Abstract
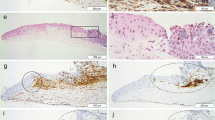
In our previous studies, grey satellite foci were found in the front of heads of pterygia. This research was designed to investigate the ultramicrostructure and clinical implications of these satellite foci. The satellite foci were observed and counted under slit lamp biomicroscope. The patients with eye pterygia were divided into groups in terms of occupation, sex, age, length of history, grade of congestion, and size of the heads. The SPSS 13.0 software was used for statistical analysis. The cap areas and satellite foci were ultramicrostructurally examined. Among the total 62 eyes with pterygium, satellite foci were found in 34. The overall incidence of satellite foci was 54.8%. There were no significant differences in incidence among the subjects of different sex, age, and length of history. There were significant differences in incidence among the patients of different occupation, grades of congestion, and size of heads. Higher grades of congestion, outdoor occupations and larger pterygium heads were associated with higher incidence of satellite foci. High grades of congestion and bigger heads were also correlated with the number of satellite foci. Length of history bore no correlation with number of satellite foci. Histologically, the components of the cap areas and the foci were identical, with both consisting of mass of active fibroblasts. The activated fibroblasts existed in the natural tissue planes between Bowman’s layer and basal cell layer. The fibroblasts in the satellite foci and the cap areas of a pterygium show some features of tumor cells and may play a vital role in the development and progression of a pterygium. The presence and amount of satellite foci around a pterygium can be used as an indicator for the speed of its growth.
Similar content being viewed by others
References
Cameron M E. Histology of pterygium: An electron microscopic study. Br J Ophthalmol, 1983, 67(9): 604–608
Yan Q C, Wang X L, Bai Q H, Wang W, Gao Q, Zhang J S, Liu Y, Liu R. Relationship between the morbidity of pterygium and the duration of ultraviolet rays exposure in Sanya, China. Chin Med J (Engl), 2006, 119(15): 1308–1310
Taylor H R, West S K, Rosenthal F S, Munoz B, Newland H S, Emmett E A. Corneal changes associated with chronic UV irradiation. Arch ophthalmolol, 1989, 107(10): 1481–1484
Karai I, Horiguchi S. Pterygium in welders. Br J Ophthalmol, 1984, 68(5): 347–349
Moran D J, Hollows F C. Pterygium and ultraviolet radiation: A positive correlation. Br j Ophthalmol, 1984, 68(5): 343–346
Frucht-Pery J, Raiskup F, Ilsar M, Landau D, Orucov F, Solomon A. Conjunctival autografting combined with low-dose mitomycin C for prevention of primary pterygium recurrence. Am J Ophthalmol, 2006, 141(6): 1044–1050
Levy R L, Naidu S, Jacobson L. Safety and efficacy of the technique of complete tenon’s membrane excision and mitomycin C in pterygium surgery. Eye Contact Lens, 2005, 31(3): 105–108
Isohashi F, Inoue T, Xing S, Eren C B, Ozeki S, Inoue T. Postoperative irradiation for pterygium: Retrospective analysis of 1,253 patients from the Osaka University Hospital. Strahlenther Onkol, 2006, 182(8): 437–442
Dushku N, Reid T W. P53 expression in altered limbal basal cells of pingueculae, pterygia, and limbal tumors. Curr Eye Res, 1997, 16(12): 1179–1192
Dushku N, Reid T W. Immunohistochemical evidence that human pterygia originate from an invasion of vimentin-expressing altered limbal epithelial basal cells. Curr Eye Res, 1994, 13(7): 473–481
Spandidos D A, Sourvinos G, Kiaris H, Tsamparlakis J. Micro-satellite instability and loss of heterozygosity in human pterygia. Br J Ophthalmol, 1997, 81(6): 493–496
Perra M T, Maxia C, Corbu A, Minerba L, Demurtas P, Colombari R, Murtas D, Bravo s, Piras F, Sirigu P. Oxidative stress in pterygium: Relationship between p53 and 8-hydroxydeoxyguanosine. Mol Vis, 2006, 12: 1136–1142
Bendesky A, Michel A, Sordo M, Calderon-Aranda E S, Acosta-Saavedra L C, Salazar AM, Podoswa N, Ostrosky-Wegman P. DNA damage, oxidative mutagen sensitivity, and repair of oxidative DNA damage in nonmelanoma skin cancer patients. Environ Mol Mutagen, 2006, 47(7): 509–517
Nakabeppu Y, Sakumi K, Sakamoto K, Tsuchimoto D, Tsuzuki T, Nakatsu Y. Mutagenesis and carcinogenesis caused by the oxidation of nucleic acids. Biol Chem, 2006, 387(4): 373–379
Nishigori C. Cellular aspects of photocarcinogenesis. Photochem Photobiol Sci, 2006, 5(2): 208–214
Chen J K, Tsai R J, Lin S S. Fibroblasts isolated from human pterygia exhibit transformed cell characteristics. In Vitro Cell Dev Biol, 1994, 30A(4): 243–248
Solomon A, Grueterich M, Li D Q, Meller D, Lee S B, Tseng S C. Over expression of insulin-like growth factor-binding protein-2 in pterygium body fibroblasts. Invest Ophthalmol Vis Sci, 2003, 44(2): 573–580
Solomon A S. Pterygium. Br J Ophthalmol, 2006, 90(6):665–666
Yu H, Rohan T. Role of the insulin-like growth factor family in cancer development and progression. J Natl Cancer Inst, 2000, 92(18): 1472–1489
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, H., Xiang, N., Zhou, X. et al. Ultramicrostructure and clinical implications of satellite foci in front of the head of pterygium. Front. Med. China 1, 211–214 (2007). https://doi.org/10.1007/s11684-007-0040-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11684-007-0040-4