Abstract
Introduction
The aim of this study was to develop a synthesis of the evidence available regarding verified E. granulosus sensu lato (s.l.) genotypes in different species worldwide.
Material and Methods
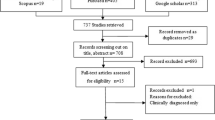
A systematic review was performed including studies concerning genotypes of E. granulosus s.l. without language or genotyped method restriction, published between 1990 and 2020. A systematic search was carried out in Trip Database, BIREME, SciELO, LILACS, IBECS, PAHO-WHO, EMBASE, PubMed, Scopus, and WoS. Variables of interest were year of publication, country, number of samples, and hosts; genotypes, molecular marker, haplotypes and molecular biology techniques used. Descriptive statistics were applied.
Results
2411 articles were analyzed, however 135 met the selection criteria, representing 8643 liver and lung samples. Of the samples selected 24% were human, the remaining samples pertained to non-human animal hosts; cattle and sheep prevailed with 28.6% and 26.6% of the studied samples, respectively. The reported evidence is mainly from Iran, Turkey, Argentina, China and Chile; with 50, 11, 6, 6 and 5 studies, respectively, published between 1992 and 2020 [most frequently during 2015–2020 (76/135 studies; 56.3%)]. The mitochondrial gene cox1 was generally sequenced and informative (91.8%). Genotypes most frequently identified were E. granulosus sensu stricto (s.s.) (83.2%).
Conclusions
Based on this overall evidence, it can be concluded that publications related to genotypes of E. granulosus s.l. are heterogeneous. E. granulosus ss accounts for the vast majority of the global burden of E. granulosus s.l. worldwide. Further studies including larger number of cases and adequate internal validity are required to specify the distribution of genotypes in various host species.
Trial registration:
PROSPERO CRD42018099827.




Similar content being viewed by others
References
Higuita NIA, Brunetti E, McCloskey C (2016) Cystic echinococcosis. J Clin Microbiol 54:518–523. https://doi.org/10.1128/JCM.02420-15
Otero-Abad B, Torgerson PR (2013) A systematic review of the epidemiology of echinococcosis in domestic and wild animals. PLoS Negl Trop Dis 7:e2249. https://doi.org/10.1371/journal.pntd.0002249
Cucher MA, Macchiaroli N, Baldi G, Camicia F, Prada L, Maldonado L et al (2016) Cystic echinococcosis in South America: systematic review of species and genotypes of Echinococcus granulosus sensu lato in humans and natural domestic hosts. Trop Med Int Heal 21:166–175. https://doi.org/10.1111/tmi.12647
Bowles J, Blair D, McManus DP (1992) Genetic variants within the genus Echinococcus identified by mitochondrial DNA sequencing. Mol Biochem Parasitol 54:165–173. https://doi.org/10.1016/0166-6851(92)90109-W
Mwambete KD, Ponce-Gordo F, Cuesta-Bandera C (2004) Genetic identification and host range of the Spanish strains of Echinococcus granulosus. Acta Trop 91:87–93. https://doi.org/10.1016/j.actatropica.2004.04.001
Manterola C, Otzen T (2016) Molecular epidemiology of cystic echinococcosis: genotypic characterization in humans and different livestock. Int J Morphol 34:1472–1481. https://doi.org/10.4067/S0717-95022016000400048
Busi M, Snábel V, Varcasia A, Garippa G, Perrone V, Liberato CD, D’Amelio SD (2007) Genetic variation within and between G1 and G3 genotypes of Echinococcus granulosus in Italy revealed by multilocus DNA sequencing. Vet Parasitol 150:75–83. https://doi.org/10.1016/j.vetpar.2007.09.003
Miambo RD, Afonso SMS, Noormahomed EV, Pondja A, Mukaratirwa S (2020) Echinococcosis in humans and animals in Southern Africa development community countries: a systematic review. Food Waterborne Parasitol 20:e00087. https://doi.org/10.1016/j.fawpar.2020.e00087
Huttner M, Romig T (2009) Echinococcus species in Africa wildlife. Parasitology 136:1089–1095. https://doi.org/10.1017/S0031182009990461
Nakao M, Yanagida T, Konyaev S, Lavikainen A, Odonokurtsev VA, Zaikov VA, Ito A (2013) Mitochondrial phylogeny of the genus Echinococcus (Cestoda: Taenidae) with emphasis on relationship among Echinococcus canadensis genotypes. Parasitology 140:1625–1636. https://doi.org/10.1017/S0031182013000565
Wassermann M, Woldeyes D, Gerbi BM, Ebi D, Zeyhle E, Mackenstedt U et al (2016) A novel zoonotic genotype related to Echinococcus granulosus sensu stricto from southern Ethiopia. Int J Parasitol 46:663–668. https://doi.org/10.1016/j.ijpara.2016.04.005
Balbinotti H, Santos GB, Badaraco J, Arend AC, Graichen DC, Haag KL, Zaha A (2012) Echinococcus ortleppi (G5) and Echinococcus granulosus sensu stricto (G1) loads in cattle from Southern Brazil. Vet Parasitol 188:255–260. https://doi.org/10.1016/j.vetpar.2012.04.007
Nungari L, Mbae C, Gikunju J, Mulinge E, Kaburu T, Zeyhle E, Magambo J (2019) Prevalence and genotyping of Echinococcus species from livestock in Kajiado County, Kenya. Biomed Res Int 2019:4798906. https://doi.org/10.1155/2019/4798906
Khademvatan S, Yousefi E, Rafiei A, Rahdar M, Saki J (2013) Molecular characterization of livestock and human isolates of Echinococcus granulosus from south-west Iran. J Helminthol 87:240–244. https://doi.org/10.1017/S0022149X12000296
Moher D, Liberati A, Prisma Group (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Ann Intern Med 151:264–269. https://doi.org/10.7326/0003-4819-151-4-200908180-00135
Shea BJ, Reeves BC, Wells G, Thuku M, Hamel C, Moran J et al (2017) AMSTAR 2: a critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ 358:j4008. https://doi.org/10.1136/bmj.j4008
Aaty HE, Abdel-Hameed DM, Alam-Eldin YH, El-Shennawy SF, Aminou HA, Makled SS, Darweesh SK (2012) Molecular genotyping of E. granulosus in animal and human isolates from Egypt. Acta Trop 121:125–128. https://doi.org/10.1016/j.actatropica.2011.10.014
Addy F, Alakonya A, Wamae N, Magambo J, Mbae C, Mulinge E et al (2012) Prevalence and diversity of cystic echinococcosis in livestock in Maasailand, Kenya. Parasitol Res 111:2289–2294. https://doi.org/10.1007/s00436-012-3082-8
Ahmed ME, Salim B, Grobusch MP, Aradaib IE (2018) First molecular characterization of Echinococcus granulosus (sensustricto) genotype 1 among cattle in Sudan. BMC Vet Res 14:36. https://doi.org/10.1186/s12917-018-1348-9
AlKitani FA, Baqir S, Mansoor MK, AlRiyami S, Hussain MH, Roberts D (2020) Genetic survey of cystic echinococcosis in farm animals in Oman. Trop Anim Health Prod 52:331–337. https://doi.org/10.1007/s11250-019-02019-5
Ali V, Martinez E, Duran P, Seláez MA, Barragan M, Nogales P, Lillo APY et al (2020) Echinococcus granulosus sensu stricto, Echinococcus ortleppi; and E. intermedius (G7) are present in Bolivia. Parasitology 147:949–956. https://doi.org/10.1017/S0031182020000529
Rojas CAA, Ebi D, Paredes R, Acosta-Jamett G, Urriola N, Roa JC et al (2017) High intraspecific variability of Echinococcus granulosus sensu stricto in Chile. Parasitol Int 66:112–115. https://doi.org/10.1016/j.parint.2016.12.001
Alvi MA, Ohiolei JA, Saqib M, Li L, Tayyab MH, Alvi AA et al (2020) Echinococcus granulosus (sensu stricto) (G1, G3) and E. ortleppi (G5) in Pakistan: phylogeny, genetic diversity and population structural analysis based on mitochondrial DNA. Parasit Vectors 13:347. https://doi.org/10.1186/s13071-020-04199-8
Andresiuk MV, Gordo FP, Saarma M, Elissondo MC, Taraborelli A, Casalongue C et al (2013) Echinococcus granulosus genotype G1 dominated in cattle and sheep during 2003–2006 in Buenos Aires, an endemic area for cystic echinococcosis in Argentina. Acta Trop 127:136. https://doi.org/10.1016/j.actatropica.2013.04.008
Arbabi M, Pirestani M, Delavari M, Hooshyar H, Abdoli A, Sarvi S (2017) Molecular and morphological characterizations of Echinococcus granulosus from human and animal isolates in Kashan, Markazi Province, Iran. Iran J Parasitol 12:177–187 (PMID: 28761477)
Armua-Fernandez MT, Castro OF, Crampet A, Bartzabal Å, Hofmann-Lehmann R, Grimm F, Deplazes P (2014) First case of peritoneal cystic echinococcosis in a domestic cat caused by Echinococcus granulosus sensu stricto (genotype 1) associated to feline immunodeficiency virus infection. Parasitol Int 63:300–302. https://doi.org/10.1016/j.parint.2013.11.005
Avila HG, Santos GB, Cucher MA, Macchiaroli N, Pérez MG, Baldi G et al (2017) Implementation of new tools in molecular epidemiology studies of Echinococcus granulosus sensu lato in South America. Parasitol Int 66:250–257. https://doi.org/10.1016/j.parint.2017.02.001
Babazadeh M, Sharifiyazdi H, Moazeni M, Gorjipour S, Heidari M (2015) Molecular characterization of a new microvariant of the G3 genotype for E. granulosus in water buffalo in Iran. Vet Res Forum Winter 6:83–87 (PMID: 25992257)
Barazesh A, Sarkari B, Sarisu G, Hami M, Mikaeili F, Aydin A et al (2019) Comparative genotyping of Echinococcus granulosus infecting livestock in Turkey and Iran. Turkiye Parazitol Derg 43:123–129. https://doi.org/10.4274/tpd.galenos.2019.6117
Bold B, Boué F, Schindler C, Badmaa B, Batbekh B, Argamjav B et al (2019) Evidence for camels (Camelus bactrianus) as the main intermediate host of Echinococcus granulosus sensu lato G6/G7 in Mongolia. Parasitol Res 118:2583–2590. https://doi.org/10.1007/s00436-019-06391-x
Bonelli P, Dei Giudici S, Peruzzu A, Piseddu T, Santucciu C, Masu G et al (2020) Genetic diversity of Echinococcus granulosus sensu stricto in Sardinia (Italy). Parasitol Int 77:102120. https://doi.org/10.1016/j.parint.2020.102120
Borji H, Bahar MM, Naghibi A, Masoom SHF (2018) Genotyping of human cystic echinococcosis in northeastern Iran. Ann Parasitol 64:323–329. https://doi.org/10.17420/ap6404.167
Boufana B, Lett WS, Lahmar S, Buishi I, Bodell AJ, Varcasia A et al (2015) Echinococcus equinus and Echinococcus granulosus sensustricto from the United Kingdom: genetic diversity and haplotypic variation. Int J Parasitol 45:161–166. https://doi.org/10.1016/j.ijpara.2014.10.005
Boufana B, Lahmar S, Rebaï W, Ben Safta Z, Jebabli L, Ammar A et al (2014) Genetic variability and haplotypes of Echinococcus isolates from Tunisia. Trans R Soc Trop Med Hyg 108:706–714. https://doi.org/10.1093/trstmh/tru138
Bowles J (1994) Molecular genetic characterization of the cervid strain of Echinococcus granulosus. Parasitology 109:215–221. https://doi.org/10.1017/S0031182000076332
Casulli A, Zeyhle E, Brunetti E, Pozio E, Meroni V, Genco F, Filice C (2010) Molecular evidence of the camel strain (G6 genotype) of E. granulosus in humans from Turkana, Kenya. Trans R Soc Trop Med Hyg 104:29–32. https://doi.org/10.1016/j.trstmh.2009.08.001
Cengiz G, Gonenc B (2020) Comparison of molecular and morphological characterization and haplotype analysis of cattle and sheep isolates of cystic echinococcosis. Vet Parasitol 282:109132. https://doi.org/10.1016/j.vetpar.2020.109132
Correa F, Stoore C, Horlacher P, Jiménez M, Hidalgo C, Rojas CAA, Barros GF et al (2018) First description of Echinococcus ortleppi and cystic echinococcosis infection status in Chile. PLoS ONE 13:e0197620. https://doi.org/10.1371/journal.pone.0197620
Cruz-Reyes A, Constantine CC, Boxell AC, Hobbs RP, Thompson RC (2007) Echinococcus granulosus from Mexican pigs is the same strain as that in Polish pigs. J Helminthol 81:287–292. https://doi.org/10.1017/S0022149X07787564
Dán Á, Rónai Z, Széll Z, Sréter T (2018) Prevalence and genetic characterization of Echinococcus spp. in cattle, sheep, and swine in Hungary. Parasitol Res 117:3019–3022. https://doi.org/10.1007/s00436-018-5977-5
Debiaggi MF, Soriano SV, Pierangeli NB, Lazzarini LE, Pianciola LA, Mazzeo ML et al (2017) (2017) Genetic characterization of human hydatid cysts shows coinfection by Echinococcus canadensis G7 and E. granulosus sensu stricto G1 in Argentina. Parasitol Res 116:2599–2604. https://doi.org/10.1007/s00436-017-5555-2
Debeljak Z, Boufana B, Interisano M, Vidanovic D, Kulisic Z, Casulli A (2016) First insights into the genetic diversity of Echinococcus granulosus sensu stricto (s.s.) in Serbia. Vet Parasitol 223:57–62. https://doi.org/10.1016/j.vetpar.2016.04.007
De la Rue ML, Takano K, Brochado JF, Costa CV, Soares AG, Yamano K et al (2011) Infection of humans and animals with Echinococcus granulosus (G1 and G3 strains) and E. ortleppi in Southern Brazil. Vet Parasitol 177:97–103. https://doi.org/10.1016/j.vetpar.2010.11.018
Dousti M, Abdi J, Bakhtiyari S, Mohebali M, Mirhendi S, Rokni M (2013) Genotyping of hydatid cyst isolated from human and domestic animals in Ilam Province, Western Iran using PCR-RFLP. Iran J Parasitol 8:47–52 (PMID: 23682259)
Erdoğan E, Özkan B, Mutlu F, Karaca S, Şahin İ (2017) Molecular characterization of E. granulosus isolates obtained from different hosts. Mikrobiyol Bul 51:79–86. https://doi.org/10.5578/mb.45452
Espinoza S, Salas AM, Vargas A, Freire V, Diaz E, Sánchez G, Venegas J (2014) Detection of the G3 genotype of Echinococcus granulosus from hydatid cysts of Chilean cattle using COX1 and ND1 mitochondrial markers. Parasitol Res 113:139–147. https://doi.org/10.1007/s00436-013-3636-4
Fadakar B, Tabatabaei N, Borji H, Naghibi A (2015) Genotyping of Echinococcus granulosus from goats and sheep indicating G7 genotype in goats in the northeast of Iran. Vet Parasitol 214:204–207. https://doi.org/10.1016/j.vetpar.2015.09.029
Fallahizadeh S, Arjmand R, Jelowdar A, Rafiei A, Kazemi F (2019) Determination of Echinococcus granulosus genotypes in livestock slaughtered in Shush County, Southwest Iran using PCR-RFLP. Helminthologia 56:196–201. https://doi.org/10.2478/helm-2019-0023
Farhadi M, Fazaeli A, Haniloo A (2015) Genetic characterization of livestock and human hydatid cyst isolates from northwest Iran, using the mitochondrial cox1 gene sequence. Parasitol Res 114:4363–4370. https://doi.org/10.1007/s00436-015-4673-y
Gholami Sh, Sosari M, Fakhar M, Sharif M, Daryani A, Hashemi M et al (2012) Molecular characterization of Echinococcus granulosus from hydatid cysts isolated from human and animals in Golestan Province, North of Iran. Iran J Parasitol 7:8–16 (PMID: 23323086)
González LM, Daniel-Mwambete K, Montero E, Rosenzvit MC, McManus DP, Gárate T, Cuesta-Bandera C (2002) Further molecular discrimination of Spanish strains of Echinococcus granulosus. Exp Parasitol 102:45–56. https://doi.org/10.1016/s0014-4894(02)00146-7
Gorgani-Firouzjaee T, Kalantrai N, Ghaffari S, Alipour J, Siadati S (2018) Genotype characterization of livestock and human cystic echinococcosis in Mazandaran Province, Iran. J Helminthol 93:255–259. https://doi.org/10.1017/S0022149X1800010X
Guarnera EA, Parra A, Kamenetzky L, García G, Gutiérrez A (2004) Cystic echinococcosis in Argentina: evolution of metacestode and clinical expression in various Echinococcus granulosus strains. Acta Trop 92:153–159. https://doi.org/10.1016/j.actatropica.2004.06.008
Guo B, Zhang Z, Zheng X, Guo Y, Guo G, Zhao L et al (2019) Prevalence and molecular characterization of Echinococcus granulosus sensu stricto in Northern Xinjiang, China. Korean J Parasitol 57:153–159. https://doi.org/10.3347/kjp.2019.57.2.153
Hajialilo E, Harandi MF, Sharbatkhori M, Mirhendi H, Rostami S (2012) Genetic characterization of Echinococcus granulosus in camels, cattle and sheep from the south-east of Iran indicates the presence of the G3 genotype. J Helminthol 86:263–270. https://doi.org/10.1017/S0022149X11000320
Hammad SJ, Cavallero S, Milardi GL, Gabrielli S, D’Amelio S, Al-Nasiri FS (2018) Molecular genotyping of Echinococcus granulosus in the North of Iraq. Vet Parasitol 249:82–87. https://doi.org/10.1016/j.vetpar.2017.11.010
Han X, Jian Y, Zhang X, Ma L, Zhu W, Cai Q et al (2019) Genetic characterization of Echinococcus isolates from various intermediate hosts in the Qinghai-Tibetan Plateau Area, China. Parasitology 146:1305–1312. https://doi.org/10.1017/S0031182019000544
Hanifian H, Diba K, Tappeh KH, Mohammadzadeh H, Mahmoudlou R (2013) Identification of Echinococcus granulosus strains in isolated hydatid cyst specimens from animals by PCR-RFLP method in West Azerbaijan-Iran. Iran J Parasitol 8:376–381 (PMID: 24454429)
Haniloo A, Farhadi M, Fazaeli A, Nourian N (2013) Genotype characterization of hydatid cysts isolated from Zanjan using PCR-RFLP technique. J Zanjan Univ Med Sci Health Services 21:57–65
Harandi MF, Hobbs RP, Adams PJ, Mobedi I, Morgan-Ryan UM, Thompson RCA (2002) Molecular and morphological characterization of Echinococcus granulosus of human and animal origin in Iran. Parasitology 125:367–373. https://doi.org/10.1017/s0031182002002172
Hasan HF, Fadhil MH, Fadhil ZH (2016) Molecular characterization of Echinococcus granulosus isolated from human and domestic animals in Kirkuk, Iraq. Anim Res Int 13:2544–2547
Hedayati Z, Daryani A, Sarvi S, Gholami S, Sharif M, Pirestani M et al (2019) Molecular genotyping of the human cystic echinococcosis in Mazandaran Province, North of Iran. Iran J Parasitol 14:151–158 (PMID: 31123480)
Hernandez F, Verdugo C, Cardenas F, Sandoval R, Morales N, Olmedo P et al (2019) Echinococcus granulosus in the endangered Patagonian huemul (Hippocamelus bisulcus). J Wildl Dis 55:694–698. https://doi.org/10.7589/2018-09-215
Hidalgo C, Stoore C, Pereira I, Paredes R, Rojas CAA (2020) Multiple haplotypes of Echinococcus granulosus sensu stricto in single naturally infected intermediate hosts. Parasitol Res 119:763–770. https://doi.org/10.1007/s00436-019-06578-2
Hodžić A, Alić A, Šupić J, Škapur V, Dusche GG (2018) Echinococcus ortleppi, the cattle strain in a crested porcupine (Hystrix cristata): a new host record. Vet Parasitol 256:32–34. https://doi.org/10.1016/j.vetpar.2018.05.004
Hüttner M, Siefert L, Mackenstedt U, Romig T (2009) A survey of Echinococcus species in wild carnivores and livestock in East Africa. Int J Parasitol 39:1269–1276. https://doi.org/10.1016/j.ijpara.2009.02.015
Jafari R, Sanei B, Baradaran A, Spotin A, Bagherpour B, Darani HY (2018) Genetic characterization of Echinococcus granulosus strains isolated from humans based on nad1 and cox1 gene analysis in Isfahan, central Iran. J Helminthol 92:696–702. https://doi.org/10.1017/S0022149X17000967
Kagendo D, Magambo J, Agola EL, Njenga SM, Zeyhle E, Mulinge E et al (2014) A survey for Echinococcus spp. of carnivores in six wildlife conservation areas in Kenya. Parasitol Int 63:604–611. https://doi.org/10.1016/j.parint.2014.04.001
Karamian M, Haghighi F, Hemmati M, Taylor WR, Salehabadi A, Ghatee MA (2017) Heterogenity of Echinococcus canadensis genotype 6—the main causative agent of cystic echinococcosis in Birjand, Eastern Iran. Vet Parasitol 245:78–85. https://doi.org/10.1016/j.vetpar.2017.08.018
Kesik HK, Kilinc SG, Simsek S, Gul A (2019) Occurrence of liver hydatid cysts in a donkey and molecular characterization of Echinococcus equinus. J Parasitol 105:442–445 (PMID: 31192761)
Khan A, Ahmed H, Simsek S, Liu H, Yin J, Wang Y et al (2020) Molecular characterization of human Echinococcus isolates and the first report of E. canadensis (G6/G7) and E. multilocularis from the Punjab Province of Pakistan using sequence analysis. BMC Infect Dis 20:262. https://doi.org/10.1186/s12879-020-04989-6
Kheirandish F, Badparva E, Mahmoudvand H, Beiranvand E, Babaei S, Nasiri B (2018) Genetic characterization of hydatid cysts isolated from domestic animals in Lorestan Province, Western Iran. Iran J Parasitol 13:120–126 (PMID: 29963094)
Kia EB, Rahimi H, Sharbatkhori M, Talebi A, Harandi MF, Mirhendi H (2010) Genotype identification of human cystic echinococcosis in Isfahan, central Iran. Parasitol Res 107:757–760. https://doi.org/10.1007/s00436-010-1947-2
Kinkar L, Laurimäe T, Sharbatkhori M, Mirhendi H, Kia EB, Ponce-Gordo F, Andresiuk V (2017) New mitogenome and nuclear evidence on the phylogeny and taxonomy of the highly zoonotic tapeworm Echinococcus granulosus sensu stricto. Infect Genet Evol 52:52–58. https://doi.org/10.1016/j.meegid.2017.04.023
Konyaev SV, Yanagida T, Nakao M, Ingovatova GM, Shoykhet YN, Bondarev AY et al (2013) Genetic diversity of Echinococcus spp. in Russia. Parasitology 140:1637–1647. https://doi.org/10.1017/S0031182013001340
Konyaev SV, Yanagida T, Ivanov MV, Ruppel VV, Sako Y, Nakao M, Ito A (2012) The first report on cystic echinococcosis in a cat caused by Echinococcus granulosus sensu stricto (G1). J Helminthol 86:391–394. https://doi.org/10.1017/S0022149X1100054X
Laatamna AE, Ebi D, Brahimi K, Bediaf K, Wassermann M, Souttou K, Romig T (2019) Frequency and genetic diversity of Echinococcus granulosus sensu stricto in sheep and cattle from the steppe region of Djelfa. Algeria Parasitol Res 118:89–96. https://doi.org/10.1007/s00436-018-6118-x
Laurimäe T, Kinkar L, Romig T, Umhang G, Casulli A, Omer RA et al (2019) Analysis of nad2 and nad5 enables reliable identification of genotypes G6 and G7 within the species complex Echinococcus granulosus sensu lato. Infect Genet Evol 74:103941. https://doi.org/10.1016/j.meegid.2019.103941
Laurimäe T, Kinkar L, Moks E, Romig T, Omer RA, Casulli A et al (2018) Molecular phylogeny based on six nuclear genes suggests that Echinococcus granulosus sensu lato genotypes G6/G7 and G8/G10 can be regarded as two distinct species. Parasitology 145:1929–1937. https://doi.org/10.1017/S0031182018000719
Lavikainen A, Lehtinen MJ, Meri T, Hirvelä-Koski V, Meri S (2003) Molecular genetic characterization of the Fennoscandian cervid strain, a new genotypic group (G10) of Echinococcus granulosus. Parasitology 127:207–215. https://doi.org/10.1017/s0031182003003780
Oskouei MM, Mehrabani NG, Miahipour A, Fallah E (2016) Molecular characterization and sequence analysis of Echinococcus granulosus from sheep isolates in East Azerbaijan province, northwest of Iran. J Parasit Dis 40:785–790. https://doi.org/10.1007/s12639-014-0579-3
Manterola C, Benavente F, Melo A, Vial M, Roa JC (2008) Description of Echinococcus granulosus genotypes in human hydatidosis in a region of southern Chile. Parasitol Int 57:342–346. https://doi.org/10.1016/j.parint.2008.02.005
Maravilla P, Thompson RBA, Palacios-Ruiz JA, Estcourt A, Ramirez-Solisa E, de la Peña CM et al (2004) Echinococcus granulosus cattle strain identification in an autochthonous case of cystic echinococcosis in central Mexico. Acta Trop 92:231–236. https://doi.org/10.1016/j.actatropica.2004.07.001
Marinova I, Spiliotis M, Wang J, Muhtarov M, Chaligiannis I, Sotiraki S et al (2017) Molecular characterization of Echinococcus granulosus isolates from Bulgarian human cystic echinococcosis patients. Parasitol Res 116:1043–1054. https://doi.org/10.1007/s00436-017-5386-1
Matini M, Roostaei M, Fallah M, Maghsood AH, Saidijam M, Harandi MF (2018) Genetic Identification of Echinococcus granulosus isolates in Hamadan, Western Iran. Iran J Parasitol 13:423–429 (PMID: 30483334)
Matini M, Fallah M, Maghsood AH, Saidijam M, Harandi MF (2019) Echinococcus granulosus sensu stricto in livestock and human in Hamadan, Western Iran. Iran J Parasitol 14:288–296 (PMID: 31543917)
Maurelli MP, Rinaldi L, Capuano F, Perugini AG, Cringoli G (2009) Development of a real-time PCR for the differentiation of the G1 and G2/G3 genotypes of Echinococcus granulosus. Parasitol Res 105:255–259. https://doi.org/10.1007/s00436-009-1388-y
Mehmood S, Simsek S, Celik F, Kesik HK, Kilinc SG, Ahmed H (2020) Molecular survey on cattle and sheep hydatidosis and first detection of Echinococcus canadensis (G6/G7) in sheep in Turkey. Parasitology 147:1055–1062. https://doi.org/10.1017/S0031182020000785
Metwally DM, Qassim LE, Al-Turaiki IM, Almeer RS, El-Khadragy MF (2018) Gene-based molecular analysis of COX1 in Echinococcus granulosus cysts isolated from naturally infected livestock in Riyadh, Saudi Arabia. PLoS ONE 13:e0195016. https://doi.org/10.1371/journal.pone.0195016
Mirahmadi H, Behravan M, Raz A, Tasa D, Namaei MH, Solgi R (2021) Genotyping of the Echinococcus granulosus in paraffin-embedded human tissue samples from Iran. Acta Parasitol 66:535–542. https://doi.org/10.1007/s11686-020-00309-9
Mohaghegh MA, Yousofi-Darani H, Jafarian AH, Mirbadie SR, Fasihi-Harandi M, Ghavimi R et al (2019) Isolated human and livestock Echinococcus granulosus genotypes using real-time PCR of cox1 gene in Northeast Iran. Acta Parasitol 64:679–685. https://doi.org/10.2478/s11686-019-00117-w
Moro PL, Nakao M, Ito A, Schantz PM, Cavero C, Cabrera L (2009) Molecular identification of Echinococcus isolates from Peru. Parasitol Int 58:184–186. https://doi.org/10.1016/j.parint.2009.01.005
Moudgil AD, Moudgil P, Asrani RK, Agnihotri RK (2019) Hydatidosis in slaughtered sheep and goats in India: prevalence, genotypic characterization and pathological studies. J Helminthol 2019:1–5. https://doi.org/10.1017/S0022149X18001219
Muqaddas H, Mehmood N, Arshad M (2020) Genetic variability and diversity of Echinococcus granulosus sensu lato in human isolates of Pakistan based on cox1 mt-DNA sequences (366bp). Acta Trop 207:105470. https://doi.org/10.1016/j.actatropica.2020.105470
M’rad S, Filisetti D, Oudni M, Mekki M, Belguith M, Nouri A et al (2005) Molecular evidence of ovine (G1) and camel (G6) strains of E. granulosus in Tunisia and putative role of cattle in human contamination. Vet Parasitol 129:267–272. https://doi.org/10.1016/j.vetpar.2005.02.006
Nakao M, Li T, Han X, Ma X, Xiao N, Qiu J et al (2010) Genetic polymorphisms of Echinococcus tapeworms in China as determined by mitochondrial and nuclear DNA sequences. Int J Parasitol 40:379–385. https://doi.org/10.1016/j.ijpara.2009.09.006
Nikmanesh B, Mirhendi H, Mahmoudi S, Rokni MB (2017) Multilocus sequence analysis of Echinococcus granulosus strains isolated from humans and animals in Iran. Exp Parasitol 183:50–55. https://doi.org/10.1016/j.exppara.2017.10.002
Nikmanesh B, Mirhendi H, Ghalavand Z, Alebouyeh M, Sharbatkhori M, Kia E et al (2014) Genotyping of Echinococcus granulosus isolates from human clinical samples based on sequencing of mitochondrial genes in Iran, Tehran. Iran J Parasitol 9:20–27 (PMID: 25642256)
Odongo DO, Tiampati CM, Mulinge E, Mbae CK, Bishop RP, Zeyhle E et al (2018) Prevalence and genotyping of Echinococcus granulosus in sheep in Narok County, Kenya. Parasitol Res 117:2065–2073. https://doi.org/10.1007/s00436-018-5889-4
Oguz B, Ozdal N, Kilinc OO, Deger MS (2018) Preliminary studies on the prevalence and genotyping of Echinococcus granulosus infection in stray dogs in Van Province, Turkey. J Vet Res 62:497–502. https://doi.org/10.2478/jvetres-2018-0061
Ohiolei JA, Li L, Yan HB, Fu BQ, Jia WZ (2020) Complete mitochondrial genome analysis confirms the presence of Echinococcus granulosus sensu lato genotype G6 in Nigeria. Infect Genet Evol 84:104377. https://doi.org/10.1016/j.meegid.2020.104377
Omondi HA, Gitau G, Gathura P, Mulinge E, Zeyhle E, Kimeli P, Bett B (2020) Prevalence and genotyping of Echinococcus granulosus sensu lato from livestock in north-eastern Kenya. J Helminthol 94:e205. https://doi.org/10.1017/S0022149X20000899
Babaoğlu AO, Soyder A, Erdoğan M, Ertuğ S, Ertabaklar H (2018) Genotyping of Echinococcus granulosus isolates by sequencing of mitochondrial cytochrome C oxidase subunit 1 (cox1) gene 601 in Aydin. Mikrobiyol Bul 52:198–205. https://doi.org/10.5578/mb.66711
Orsten S, Boufana B, Ciftci T, Akinci D, Karaagaoglu E, Ozkuyumcu C et al (2018) Human cystic echinococcosis in Turkey: a preliminary study on DNA polymorphisms of hydatid cysts removed from confirmed patients. Parasitol Res 117:1257–1263. https://doi.org/10.5578/mb.66711
Parsa F, Harandi MF, Rostami S, Sharbatkhori M (2012) Genotyping Echinococcus granulosus from dogs from Western Iran. Exp Parasitol 132:308–312. https://doi.org/10.1016/j.exppara.2012.07.010
Pestechian N, Safa AH, Tajedini M, Rostami-Nejad M, Mousavi M, Yousofi H, Javanmard SH (2014) Genetic diversity of Echinococcus granulosus in center of Iran. Korean J Parasitol 52:413. https://doi.org/10.3347/kjp.2014.52.4.413
Pezeshki A, Akhlaghi L, Sharbatkhori M, Razmjou E, Oormazdi H, Mohebali M, Meamar AR (2013) Genotyping of Echinococcus granulosus from domestic animals and humans from Ardabil Province, northwest Iran. J Helminthol 87:387–391. https://doi.org/10.1017/S0022149X1200051X
Piccoli L, Bazzocchi C, Brunetti E, Mihailescu P, Bandi C, Mastalier B et al (2013) Molecular characterization of Echinococcus granulosus in south-eastern Romania: evidence of G1–G3 and G6–G10 complexes in humans. Clin Microbiol Infect 19:578–582. https://doi.org/10.1111/j.1469-0691.2012.03993.x
Pour AA, Hosseini SH, Shayan P (2011) Comparative genotyping of Echinococcus granulosus infecting buffalo in Iran using cox1 gene. Parasitol Res 108:1229–1234. https://doi.org/10.1007/s00436-010-2170-x
Rajabloo M, Hosseini SH, Jalousian F (2012) Morphological and molecular characterisation of Echinococcus granulosus from goat isolates in Iran. Acta Trop 123:67–71. https://doi.org/10.1016/j.actatropica.2012.03.006
Rodriguez-Prado U, Jimenez-Gonzalez DE, Avila G, Gonzalez AE, Martinez-Flores WA, de la Peña CM et al (2014) Short report: genetic variation of Echinococcus canadensis (G7) in Mexico. Am J Trop Med Hyg 91:1149–1153. https://doi.org/10.4269/ajtmh.14-0317
Roinioti E, Papathanassopoulou A, Theodoropoulou I, Simsek S, Theodoropoulos G (2016) Molecular identification of Echinococcus granulosus isolates from ruminants in Greece. Vet Parasitol 226:138–144. https://doi.org/10.1016/j.vetpar.2016.06.040
Rosenzvit M, Zhang L-H, Kamenetzky L, Canova S, Guarnera E, McManus D (1999) Genetic variation and epidemiology of Echinococcus granulosus in Argentina. Parasitology 118:523–530. https://doi.org/10.1017/s0031182099004035
Rostami S, Torbaghan SS, Dabiri S, Babaei Z, Mohammadi MA, Sharbatkhori M, Harandi MF (2015) Genetic characterization of Echinococcus granulosus from a large number of formalin fixed, paraffin-embedded tissue samples of human isolates in Iran. Am J Trop Med Hyg 92:588–594. https://doi.org/10.4269/ajtmh.14-0585
Nejad MR, Taghipour N, Nochi Z, Mojarad EN, Mohebbi SR, Harandi MF, Zali MR (2012) Molecular identification of animal isolates of Echinococcus granulosus from Iran using four mitochondrial genes. J Helminthol 86:485–492. https://doi.org/10.1017/S0022149X1100071X
Sadjjadi SM, Mikaeili F, Karamian M, Maraghi S, Sadjjadi FS, Shariat-Torbaghan S et al (2013) Evidence that the Echinococcus granulosus G6 genotype has an affinity for the brain in humans. Int J Parasitol 43:875–877. https://doi.org/10.1016/j.ijpara.2013.06.008
Salamatin R, Kowal J, Nosal P, Kornaá S, Cielecka D, Jańczak D et al (2017) Cystic echinococcosis in Poland: genetic variability and the first record of Echinococcus granulosus (G1 genotype) in the country. Parasitol Res 116:3077–3085. https://doi.org/10.1007/s00436-017-5618-4
Sánchez E, Cáceres O, Náquira C, Miranda E, Samudio F, Fernandes O (2012) Echinococcus granulosus genotypes circulating in alpacas (Lama pacos) and pigs (Sus scrofa) from an endemic region in Peru. Mem Inst Oswaldo Cruz 107:275–278. https://doi.org/10.1590/s0074-02762012000200019
Sánchez E, Cáceres O, Náquira C, Garcia D, Patiño G, Silvia H et al (2010) Molecular characterization of Echinococcus granulosus from Peru by sequencing of the mitochondrial cytochrome C oxidase subunit 1 gene. Mem Inst Oswaldo Cruz 105:806–810. https://doi.org/10.1590/s0074-02762010000600013
Santivañez SJ, Gutierrez AM, Rosenzvit MC, Muzulin PM, Rodriguez ML, Vasquez JC et al (2008) Human hydatid disease in Peru is basically restricted to E. granulosus genotype G1. Am J Trop Med Hyg 79:89–92. https://doi.org/10.4269/ajtmh.2008.79.89
Scott JC, Stefaniak J, Pawlowski ZS, McManus DP (1997) Molecular genetic analysis of human cystic hydatid cases from Poland: identification of a new genotypic group (G9) of Echinococcus granulosus. Parasitology 114:37–43. https://doi.org/10.1017/s0031182096008062
Sgroi G, Varcasia A, Dessi G, D’Alessio N, Tamponi C, Saarma U et al (2019) Cystic echinococcosis in wild boars (Sus scrofa) from southern Italy: epidemiological survey and molecular characterization. Int J Parasitol Parasites Wildl 9:305–311. https://doi.org/10.1016/j.ijppaw.2019.04.013
Shahnazi M, Hejazi H, Salehi M, Andalib AR (2011) Molecular characterization of human and animal Echinococcus granulosus isolates in Isfahan, Iran. Acta Trop 117:47–50. https://doi.org/10.1016/j.actatropica.2010.09.002
Shi Y, Wan X, Wang Z, Li J, Jiang Z, Yang Y (2019) First description of Echinococcus ortleppi infection in China. Parasit Vectors 12:398. https://doi.org/10.1186/s13071-019-3653-y
Shamsi M, Dalimi A, Khosravi A, Ghafarifar F (2015) Determination of genotype isolates of human and sheep hydatid cyst in Ilam. J Ilam Univ Med Sci 23:111–119 (PMID: 23682259)
Shang J, Zhang G, Yu W, He W, Wang Q, Zhong B et al (2019) Molecular characterization of human echinococcosis in Sichuan, Western China. Acta Trop 190:45–51. https://doi.org/10.1016/j.actatropica.2018.09.019
Sharbatkhori M, Tanzifi A, Rostami S, Rostami M, Harandi MF (2016) Echinococcus granulosus sensulato genotypes in domestic livestock and humans in Golestan Province, Iran. Rev Inst Med Trop Sao Paulo 58:38. https://doi.org/10.1590/S1678-9946201658038
Shariatzadeh SA, Spotin A, Gholami S, Fallah E, Hazratian T, Mahami-Oskouei M et al (2015) The first morphometric and phylogenetic perspective on molecular epidemiology of Echinococcus granulosus sensulato in stray dogs in a hyperendemic Middle East focus, northwestern Iran. Parasit Vectors 8:409. https://doi.org/10.1186/s13071-015-1025-9
Sharifiyazdi H, Oryan A, Ahmadnia S, Valinezhad A (2011) Genotypic characterization of Iranian camel (Camelus dromedarius) isolates of Echinoccocus granulosus. J Parasitol 97:251–255. https://doi.org/10.1645/GE-2642.1
Snábel V, D’Amelio S, Mathiopoulos K, Turceková L, Dubinský P (2000) Molecular evidence for the presence of a G7 genotype of E. granulosus in Slovakia. J Helminthol 74:177–181. https://doi.org/10.1017/S0022149X00000251
Šoba B, Gašperšič Š, Keše D, Kotar T (2020) Molecular characterization of Echinococcus granulosus sensu lato from humans in Slovenia. Pathogens 9:562. https://doi.org/10.3390/pathogens9070562
Soriano SV, Pierangeli NB, Pianciola L, Mazzeo M, Lazzarini LE, Saiz MS et al (2010) Molecular characterization of Echinococcus isolates indicates goats as reservoir for Echinococcus canadensis G6 genotype in Neuquen, Patagonia Argentina. Parasitol Int 59:626–628. https://doi.org/10.1016/j.parint.2010.07.003
Spotin A, Mahami-Oskouei M, Harandi MF, Baratchian M, Bordbar A, Ahmadpour E, Ebrahimi S (2017) Genetic variability of E. granulosus complex in various geographical populations of Iran inferred by mitochondrial DNA sequences. Acta Trop 165:10–16. https://doi.org/10.1016/j.actatropica.2016.03.002
Tashani O, Zhang LH, Boufana B, Jegi A, McManus DP (2002) Epidemiology and strain characteristics of Echinococcus granulosus in the Benghazi area of eastern Libya. Ann Trop Med Parasitol 96:369–381. https://doi.org/10.1179/000349802125000952
Terefe Y, Addy F, Alemu S, Mackenstedt U, Romig T, Wassermann M (2019) Genetic characterization of Echinococcus species in eastern Ethiopia. Vet Parasitol Reg Stud Reports 17:100302. https://doi.org/10.1016/j.vprsr.2019.100302
Tigre W, Deresa B, Haile A, Gabriël S, Victor B, Pelt JV et al (2016) Molecular characterization of Echinococcus granulosus s.l. cysts from cattle, camels, goats and pigs in Ethiopia. Vet Parasitol 215:17–21. https://doi.org/10.1016/j.vetpar.2015.10.022
Umhang G, Chihai O, Boué F (2014) Molecular characterization of Echinococcus granulosus in a hyperendemic European focus, the Republic of Moldova. Parasitol Res 113:4371–4376. https://doi.org/10.1007/s00436-014-4112-5
Umhang G, Richomme C, Boucher JM, Hormaz V, Boué F (2013) Prevalence survey and first molecular characterization of Echinococcus granulosus in France. Parasitol Res 112:1809–1812. https://doi.org/10.1007/s00436-012-3245-7
Vahedi A, Mahdavi M, Ghazanchaei A, Shokouhi B (2014) Genotypic characteristics of hydatid cysts isolated from humans in East Azerbaijan Province (2011–2013). J Anal Res Clin Med 2:152–157. https://doi.org/10.5681/jarcm.2014.025
Villalobos N, González LM, Morales J, de Aluja AS, Jiménez MI, Blanco MA et al (2007) Molecular identification of Echinococcus granulosus genotypes (G1 and G7) isolated from pigs in Mexico. Vet Parasitol 147:185–189. https://doi.org/10.1016/j.vetpar.2007.03.021
Yan B, Liu X, Wu J, Zhao S, Yuan W, Wang B et al (2018) Genetic diversity of Echinococcus granulosus genotype G1 in Xinjiang, Northwest of China. Korean J Parasitol 56:391–396. https://doi.org/10.3347/kjp.2018.56.4.391
Youssefi MR, Tabaripour R, Omrani VF, Spotin A, Esfandiari B (2013) Genotypic characterization of Echinococcus granulosus in Iranian goats. Asian Pac J Trop Dis 3:362–366. https://doi.org/10.1016/S2222-1808(13)60085-7
Zait H, Kouidri M, Grenouillet FE, Umhang G, Millon L, Hamrioui B, Grenouillet F (2016) Molecular characterization of Echinococcus granulosus sensu stricto and Echinococcus canadensis in humans and livestock from Algeria. Parasitol Res 115:2423–2431. https://doi.org/10.1007/s00436-016-4994-5
Zhang LH, Chai JJ, Jiao W, Osman Y, McManus DP (1998) Mitochondrial genetic markers confirm the presence of the camel strain (G6 genotype) of Echinococcus granulosus in north-western China. Parasitology 116:29–33. https://doi.org/10.1017/s0031182097001881
Zhang L, Eslami A, Hosseini SH, McManus DP (1998) Indication of the presence of two distinct strains of Echinococcus granulosus in Iran by mitochondrial DNA markers. Am J Trop Med Hyg 59:171–174. https://doi.org/10.4269/ajtmh.1998.59.171
Siyadatpanah A, Anvari D, Emami Zeydi A, Hosseini SA, Daryani A, Sarvi S et al (2019) A systematic review and meta-analysis of the genetic characterization of human echinococcosis in Iran, an endemic country. Epidemiol Health 41:e2019024. https://doi.org/10.4178/epih.e2019024
Khademvatan S, Majidiani H, Foroutan M, Tappeh KH, Aryamand S, Khalkhali HR (2019) Echinococcus granulosus genotypes in Iran: a systematic review. J Helminthol 93:131–138. https://doi.org/10.1017/S0022149X18000275
Manterola C, Rojas C, Totomoch-Serra A, García-Méndez N, Riffo-Campos A (2020) Echinococcus granulosus genotypes verified in human hydatid disease around the world. Systematic review. Rev Chilena Infectol 37:541–549. https://doi.org/10.4067/S0716-10182020000500541
Amarasinghe SL, Su S, Dong X, Zapppia L, Ritchie ME, Gouil Q (2020) Opportunities and challenges in long-read sequencing data analysis. Genome Biol 21:30. https://doi.org/10.1186/s13059-020-1935-5
Umhang G, Grenouillet F, Bastid V, M’Rad S, Valot B, Oudni-M’Rad M et al (2018) Investigating the genetic diversity of Echinococcus granulosus sensu stricto with new microsatellites. Parasitol Res 117:2743–2755. https://doi.org/10.1007/s00436-018-5963-y
M’Rad S, Oudni-M’Rad M, Bastid V, Bournez L, Mosbahi S, Nouri A et al (2020) Microsatellite investigations of multiple Echinococcus granulosus sensu stricto cysts in single hosts reveal different patterns of infection events between livestock and humans. Pathogens 9:444. https://doi.org/10.3390/pathogens9060444
Jenkins DJ, Morris B (2003) Echinococcus granulosus in wildlife in and around Kouscjuszko National Park, south-eastern Australia. Aust Vet J 81:81–85. https://doi.org/10.1111/j.1751-0813.2003.tb11440.x
Totomoch-Serra A, Manterola C (2020) Importance of genotyping cystic echinococcosis in humans from Mexico. Arch Med Res 51:63–64. https://doi.org/10.1016/j.arcmed.2019.12.009
Totomoch-Serra A, García-Méndez N, Manterola C (2020) Echinococcus granulosus haplotypes in the scientific literature. Rev Invest Clin 72:394. https://doi.org/10.4875/RIC.20000194
Bowles J, McManus DP (1993) NADH dehydrogenase 1 gene sequences compared for species and strains of the genus Echinococcus. Int J Parasitol 23:969–972. https://doi.org/10.1016/0020-7519(93)90065-7
Funding
This work was supported by the Universidad de La Frontera DIUFRO under Grant DI19-0030.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Manterola, C., Totomoch-Serra, A., Rojas, C. et al. Echinococcus granulosus sensu lato Genotypes in Different Hosts Worldwide: A Systematic Review. Acta Parasit. 67, 161–185 (2022). https://doi.org/10.1007/s11686-021-00439-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11686-021-00439-8