Abstract
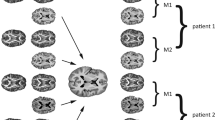
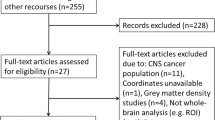
Brain gray matter (GM) reductions have been reported after breast cancer chemotherapy, typically in small and/or cross-sectional cohorts, most commonly using voxel-based morphometry (VBM). There has been little examination of approaches such as deformation-based morphometry (DBM), machine-learning-based brain aging metrics, or the relationship of clinical and demographic risk factors to GM reduction. This international data pooling study begins to address these questions. Participants included breast cancer patients treated with (CT+, n = 183) and without (CT-, n = 155) chemotherapy and noncancer controls (NC, n = 145), scanned pre- and post-chemotherapy or comparable intervals. VBM and DBM examined GM volume. Estimated brain aging was compared to chronological aging. Correlation analyses examined associations between VBM, DBM, and brain age, and between neuroimaging outcomes, baseline age, and time since chemotherapy completion. CT+ showed longitudinal GM volume reductions, primarily in frontal regions, with a broader spatial extent on DBM than VBM. CT- showed smaller clusters of GM reduction using both methods. Predicted brain aging was significantly greater in CT+ than NC, and older baseline age correlated with greater brain aging. Time since chemotherapy negatively correlated with brain aging and annual GM loss. This large-scale data pooling analysis confirmed findings of frontal lobe GM reduction after breast cancer chemotherapy. Milder changes were evident in patients not receiving chemotherapy. CT+ also demonstrated premature brain aging relative to NC, particularly at older age, but showed evidence for at least partial GM recovery over time. When validated in future studies, such knowledge could assist in weighing the risks and benefits of treatment strategies.




Similar content being viewed by others
Data availability
Inter-institutional terms of access agreements enacted for the purposes of this data pooling study do not permit resharing of the study data. Those interested on obtaining data could contact the individual participating institutions, and the corresponding author can facilitate reasonable such requests.
References
Ahles, T. A., & Root, J. C. (2018). Cognitive effects of cancer and cancer treatments. Annu Rev Clin Psychol,14, 425–451. https://doi.org/10.1146/annurev-clinpsy-050817-084903
Ahles, T. A., Root, J. C., & Ryan, E. L. (2012). Cancer- and cancer treatment-associated cognitive change: An update on the state of the science. Journal Of Clinical Oncology,30(30), 3675–3686. https://doi.org/10.1200/jco.2012.43.0116
Ahles, T. A., Saykin, A. J., McDonald, B. C., Li, Y., Furstenberg, C. T., Hanscom, B. S., Mulrooney, T. J., Schwartz, G. N., & Kaufman, P. A. (2010). Longitudinal assessment of cognitive changes associated with adjuvant treatment for breast cancer: Impact of age and cognitive reserve. Journal Of Clinical Oncology, 28(29), 4434–4440. https://doi.org/10.1200/jco.2009.27.0827
Amidi, A., Agerbæk, M., Wu, L. M., Pedersen, A. D., Mehlsen, M., Clausen, C. R., Demontis, D., Børglum, A. D., Harbøll, A., & Zachariae, R. (2017). Changes in cognitive functions and cerebral grey matter and their associations with inflammatory markers, endocrine markers, and APOE genotypes in testicular cancer patients undergoing treatment. Brain Imaging and Behavior, 11(3), 769–783. https://doi.org/10.1007/s11682-016-9552-3
Amidi, A., & Wu, L. M. (2019). Structural brain alterations following adult non-CNS cancers: A systematic review of the neuroimaging literature. Acta Oncologica,58(5), 522–536. https://doi.org/10.1080/0284186x.2018.1563716
Apple, A. C., Ryals, A. J., Alpert, K. I., Wagner, L. I., Shih, P. A., Dokucu, M., & Wang, L. (2017). Subtle hippocampal deformities in breast cancer survivors with reduced episodic memory and self-reported cognitive concerns. NeuroImage: Clinical,14, 685–691. https://doi.org/10.1016/j.nicl.2017.03.004
Ashburner, J., & Friston, K. J. (2000). Voxel-based morphometry–the methods. Neuroimage,11(6 Pt 1), 805–821. https://doi.org/10.1006/nimg.2000.0582
Ashburner, J., & Friston, K. J. (2001). Why voxel-based morphometry should be used. Neuroimage,14(6), 1238–1243. https://doi.org/10.1006/nimg.2001.0961
Ashburner, J., Hutton, C., Frackowiak, R., Johnsrude, I., Price, C., & Friston, K. (1998). Identifying global anatomical differences: Deformation-based morphometry. Human Brain Mapping,6(5–6), 348–357. https://doi.org/10.1002/(SICI)1097-0193(1998)6:5/6%3c348::AID-HBM4%3e3.0.CO;2-P
Bergouignan, L., Lefranc, J. P., Chupin, M., Morel, N., Spano, J. P., & Fossati, P. (2011). Breast cancer affects both the hippocampus volume and the episodic autobiographical memory retrieval. PLoS One,6(10), e25349. https://doi.org/10.1371/journal.pone.0025349
Blommaert, J., Schroyen, G., Vandenbulcke, M., Radwan, A., Smeets, A., Peeters, R., Sleurs, C., Neven, P., Wildiers, H., Amant, F., Sunaert, S., & Deprez, S. (2019). Age-dependent brain volume and neuropsychological changes after chemotherapy in breast cancer patients. Human Brain Mapping, 40(17), 4994–5010. https://doi.org/10.1002/hbm.24753
Boyle, R., Jollans, L., Rueda-Delgado, L. M., Rizzo, R., Yener, G. G., McMorrow, J. P., Knight, S. P., Carey, D., Robertson, I. H., Emek-Savaş, D. D., Stern, Y., Kenny, R. A., & Whelan, R. (2021). Brain-predicted age difference score is related to specific cognitive functions: A multi-site replication analysis. Brain Imaging and Behavior, 15(1), 327–345. https://doi.org/10.1007/s11682-020-00260-3
Carroll, J. E., Van Dyk, K., Bower, J. E., Scuric, Z., Petersen, L., Schiestl, R., & Ganz, P. A. (2019). Cognitive performance in survivors of breast cancer and markers of biological aging. Cancer,125(2), 298–306. https://doi.org/10.1002/cncr.31777
Chen, B. T., Jin, T., Patel, S. K., Ye, N., Sun, C. L., Ma, H., Rockne, R. C., Root, J. C., Saykin, A. J., Ahles, T. A., Holodny, A. I., Prakash, N., …, & Hurria, A. (2018). Gray matter density reduction associated with adjuvant chemotherapy in older women with breast cancer. Breast Cancer Research And Treatment, 172(2), 363–370. https://doi.org/10.1007/s10549-018-4911-y
Cole, J. H. (2018). Neuroimaging studies illustrate the commonalities between ageing and brain diseases. Bioessays,40(7), e1700221. https://doi.org/10.1002/bies.201700221
Cole, J. H. (2020). Multimodality neuroimaging brain-age in UK biobank: Relationship to biomedical, lifestyle, and cognitive factors. Neurobiology Of Aging,92, 34–42. https://doi.org/10.1016/j.neurobiolaging.2020.03.014
Cole, J. H., & Franke, K. (2017). Predicting age using neuroimaging: Innovative brain ageing biomarkers. Trends In Neurosciences,40(12), 681–690. https://doi.org/10.1016/j.tins.2017.10.001
Cole, J. H., Underwood, J., Caan, M. W., De Francesco, D., van Zoest, R. A., Leech, R., & Sharp, D. J. (2017). Increased brain-predicted aging in treated HIV disease. Neurology,88(14), 1349–1357. https://doi.org/10.1212/wnl.0000000000003790
Conroy, S. K., McDonald, B. C., Smith, D. J., Moser, L. R., West, J. D., Kamendulis, L. M., Klaunig, J. E., Champion, V. L., Unverzagt, F. W., & Saykin, A. J. (2013). Alterations in brain structure and function in breast cancer survivors: Effect of post-chemotherapy interval and relation to oxidative DNA damage. Breast Cancer Research And Treatment, 137(2), 493–502. https://doi.org/10.1007/s10549-012-2385-x
Correa, D. D., Root, J. C., Baser, R., Moore, D., Peck, K. K., Lis, E., Shore, T. B., Thaler, H. T., Jakubowski, A., & Relkin, N. (2013). A prospective evaluation of changes in brain structure and cognitive functions in adult stem cell transplant recipients. Brain Imaging Behav, 7(4), 478–490. https://doi.org/10.1007/s11682-013-9221-8
Correa, D. D., Root, J. C., Kryza-Lacombe, M., Mehta, M., Karimi, S., Hensley, M. L., & Relkin, N. (2017). Brain structure and function in patients with ovarian cancer treated with first-line chemotherapy: A pilot study. Brain Imaging Behav,11(6), 1652–1663. https://doi.org/10.1007/s11682-016-9608-4
de Ruiter, M. B., Reneman, L., Boogerd, W., Veltman, D. J., Caan, M., Douaud, G., Lavini, C., Linn, S. C., Boven, E., van Dam, F. S. A. M., & Schagen, S. B. (2012). Late effects of high-dose adjuvant chemotherapy on white and gray matter in breast cancer survivors: Converging results from multimodal magnetic resonance imaging. Human Brain Mapping, 33(12), 2971–2983. https://doi.org/10.1002/hbm.21422
Franke, K., & Gaser, C. (2019). Ten years of BrainAGE as a neuroimaging biomarker of brain aging: What insights have we gained? Frontiers In Neurology,10, 789. https://doi.org/10.3389/fneur.2019.00789
Franke, K., Gaser, C., Manor, B., & Novak, V. (2013). Advanced BrainAGE in older adults with type 2 diabetes mellitus. Frontiers In Aging Neuroscience,5, 90. https://doi.org/10.3389/fnagi.2013.00090
Gaser, C., Franke, K., Klöppel, S., Koutsouleris, N., & Sauer, H. (2013). BrainAGE in mild cognitive impaired patients: Predicting the conversion to Alzheimer’s disease. PLoS One,8(6), e67346. https://doi.org/10.1371/journal.pone.0067346
Good, C. D., Johnsrude, I. S., Ashburner, J., Henson, R. N., Friston, K. J., & Frackowiak, R. S. (2001). A voxel-based morphometric study of ageing in 465 normal adult human brains. Neuroimage,14(1 Pt 1), 21–36. https://doi.org/10.1006/nimg.2001.0786
Guida, J. L., Ahles, T. A., Belsky, D., Campisi, J., Cohen, H. J., DeGregori, J., Fuldner, R., Ferrucci, L., Gallicchio, L., Gavrilov, L., Gavrilova, N., Green, P. A., …, & Hurria, A. (2019). Measuring aging and identifying aging phenotypes in cancer survivors. Journal of the National Cancer Institute, 111(12), 1245–1254. https://doi.org/10.1093/jnci/djz136
Henneghan, A., Rao, V., Harrison, R. A., Karuturi, M., Blayney, D. W., Palesh, O., & Kesler, S. R. (2020). Cortical brain age from pre-treatment to post-chemotherapy in patients with breast cancer. Neurotoxicity Research,37(4), 788–799. https://doi.org/10.1007/s12640-019-00158-z
Hosseini, S. M., Koovakkattu, D., & Kesler, S. R. (2012). Altered small-world properties of gray matter networks in breast cancer. Bmc Neurology,12, 28. https://doi.org/10.1186/1471-2377-12-28
Hurria, A., Jones, L., & Muss, H. B. (2016). Cancer treatment as an accelerated aging process: Assessment, biomarkers, and interventions. American Society of Clinical Oncology Education Book,35, e516-522. https://doi.org/10.1200/edbk_156160
Inagaki, M., Yoshikawa, E., Matsuoka, Y., Sugawara, Y., Nakano, T., Akechi, T., Wada, N., Imoto, S., Murakami, K., & Uchitomi, Y. (2007). Smaller regional volumes of brain gray and white matter demonstrated in breast cancer survivors exposed to adjuvant chemotherapy. Cancer, 109(1), 146–156. https://doi.org/10.1002/cncr.22368
Janelsins, M. C., Kesler, S. R., Ahles, T. A., & Morrow, G. R. (2014). Prevalence, mechanisms, and management of cancer-related cognitive impairment. Int Rev Psychiatry,26(1), 102–113. https://doi.org/10.3109/09540261.2013.864260
Jenkins, V., Thwaites, R., Cercignani, M., Sacre, S., Harrison, N., Whiteley-Jones, H., Mullen, L., Chamberlain, G., Davies, K., Zammit, C., Matthews, L., & Harder, H. (2016). A feasibility study exploring the role of pre-operative assessment when examining the mechanism of ‘chemo-brain’ in breast cancer patients. Springerplus, 5, 390. https://doi.org/10.1186/s40064-016-2030-y
Kaiser, J., Bledowski, C., & Dietrich, J. (2014). Neural correlates of chemotherapy-related cognitive impairment. Cortex; A Journal Devoted to the Study of the Nervous System and Behavior,54, 33–50. https://doi.org/10.1016/j.cortex.2014.01.010
Kesler, S., Janelsins, M., Koovakkattu, D., Palesh, O., Mustian, K., Morrow, G., & Dhabhar, F. S. (2013). Reduced hippocampal volume and verbal memory performance associated with interleukin-6 and tumor necrosis factor-alpha levels in chemotherapy-treated breast cancer survivors. Brain, Behavaior, and Immunity,30 Suppl(0), S109-116. https://doi.org/10.1016/j.bbi.2012.05.017
Koppelmans, V., de Ruiter, M. B., van der Lijn, F., Boogerd, W., Seynaeve, C., van der Lugt, A., Vrooman, H., Niessen, W. J., Breteler, M. M. B., & Schagen, S. B. (2012). Global and focal brain volume in long-term breast cancer survivors exposed to adjuvant chemotherapy. Breast Cancer Research and Treatment, 132(3), 1099–1106. https://doi.org/10.1007/s10549-011-1888-1
Lange, M., Heutte, N., Noal, S., Rigal, O., Kurtz, J. E., Lévy, C., Allouache, D., Rieux, C., Lefel, J., Clarisse, B., Leconte, A., Veyret, C., …, & Joly, F. (2019). Cognitive changes after adjuvant treatment in older adults with early-stage breast cancer. The Oncologist, 24(1), 62–68. https://doi.org/10.1634/theoncologist.2017-0570
Lange, M., Joly, F., Vardy, J., Ahles, T., Dubois, M., Tron, L., Winocur, G., de Ruiter, M. B., & Castel, H. (2019). Cancer-related cognitive impairment: An update on state of the art, detection, and management strategies in cancer survivors. Annals of Oncology, 30(12), 1925–1940. https://doi.org/10.1093/annonc/mdz410
Lepage, C., Smith, A. M., Moreau, J., Barlow-Krelina, E., Wallis, N., Collins, B., MacKenzie, J., & Scherling, C. (2014). A prospective study of grey matter and cognitive function alterations in chemotherapy-treated breast cancer patients. Springerplus, 3, 444. https://doi.org/10.1186/2193-1801-3-444
Li, M., & Caeyenberghs, K. (2018). Longitudinal assessment of chemotherapy-induced changes in brain and cognitive functioning: A systematic review. Neuroscience and Biobehavioral Reviews,92, 304–317. https://doi.org/10.1016/j.neubiorev.2018.05.019
McDonald, B. C., Conroy, S. K., Ahles, T. A., West, J. D., & Saykin, A. J. (2010). Gray matter reduction associated with systemic chemotherapy for breast cancer: A prospective MRI study. Breast Cancer Research and Treatment,123(3), 819–828. https://doi.org/10.1007/s10549-010-1088-4
McDonald, B. C., Conroy, S. K., Smith, D. J., West, J. D., & Saykin, A. J. (2013). Frontal gray matter reduction after breast cancer chemotherapy and association with executive symptoms: a replication and extension study. Brain, Behavior, and Immunity,30 Suppl(0), S117-125. https://doi.org/10.1016/j.bbi.2012.05.007
McDonald, B. C., & Saykin, A. J. (2013). Alterations in brain structure related to breast cancer and its treatment: Chemotherapy and other considerations. Brain Imaging Behav,7(4), 374–387. https://doi.org/10.1007/s11682-013-9256-x
Mentzelopoulos, A., Gkiatis, K., Karanasiou, I., Karavasilis, E., Papathanasiou, M., Efstathopoulos, E., Kelekis, N., Kouloulias, V., & Matsopoulos, G. K. (2021). Chemotherapy-induced brain effects in small-cell lung cancer patients: A multimodal MRI study. Brain Topography, 34(2), 167–181. https://doi.org/10.1007/s10548-020-00811-3
Niu, R., Du, M., Ren, J., Qing, H., Wang, X., Xu, G., Lei, D., & Zhou, P. (2021). Chemotherapy-induced grey matter abnormalities in cancer survivors: A voxel-wise neuroimaging meta-analysis. Brain Imaging and Behavior, 15(4), 2215–2227. https://doi.org/10.1007/s11682-020-00402-7
Schilder, C. M., Seynaeve, C., Beex, L. V., Boogerd, W., Linn, S. C., Gundy, C. M., Huizenga, H. M., Nortier, J. W., van de Velde, C. J., van Dam, F. S., & Schagen, S. B. (2010). Effects of tamoxifen and exemestane on cognitive functioning of postmenopausal patients with breast cancer: Results from the neuropsychological side study of the tamoxifen and exemestane adjuvant multinational trial. Journal of Clinical Oncology, 28(8), 1294–1300. https://doi.org/10.1200/jco.2008.21.3553
Schwarz, D., & Kašpárek, T. (2011). Comparison of two methods for automatic brain morphometry analysis. Radioengineering,20(4), 996–1001.
Seigers, R., Schagen, S. B., Van Tellingen, O., & Dietrich, J. (2013). Chemotherapy-related cognitive dysfunction: Current animal studies and future directions. Brain Imaging Behav,7(4), 453–459. https://doi.org/10.1007/s11682-013-9250-3
Simó, M., Root, J. C., Vaquero, L., Ripollés, P., Jové, J., Ahles, T., & Rodríguez-Fornells, A. (2015). Cognitive and brain structural changes in a lung cancer population. Journal of Thoracic Oncology : Official Publication of the International Association for the Study of Lung Cancer,10(1), 38–45. https://doi.org/10.1097/jto.0000000000000345
Sousa, H., Almeida, S., Bessa, J., & Pereira, M. G. (2020). The developmental trajectory of cancer-related cognitive impairment in breast cancer patients: A systematic review of longitudinal neuroimaging studies. Neuropsychology Review,30(3), 287–309. https://doi.org/10.1007/s11065-020-09441-9
Stouten-Kemperman, M. M., de Ruiter, M. B., Caan, M. W., Boogerd, W., Kerst, M. J., Reneman, L., & Schagen, S. B. (2015). Lower cognitive performance and white matter changes in testicular cancer survivors 10 years after chemotherapy. Human Brain Mapping,36(11), 4638–4647. https://doi.org/10.1002/hbm.22942
Stouten-Kemperman, M. M., de Ruiter, M. B., Koppelmans, V., Boogerd, W., Reneman, L., & Schagen, S. B. (2015). Neurotoxicity in breast cancer survivors ≥ 10 years post-treatment is dependent on treatment type. Brain Imaging and Behavior,9(2), 275–284. https://doi.org/10.1007/s11682-014-9305-0
Yoshikawa, E., Matsuoka, Y., Inagaki, M., Nakano, T., Akechi, T., Kobayakawa, M., Fujimori, M., Nakaya, N., Akizuki, N., Imoto, S., Murakami, K., & Uchitomi, Y. (2005). No adverse effects of adjuvant chemotherapy on hippocampal volume in japanese breast cancer survivors. Breast Cancer Research and Treatment, 92(1), 81–84. https://doi.org/10.1007/s10549-005-1412-6
Acknowledgements
The authors wish to posthumously acknowledge the contributions of John D. West, MS to this research.
Funding
Sources of support for gathering and sharing the data analyzed for this paper include National Institutes of Health grants R03 AG045090, R01 NR010939, and R01 CA101318, Breast Cancer Research Foundation grant BCRF-9 2008-2016, Dutch Cancer Society grant 2009-4284, Research Foundation Flanders (FWO) grant G.048010 N, and Stichting tegen Kanker.
Author information
Authors and Affiliations
Contributions
The primary writing committee for this manuscript consisted of RLD, MBR, and BCM, who also conducted the data analyses. BC, BTC, JAD, KD, SD, BCM, PAN, MBR, AJS, SS, and SBS were responsible for obtaining funding and data acquisition for the original study cohorts. All authors contributed to manuscript writing and editing.
Corresponding author
Ethics declarations
Written informed consent was obtained from all participants as per protocols approved by the respective Institutional Review Board at each participating institution (City of Hope, Indiana University, KU Leuven, The Netherlands Cancer Institute, University of Michigan, and University of Vermont).
Competing interests
Sources of support for gathering and sharing the data analyzed for this paper include National National Institutes of Health grants R03 AG045090, R01 NR010939, and R01 CA101318, Breast Cancer Research Foundation grant BCRF-9 2008-2016, Dutch Cancer Society grant 2009-4284, Research Foundation Flanders (FWO) grant G.048010 N, and Stichting tegen Kanker. AJS has received support from Springer-Nature Publishing (Editorial Office Support as Editor-in-Chief, Brain Imaging and Behavior).
Conflict of interest
All other authors declared no conflicts of interest that are relevant to the content of this article.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 48.8 KB)

Supplementary Fig. 1
Scatter plot of age and brain age values at baseline measurement. CT+ = Breast cancer patients treated with chemotherapy; CT- = Breast cancer patients treated without chemotherapy; NC = Healthy controls with no cancer diagnosis. (PNG 235 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
de Ruiter, M.B., Deardorff, R.L., Blommaert, J. et al. Brain gray matter reduction and premature brain aging after breast cancer chemotherapy: a longitudinal multicenter data pooling analysis. Brain Imaging and Behavior 17, 507–518 (2023). https://doi.org/10.1007/s11682-023-00781-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11682-023-00781-7