (2005). Sexual size dimorphism within species increases with body size in insects. Oikos, 108(2), 321–334. https://doi.org/10.1111/j.0030-1299.2005.13609.x
Thatje, S., & Hall, S. (2016). The effect of temperature on the evolution of per offspring investment in a globally distributed family of marine invertebrates (Crustacea: Decapoda: Lithodidae). Marine Biology, 163(3), 48. https://doi.org/10.1007/s00227-015-2776-8
Turra, A. (2005). Reproductive behavior of intertidal hermit crabs (Decapoda, Anomura) in southeastern Brazil. Revista Brasileira De Zoologia, 22(2), 313–319. https://doi.org/10.1590/S0101-81752005000200003
Turra, A., & Denadai, M. R. (2001). Desiccation tolerance of four sympatric tropical intertidal hermit crabs (Decapoda, Anomura). Marine & Freshwater Behaviour & Phy, 34(4), 227–238. https://doi.org/10.1080/10236240109379076
Turra, A., & Leite, F. P. P. (1999). Population structure and fecundity of the hermit crab Clibanarius antillensis Stimpson 1862 (Anomura, Diogenidae) in southeastern Brazil. Bulletin of Marine Science, 64(2), 281–289.
Turra, A., & Leite, F. P. (2000). Population biology and growth of three sympatric species of intertidal hermit crabs in south-eastern Brazil. Journal of the Marine Biological Association of the United Kingdom, 80(6), 1061–1069. https://doi.org/10.1017/S002531540000312X
Turra, A., & Leite, F. P. (2001). Shell utilization patterns of a tropical rocky intertidal hermit crab assemblage: I. The case of Grande Beach. Journal of Crustacean Biology, 21(2), 393–406. https://doi.org/10.1163/20021975-99990140
Turra, A., & Leite, F. P. P. (2003). The molding hypothesis: Linking shell use with hermit crab growth, morphology and shell-species selection. Marine Ecology Progress Series, 265, 155–163. https://doi.org/10.3354/meps265155
van de Kerk, M., Littles, C. J., Saucedo, O., & Lorenzen, K. (2016). The effect of latitudinal variation on shrimp reproductive strategies. PLoS ONE. https://doi.org/10.1371/journal.pone.0155266
Vermeij, G. J. (1971). Temperature relationships of some tropical pacific intertidal gastropods. Marine Biology, 10(4), 308–314. https://doi.org/10.1007/BF00368090
Vinarski, M. V. (2014). On the applicability of Bergmann’s rule to ectotherms: The state of the art. Biology Bulletin Reviews, 4(3), 232–242. https://doi.org/10.1134/S2079086414030098
Vogt, G. (2013). Abbreviation of larval development and extension of brood care as key features of the evolution of freshwater Decapoda. Biological Reviews, 88(1), 81–116. https://doi.org/10.1111/j.1469-185X.2012.00241.x
Wada, S. (1999). Environmental factors affecting sexual size dimorphism in the hermit crab Pagurus middendorffii. Journal of the Marine Biological Association of the United Kingdom, 79(5), 953–954. https://doi.org/10.1017/S0025315499001150
Wada, S. (2001). Reproductive characters and population structure of the subtidal hermit crab Pagurus ochotensis (Anomura: Paguridae). Benthos Research, 56(1), 43–46. https://doi.org/10.5179/benthos1996.56.1_43
Warton, D. I., Duursma, R. A., Falster, D. S., & Taskinen, S. (2012). smatr 3–an R package for estimation and inference about allometric lines. Methods in Ecology and Evolution, 3(2), 257–259. https://doi.org/10.1111/j.2041-210X.2011.00153.x
Warton, D. I., Wright, I. J., Falster, D. S., & Westoby, M. (2006). Bivariate line-fitting methods for allometry. Biological Reviews, 81, 259–291. https://doi.org/10.1017/S1464793106007007
Watt, C., Mitchell, S., & Salewski, V. (2010). Bergmann’s rule; a concept cluster? Oikos, 119(1), 89–100. https://doi.org/10.1111/j.1600-0706.2009.17959.x
Wolverton, S., Huston, M. A., Kennedy, J. H., Cagle, K., & Cornelius, J. D. (2009). Conformation to Bergmann’s rule in white-tailed deer can be explained by food availability. The American Midland Naturalist, 162(2), 403–417. https://doi.org/10.1674/0003-0031-162.2.403
Yoshino, K., Goshima, S., & Nakao, S. (2001). Sexual difference in shell use in the hermit crab Pagurus filholi (de Man) from northern Japan. Crustacean Research, 30, 55–64. https://doi.org/10.18353/crustacea.30.0_55
Yoshino, K., Goshima, S., & Nakao, S. (2002). Temporal reproductive patterns within a breeding season of the hermit crab Pagurus filholi: Effects of crab size and shell species. Marine Biology, 141(6), 1069–1075. https://doi.org/10.1007/s00227-002-0876-8
Young, A. M. (1979). Osmoregulation in three hermit crab species, Clibanarius vittatus (Bosc), Pagurus longicarpus Say and P. pollicaris Say (Crustacea: Decapoda; Anomura). Comparative Biochemistry and Physiology Part A: Physiology, 63(3), 377–382. https://doi.org/10.1016/0300-9629(79)90606-6
Zar, J. H. (2010). Biostatistical analysis. NY: Pearson Prentice-Hall.
Acknowledgements
The authors thank the anonymous referees for their comments. We would like to thank the members of the Laboratório de Fauna Aquática (UFRN), Paulo A. Lima-Filho, Elena Thomsen, Sávio A. S. N. Moraes, Isabela C. Alves, Origilene B. Dantas, and Jefferson C. M. Santos for their help in the field collecting specimens. We acknowledge the Ministério do Meio Ambiente (MMA), Instituto Chico Mendes de Conservação da Biodiversidade (ICMBio) and Sistema de Autorização e Informação em Biodiversidade (SISBIO) for license #64213-1.
Funding
The Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brazil (CAPES)—Funding Code 001, for granting scholarship to VFV (#88882.376154/2019-01). For the National Council for Scientific and Technological Development—CNPq (PQ 311521/2011-8) (PQ 309771/2014-5) for granting the research scholarship to FAMF.
Author information
Authors and Affiliations
Contributions
Conceptualization: VFV, CERDA. Methodology: VFV, CERDA. Formal analysis and research: VFV, CERDA. Laboratory support: FAMF. Writing—preparation of original draft: VFV, CERDA. Writing—proofreading and editing: VFV, CERDA, FAMF. Supervision: CERDA, FAMF. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Vale, V.F., Freire, F.A.d.M. & Alencar, C.E.R.D. Bergmann-Rensch Continuum Under Shell: Gender-Specific Trend in Response to Latitudinal Gradient. Evol Biol 51, 217–234 (2024). https://doi.org/10.1007/s11692-024-09628-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11692-024-09628-5