Abstract
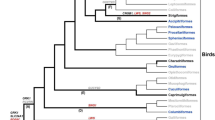
Diurnality, associated with enhanced visual acuity and color vision, is typical of most modern Primates. However, it remains a matter of debate when and how many times primates re-acquired diurnality or returned to nocturnality. We analyzed the features specific to nocturnal and diurnal vision that were recently found in the nuclei of mammalian rod photoreceptor cells in 11 species representing various groups of the Primates and related tree shrew and colugo. In particular, heterochromatin in rod nuclei of nocturnal mammals is clustered in the center of rod nuclei (inverted architecture), whereas rods of diurnal mammals retain rods with peripheral heterochromatin (conventional architecture). Rod nuclei of the nocturnal owl monkey have a state transitional to the inverted one. Surprisingly, rod nuclei of the tarsier have a conventional nuclear architecture typical for diurnal mammals, strongly implying that recent Tarsiiformes returned to nocturnality from the diurnal state. Diurnal lemurs retain inverted rod nuclei typical of nocturnal mammals, which conforms to the notion that the ancestors of all Lemuroidea were nocturnal. Data on the expression of proteins indispensable for peripheral heterochromatin maintenance (and, respectively, conventional or inverted nuclear organization) in rod cells support the view that the primate ancestors were nocturnal and transition to diurnality occurred independently in several primate and related groups: Tupaia, diurnal lemurs, and, at least partially independently, in Simiiformes (monkeys and apes) and Tarsiiformes.





Similar content being viewed by others
References
Acosta, M. L., Bumsted O’Brien, K. M., Tan, S. S., & Kalloniatis, M. (2008). Emergence of cellular markers and functional ionotropic glutamate receptors on tangentially dispersed cells in the developing mouse retina. The Journal of Comparative Neurology, 506, 506–523.
Ankel-Simons, F., & Rasmussen, D. T. (2008). Diurnality, nocturnality, and the evolution of primate visual systems. American Journal of Physical Anthropology, Suppl 47, 100–117.
Baylor, D. A., Lamb, T. D., & Yau, K. W. (1979). Responses of retinal rods to single photons. Journal of Physiology, 288, 613–634.
Chaimanee, Y., Lebrun, R., Yamee, C., & Jaeger, J. J. (2011). A new Middle Miocene tarsier from Thailand and the reconstruction of its orbital morphology using a geometric-morphometric method. Proceedings of the Royal Society B—Biological Sciences, 278, 1956–1963.
Collins, C. E., Hendrickson, A., & Kaas, J. H. (2005). Overview of the visual system of Tarsius. The Anatomical Record. Part A, Discoveries in Molecular, Cellular, and Evolutionary Biology, 287, 1013–1025.
Cone, R. A. (1972). Rotational diffusion of rhodopsin in the visual receptor membrane. Nature: New Biology, 236, 39–43.
Davies, W. I., Collin, S. P., & Hunt, D. M. (2012). Molecular ecology and adaptation of visual photopigments in craniates. Molecular Ecology, 21, 3121–3158.
Donati, G., Santini, L., Razafindramanana, J., Boitani, L., & Borgognini-Tarli, S. (2013). (Un-)expected nocturnal activity in “Diurnal” Lemur catta supports cathemerality as one of the key adaptations of the lemurid radiation. American Journal of Physical Anthropology, 150, 99–106.
Eberhart, A., Kimura, H., Leonhardt, H., Joffe, B., & Solovei, I. (2012). Reliable detection of epigenetic histone marks and nuclear proteins in tissue cryosections. Chromosome Research, 20, 849–858.
Fernandez-Duque, E., de la Iglesia, H., & Erkert, H. G. (2010). Moonstruck primates: owl monkeys (Aotus) need moonlight for nocturnal activity in their natural environment. PLoS One, 5, e12572.
Finotelo, L. F., Amaral, P. J., Pieczarka, J. C., de Oliveira, E. H., Pissinati, A., Neusser, M., et al. (2010). Chromosome phylogeny of the subfamily Pitheciinae (Platyrrhini, Primates) by classic cytogenetics and chromosome painting. BMC Evolutionary Biology, 10, 189.
Gursky, S. (2003). Lunar philia in a nocturnal primate. International Journal of Primatology, 24, 351–356.
Hall, M. I., Kamilar, J. M., & Kirk, E. C. (2012). Eye shape and the nocturnal bottleneck of mammals. Proceedings of the Royal Society B—Biological Sciences, 279, 4962–4968.
Heesy, C. P., & Hall, M. I. (2010). The nocturnal bottleneck and the evolution of mammalian vision. Brain, Behavior and Evolution, 75, 195–203.
Hendrickson, A., Djajadi, H. R., Nakamura, L., Possin, D. E., & Sajuthi, D. (2000). Nocturnal tarsier retina has both short and long/medium-wavelength cones in an unusual topography. The Journal of Comparative Neurology, 424, 718–730.
Hiwatashi, T., Okabe, Y., Tsutsui, T., Hiramatsu, C., Melin, A. D., Oota, H., et al. (2010). An explicit signature of balancing selection for color-vision variation in new world monkeys. Molecular Biology and Evolution, 27, 453–464.
Jacobs, G. H. (2008). Primate color vision: A comparative perspective. Visual Neuroscience, 25, 619–633.
Jacobs, G. H. (2013). Losses of functional opsin genes, short-wavelength cone photopigments, and color vision—A significant trend in the evolution of mammalian vision. Visual Neuroscience, 30, 39–53.
Jacobs, G. H., & Deegan, J. F., 2nd. (2005). Polymorphic New World monkeys with more than three M/L cone types. Journal of the Optical Society of America. A, Optics, Image Science, and Vision, 22, 2072–2080.
Jacobs, G. H., Neitz, M., & Neitz, J. (1996). Mutations in S-cone pigment genes and the absence of colour vision in two species of nocturnal primate. Proceedings of the Royal Society B—Biological Sciences, 263, 705–710.
Jacobs, G. H., Williams, G. A., Cahill, H., & Nathans, J. (2007). Emergence of novel color vision in mice engineered to express a human cone photopigment. Science, 315, 1723–1725.
Kay, R. F., & Kirk, E. C. (2000). Osteological evidence for the evolution of activity pattern and visual acuity in primates. American Journal of Physical Anthropology, 113, 235–262.
Knabe, W., Skatchkov, S., & Kuhn, H. J. (1997). “Lens mitochondria” in the retinal cones of the tree-shrew Tupaia belangeri. Vision Research, 37, 267–271.
Levenson, D. H., Fernandez-Duque, E., Evans, S., & Jacobs, G. H. (2007). Mutational changes in S-cone opsin genes common to both nocturnal and cathemeral Aotus monkeys. American Journal of Primatology, 69, 757–765.
Martin, R. D., & Ross, C. F. (2005). The evolutionary and ecological context of primate vision. In J. Kremers (Ed.), The primate visual system: A comparative approach (pp. 1–36). Chichester: John Wiley and Sons.
Melin, A. D., Matsushita, Y., Moritz, G. L., Dominy, N. J., & Kawamura, S. (2013). Inferred L/M cone opsin polymorphism of ancestral tarsiers sheds dim light on the origin of anthropoid primates. Proceedings of the Royal Society B—Biological Sciences, 280, 20130189.
Menezes, A. N., Bonvicino, C. R., & Seuanez, H. N. (2010). Identification, classification and evolution of owl monkeys (Aotus, Illiger 1811). BMC Evolutionary Biology, 10, 248.
Moritz, G. L., Lim, N. T.-L., Netz, M., Peichl, L., & Dominy, N. J. (2013). Expression and evolution of short wavelength sensitive opsins in colugos: A nocturnal lineage that informs debate on primate origins. Evolutionary Biology. doi:10.1007/s11692-013-9230-y.
Müller, B., Glösmann, M., Peichl, L., Knop, G. C., Hagemann, C., & Ammermüller, J. (2009). Bat eyes have ultraviolet-sensitive cone photoreceptors. PLoS One, 4, e6390.
Müller, B., Goodman, S. M., & Peichl, L. (2007). Cone photoreceptor diversity in the retinas of fruit bats (megachiroptera). Brain, Behavior and Evolution, 70, 90–104.
Nieves, M., De Oliveira, E. H., Amaral, P. J., Nagamachi, C. Y., Pieczarka, J. C., Muhlmann, M. C., et al. (2011). Analysis of the heterochromatin of Cebus (Primates, Platyrrhini) by micro-FISH and banding pattern comparisons. Journal of Genetics, 90, 111–117.
Perelman, P., Johnson, W. E., Roos, C., Seuanez, H. N., Horvath, J. E., Moreira, M. A., et al. (2011). A molecular phylogeny of living primates. PLoS Genetics, 7, e1001342.
Pieczarka, J. C., Nagamachi, C. Y., Muniz, J. A., Barros, R. M., & Mattevi, M. S. (1998). Analysis of constitutive heterochromatin of Aotus (Cebidae, Primates) by restriction enzyme and fluorochrome bands. Chromosome Research, 6, 77–83.
Prakhongcheep, O., Hirai, Y., Hara, T., Srikulnath, K., Hirai, H., & Koga, A. (2013). Two types of alpha satellite DNA in distinct chromosomal locations in Azara’s Owl Monkey. DNA Research. doi:10.1093/dnares/dst004.
Renne, P. R., Deino, A. L., Hilgen, F. J., Kuiper, K. F., Mark, D. F., Mitchell, W. S., 3rd, et al. (2013). Time scales of critical events around the Cretaceous-Paleogene boundary. Science, 339, 684–687.
Rossie, J. B., Ni, X., & Beard, K. C. (2006). Cranial remains of an Eocene tarsier. Proceedings of the National Academy of Sciences of the United States of America, 103, 4381–4385.
Solovei, I., Kreysing, M., Lanctot, C., Kösem, S., Peichl, L., Cremer, T., et al. (2009). Nuclear architecture of rod photoreceptor cells adapts to vision in mammalian evolution. Cell, 137, 356–368.
Solovei, I., Wang, A. S., Thanisch, K., Schmidt, C. S., Krebs, S., Zwerger, M., et al. (2013). LBR and lamin A/C sequentially tether peripheral heterochromatin and inversely regulate differentiation. Cell, 152, 584–598.
Talebi, M. G., Pope, T. R., Vogel, E. R., Neitz, M., & Dominy, N. J. (2006). Polymorphism of visual pigment genes in the muriqui (Primates, Atelidae). Molecular Ecology, 15, 551–558.
Tan, Y., Yoder, A. D., Yamashita, N., & Li, W. H. (2005). Evidence from opsin genes rejects nocturnality in ancestral primates. Proceedings of the National Academy of Sciences of the United States of America, 102, 14712–14716.
Tattersall, I. (2006). Origin of the Malagasy strepsirrhine primates. In L. Gould & M. L. Sauther (Eds.), Lemurs: Ecology and adaptation (pp. 3–17). New York: Springer.
Torres, O. M., Enciso, S., Ruiz, F., Silva, E., & Yunis, I. (1998). Chromosome diversity of the genus Aotus from Colombia. American Journal of Primatology, 44, 255–275.
van Oosterhout, F., Fisher, S. P., van Diepen, H. C., Watson, T. S., Houben, T., VanderLeest, H. T., et al. (2012). Ultraviolet light provides a major input to non-image-forming light detection in mice. Current Biology, 22, 1397–1402.
Warrant, E. (2004). Vision in the dimmest habitats on earth. Journal of Comparative Physiology. A, Neuroethology, Sensory, Neural, and Behavioral Physiology, 190, 765–789.
Wässle, H. (2004). Parallel processing in the mammalian retina. Nature Reviews Neuroscience, 5, 747–757.
Wildman, D. E., Jameson, N. M., Opazo, J. C., & Yi, S. V. (2009). A fully resolved genus level phylogeny of neotropical primates (Platyrrhini). Molecular Phylogenetics and Evolution, 53, 694–702.
Williams, B. A., Kay, R. F., & Kirk, E. C. (2010). New perspectives on anthropoid origins. Proceedings of the National Academy of Sciences of the United States of America, 107, 4797–4804.
Zhao, H., Rossiter, S. J., Teeling, E. C., Li, C., Cotton, J. A., & Zhang, S. (2009). The evolution of color vision in nocturnal mammals. Proceedings of the National Academy of Sciences of the United States of America, 106, 8980–8985.
Acknowledgments
We are grateful to all colleagues who supplied us with retina samples, to Stefan Müller (LMU, Munich) for a discussion of the Aotus karyotype, and to an anonymous reviewer for very helpful comments. The study was supported by the DFG (JO903/1 to BJ, SFB/TR5 to HL and SO1054/1 to IS).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Boris Joffe and Leo Peichl contributed equally.
Rights and permissions
About this article
Cite this article
Joffe, B., Peichl, L., Hendrickson, A. et al. Diurnality and Nocturnality in Primates: An Analysis from the Rod Photoreceptor Nuclei Perspective. Evol Biol 41, 1–11 (2014). https://doi.org/10.1007/s11692-013-9240-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11692-013-9240-9