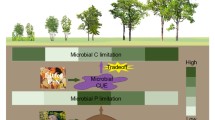
Abstract
Plant stoichiometry and nutrient allocation may reflect adaptation strategies to environmental nutrient changes. Fire, as a major disturbance in forests, mediates soil nutrient availability that may influence plant nutrient dynamics. However, plant–soil stoichiometric allocation strategies during different post-fire periods and the effects of soil, enzymes, and microbial biomass on plant stoichiometry are largely unknown. The pioneer tree species Betula platyphylla in burnt forests of northern China was the object of this study, and severely burned areas selected with different fire years. Nearby unburned areas acted as a control. Carbon (C), nitrogen (N), and phosphorus (P) contents in leaves, branches, and fine roots and rhizosphere soil, C-, N- and P-acquiring enzyme activities were examined. Microbial biomass C, N, and P were measured, and factors influencing C:N:P stoichiometry of plants during the burned area restoration were explored. Our results show that C and N contents in leaves increased with time since fire, while C and P in branches and C, N and P in fine roots decreased. Activities of C-, N-, and P-acquiring enzymes and microbial biomass N increased with time since fire. Redundancy analysis showed that changes in soil N-acquiring enzyme activity, microbial biomass C, and N had significant effects on plant ecological stoichiometry. These results show a significant flexibility in plant nutrient element allocation strategies and C:N:P stoichiometric characteristics. Soil extracellular enzyme activity drives the changes in stoichiometry during the process of post-fire restoration.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Ecological stoichiometry is used to study how plants respond to environmental changes by examining variations in the proportional content and balance of carbon (C), nitrogen (N), and phosphorus (P) (Sterner and Elser 2002). This approach particularly facilitates the establishment of links between different components of the ecosystem (Zechmeister-Boltenstern et al. 2015; Yang et al. 2018). For example, C is the structural basis of a plant and accounts for approximately 50% of its dry weight (Liu et al. 2011). N is an essential component of enzymes and proteins and plays a crucial role in regulating C uptake and photosynthesis (Chen et al. 2016). P is responsible for the composition of RNA and DNA, which transmit genetic information and store energy (Bai et al. 2012). As one of the most important disturbances in nature, fire has a significant impact on the elemental cycle and balance of forest ecosystems by increasing soil temperatures, accelerating nutrient cycling, and altering plant nutrient content (Sterner and Elser 2002; Bond-Lamberty et al. 2007; Dijkstra and Adams 2015; Cavard et al. 2019). Changes in plant elemental content after fire reflect the dynamic nature of vegetation restoration. However, changes in C, N and P levels and their driving mechanisms during post-fire vegetation restoration are unclear.
C, N and P in plants are closely related to growth and physiological metabolism, and their stoichiometric ratios can assess the nutrient limitation status of the environment (Sterner and Elser 2002). Among plant organs, leaves are the organ of photosynthesis, and their nutrient content is stable and sensitive to environmental changes (Tang et al. 2018). Fire has been shown to mitigate nutrient levels by increasing the nutrient content that plants lack when unburned (Dijkstra and Adams 2015). Branches are responsible for transporting and storing nutrients and may also reflect plant nutrient status (Hu et al. 2016). However, there are few reports on branch stoichiometry in post-fire plants. Fine roots absorb water and nutrients and transport organic matter to the soil through respiration and photosynthate consumption and are more sensitive to environmental changes than leaves (Toberman et al. 2014). Higher soil N availability promotes photosynthesis, which increases fine root P and forces plants to adjust resource allocation between organs (Scoffoni et al. 2011). During post-fire ecosystem restoration, increased availability of soil N and P promote fine root nutrient uptake and plant growth (Dijkstra and Adams 2015). Plants change their stoichiometric allocation strategies between leaves, branches, and fine roots over time to adapt to changes in the environment caused by fire (Schreeg et al. 2014; Yang et al. 2015). However, there is a lack of studies exploring the similarities and differences in the patterns of stoichiometric characteristics of different plant organs over time since fire occurrence.
Previous studies on plant stoichiometry are often linked to soil ecological stoichiometry, and a holistic view of plant–soil–microbe interactions is useful for exploring plant growth strategies during post-fire restoration (Bell et al. 2014; Zechmeister-Boltenstern et al. 2015). Soil conditions are key to measuring ecosystem recovery after a fire (Bodí et al. 2012; Schaller et al. 2015). The effect of fire on soil nutrients typically depends on the severity of the fire and the time since the fire. For example, severe fires consume surface organic matter, volatilize large amounts of nutrients (Adkins et al. 2019), and alter C, N, and P cycling. In addition, soil extracellular enzymes and microbial biomass also influence the overall health and development of forest ecosystems (Schoenholtz et al. 2000). It is now widely recognized that fire reduces extracellular enzyme activity, with recovery times ranging from years to decades (Holden et al. 2013; Pellegrini et al. 2020). Moreover, fires affect microbial mortality directly and indirectly by altering soil environmental conditions and nutrient availability (Xu et al. 2012).
The Daxing’an Mountains are in a region sensitive to climate change, and the frequent occurrence of fires has profound effects on plant nutrient utilization, ecosystem structure and function in forest ecosystems (Wu et al. 2013). Betula platyphylla Suk. is a pioneer species in post-fire forest regeneration processes and exploring its stoichiometric characteristics may provide a basis for revealing the strategy of plant resource use in forest restoration. This study will investigate the patterns of changes in C, N, P stoichiometry of different organs (leaf, branch and fine root) of B. platyphlla during post-fire ecosystem restoration and clarify their response mechanism to changes in soil properties and microorganisms. Specifically, it was hypothesized that soil properties may better explain changes in plant stoichiometry during post-fire restoration than soil extracellular enzymes and microbial biomass.
Materials and methods
Study site
The study was carried out in the Huzhong area, Huma County, Daxing’an Mountains, northern China (124°5′1″ E, 52°2′14″ N) (Fig. 1). The area has a cold temperate, continental monsoon climate, where temperatures range from − 52.0 to 32 °C with an average annual temperature of − 4.3 °C. Average annual sunshine duration is approximately 2052 h. Average precipitation in 2021 was 497.7 mm. The soil is brown coniferous forest soil with a thick layer of humus (Calcaric Cambisol, FAO classification). Mosses are abundant in the duff layer (O-horizon) and the litter is dominated by pine needles and B. platyphlla leaves. The main species are Larix gmelinii, B. platyphlla, Pinus sylvestris var. mongolica and Populus davidiana.
Sample collection
We selected five severely burnt areas with different burn times (2020, 2017, 2012, 2004, and 1991) and adjacent unburnt forests to characterize the restoration process in the burned area (Fig. 1). Based on statistical data of forest fires in the Daxing’an Mountains, the specific time of year of fire occurrence was determined by combining remote sensing image recognition, field surveys and forestry department records. Fire severity was classified according to aboveground biomass consumption and tree mortality (Keeley 2009). Areas with tree mortality > 70% were classified as severely burnt (Wei et al. 2016). Due to the large size of the severely burned areas, a central area at least 100 m from the unburned area was selected as the burned sample plot. All plots were essentially the same in terms of climate and topography, and the distance between plots was > 10 km. The selected sample plots were confirmed by field surveys to have had no repeated fires, to be sparsely populated, and to have no obvious anthropogenic disturbances (e.g., post-fire logging, post-fire reforestation).
In July 2021, five young, healthy B. platyphlla trees with diameters at breast height of approximately 5 cm were randomly selected in each sample site, with a distance of 30 m of each other to exclude the influence of tree size on stoichiometric characteristics. Disease-free and fully expanded leaves and branches were collected from each tree. At the same time, the roots of each were excavated after removal of litter and debris from the surface to maintain the integrity of the roots as much as possible. Rhizosphere soil was obtained by shaking the roots and divided into two equal parts, one air dried for physicochemical property analysis, and the other brought to the laboratory and stored at 0−4 °C for the analysis of enzyme activity and microbial biomass. Root samples were washed with deionised water, and roots with diameters < 2 mm were considered as fine roots.
Chemical and biological analyses
The carbon content of leaves, branches, and fine roots was determined using a Multi N/C 2100 analyser (Analytik Jena AG, Jena, Germany). Plant samples were digested with H2SO4–H2O2; soil samples were digested with H2SO4 with the addition of CuSO4 and HClO4. N and P contents in plant and soil solutions were determined using an automatic flow analyzer (AutoAnalyzer III, Bran+Luebbe, Norderstedt, Germany). The pH was determined using a PHS-3C pH meter at a water-soil ratio of 2.5: 1.
The following six enzymes were selected for this study: C-acquiring enzyme (β-1,4-glucosidase (BG)), N-acquiring enzyme (β-1,4-N-acetylglucosaminidase (NAG), leucine aminopeptidase (LAP)) and P-acquiring enzyme (acid phosphatase (ACP)), peroxidase (POD), and polyphenol oxidase (PHO). All enzyme activities were determined using specific kits according to the manufacturer’s instructions (Suzhou Comin Biotechnology Co., Ltd., Suzhou, Jiangsu, China).
Microbial biomass C (MBC) and microbial biomass N (MBN) were determined by the fumigation-extraction method using a Multi 3100 N/C TOC analyzer (Analytik Jena, Jena, Germany) (Vance et al 1987). Microbial biomass P (MBP) was identified by the chloroform fumigation-NaHCO3 leaching-molybdenum antimony colorimetry method using a spectrophotometer. The experimentally derived conversion factors for MBC, MBN and MBP were 0.45, 0.54 and 0.40, respectively (Joergensen and Mueller 1996).
Statistical analysis
All data were assessed for normality and homogeneity (chi-squared of variances) prior to testing. The effects of time since fire on the stoichiometry of plant, soil, enzymes and microbial biomass and their stoichiometric ratios were tested using one-way analysis of variance (ANOVA) and least significant difference (LSD) multiple comparisons. To explore the influence of soil, enzyme activity and microbial biomass on plant stoichiometry, redundancy analysis (RDA) was carried out using the Canoco5 software package after Hellinger transformation and standardization of environmental factor data (soil, enzymes and microbial biomass). All statistical analyses were performed using the R 4.3.1 (R Core Team 2021) and the level of statistically significance was p < 0.05.
Results
Effect of time since fire on the stoichiometry of leaves, branches, and fine roots
In the process of stand restoration after fire, C, N and P levels in different organs were significantly different. Their content in leaves was higher than in branches and fine roots (p < 0.05; Table 1). Leaf C and N tended to increase with time since fire, with the highest contents found in the unburned site (p < 0.05; Table 1). Branch C decreased with time since fire, while branch P content was variable, decreasing and then increasing over time, with the lowest content 30 years after fire (p < 0.05; Table 1). In addition, fine root C and P were stable during the 30 year post-fire period, with the lowest in the unburned site. However, fine root N initially decreased and then increased with time since fire, with the lowest 30 years after fire (p < 0.05; Table 1).
There were significant differences in C:N and N:P ratios in leaves and fine roots, and C:P ratio in branches during forest restoration after fire (p < 0.05; Table 1). Leaf C:N decreased with time since fire, while leaf N:P increased and fluctuated in the range of 3.96–6.01. Branch C:P and fine root C:N first increased and then decreased with time since fire. Fine root N:P fluctuated in the range of 6.89–10.73 (p < 0.05; Table 1). In addition, the interaction between time since fire and tree components had significant effects on plant stoichiometry (p < 0.05; Table 2).
Effect of time since fire on soil, enzyme and microbial biomass stoichiometry
The effects of time since fire on soil C and N contents were insignificant (p > 0.05; Fig. 2). Soil P initially increased and then decreased. In the early post-fire period (1 and 4 years after fire), soil P was 65% and 83% higher than that of the unburned area, respectively, but decreased to that of the unburned area 9 years after fire (p < 0.05; Fig. 2). The pH initially decreased and then increased with time since fire, being highest in the unburned area (p < 0.05; Supplementary Fig. S1).
There were also significant differences in BG, NAG, LAP, ACP, PHO and POD enzyme activities at different times since fire (p < 0.05; Fig. 3). The C-acquiring enzyme (BG), N-acquiring enzyme (NAG+LAP), P-acquiring enzyme (ACP) and PHO activities increased with time since fire, while POD activities decreased trend (p < 0.05; Fig. 3). In addition, enzyme N:P (NAG+LAP: ACP) first increased and then decreased with time since fire (p < 0.05; Fig. 3).
There was a significant increase in MBN with time since fire (p < 0.05; Fig. 4). It was significantly reduced by 161.4% one year after fire compared to the unburned site and recovered to the unburned level by nine years. Both MBC: MBN and MBC: MBP ratios decreased with time since fire, with MBC: MBP decreasing to unburnt levels 4 years after fire and MBC: MBN 17 years after fire (p < 0.05; Fig. 4).
Stoichiometry relationships between plant and soil, enzyme and microbial biomass
Redundancy analysis (RDA) showed that soil nutrients, enzyme activities, and microbial biomass together explained 83.4% and 83.5% of total plant variance in C, N, P and their ratios, respectively (Fig. 5). Of these, 53.3% and 21.8% were explained by RDA1 and RDA2 for C, N and P stoichiometry; 33.9% and 27.1% by RDA1 and RDA2 for stoichiometric ratios. According to the RDA results, the points in approximately the same direction indicate that the stoichiometry distribution of the plant (leaves, branches, and fine roots) and the environmental variables had a strong positive correlation. In particular, LAP, MBC, MBN, enzyme N:P ratios, and ACP had significant effects on C, N and P (Supplementary Table S1), while NAG, POD, and soil C had significant effects on stoichiometric ratios (Supplementary Table S2).
Discussion
Effects of time since fire on stoichiometries among plant organs
There were various patterns of changes in C, N and P in organs with time since fire (Table 1). Leaf C decreased, while branch and fine root C increased (Table 1), indicating that plants allocate more photosynthetic products to branches and fine roots after fire, supplying fine roots for nutrient uptake and branches for nutrient transport. Compared to the unburned area, leaf N decreased significantly in the first year after fire and gradually increased with increasing times since fire. Fine root N was opposite to leaves (Table 1). This may be because severe burning leads to a large volatilization of N compounds and N availability in the soil is reduced. At this time, plants will adopt flexible growth strategies, i.e., change the allocation of resources between organs and absorb more N by fine roots (Wright and Sutton-Grier 2012; Song and Liu 2019). At the same time, to meet growth demands, plants will improve leaf photosynthetic efficiency and N consumption during photosynthesis will be greater than N supply to leaves, resulting in a decrease in leaf N content after fire (Ordoñez et al. 2009; Muqaddas et al. 2015). With increasing time since fire, ecosystem restoration and nutrient conditions improved, and changes in N in different organs gradually stabilized (Scoffoni et al. 2011). In addition, there was no change in leaf P in the process of restoration after fire, which may be due to the need for plants to ensure the supply of P to improve photosynthesis. These results suggest that fire alters nutrient allocation strategies in plants, leading to differences in C, N and P in different organs (Pellegrini et al. 2015).
Plant stoichiometric ratios also changed significantly at different times since fire, as influenced by the absolute content of C, N, and P (Cui et al. 2010; Toberman et al. 2014). Leaf C:N ratios were higher in the first year after fire than in the unburned site, with a decrease with time, while the opposite was found for leaf N:P (Table 1) due to the decrease in N in the first year after fire. Leaf N:P ratios decreased and then increased with time but fluctuated between 3.96 and 6.01 (Table 1). According to the N:P threshold (Koerselman and Meuleman 1996), the “breakpoints” of the N:P ratio are N-limited (< 14), P-limited (> 16) and co-limited (between 14 and 16). Together, these results suggest that fire did not alter the N-limited status of plants in the Daxing’an Mountains and are similar to previous studies that N remains the limiting factor for growth in boreal forest ecosystems (Kong et al. 2022).
Effects of time since fire on the properties of rhizosphere soil
It is recognized that fire increases pH, however we found a significant decrease in pH after fire (Supplementary Fig. S1). This may be due to the differences in physical and chemical properties between rhizosphere and non-rhizosphere soils (Toberman et al. 2014; Carrillo et al. 2017). This study collected rhizosphere soils, whereas many previous studies collected non-rhizosphere soil. The form of nitrogen (i.e., NH4+, NO3−) supplied to plants determines the uptake of anions and cations (Zhang and Cao 1992). NH4+-N increased significantly after fire and uptake would make cation uptake greater than anion uptake, resulting in a decrease in rhizosphere pH (Supplementary Fig. S1). With increasing time since fire, NH4+-N gradually decreased and eventually returned to the unburnt level. At this point, plants absorb NO3−N and the uptake of anions is greater than that of cations, and thus pH began to increase (Zhang and Cao 1992).
There were no significant differences in soil C and N levels before and after fire (p > 0.05; Fig. 2). This is inconsistent with the widely recorded decrease in surface soil C and N after severe fires (Francos et al. 2018; Adkins et al. 2019), possibly due to the rhizosphere priming effect that occurs in the rhizosphere environment during the post-fire restoration process (Cheng and Kuzyakov 2005). In particular, under N-limiting conditions, microorganisms release C to access organic N (Carrillo et al. 2017). In addition, the transformation of organic matter is mediated by extracellular enzymes. C- and N-acquiring enzymes recover quickly after fire and release organic molecules into the soil through microbial cell cleavage, catalyzing the production of assimilable molecules containing C and N which effectively mitigate the decline of C and N in the rhizosphere (Sinsabaugh et al. 2009). We also found that P increased the first year after fire (p > 0.05; Fig. 2). Due to the relatively high evaporation temperature of P, there was no significant loss of phosphorous during fire, and P from plants enters the soil via ash deposition, resulting in an increase after fire (Boring et al. 2004). With the increase of time since fire, P decreased significantly at 9 and 17 years after fire, which may be related to surface runoff or erosion, and the most erodible component is ash (Bodí et al. 2012). Severe fires cause litter loss and lessen soil resistance to erosion, increasing the risk runoff (Murphy et al. 2006). In addition, it has also been suggested that the decrease in P may be due to the uptake and utilization of available P by regenerated plants (Zhang et al. 2015).
Effect of time since fire on the stoichiometry of soil extracellular enzymes and microbial biomass
Soil extracellular enzymes play an important role in the microbial decomposition of complex organic matter and the transformation of nutrients, and act as key drivers of nutrient cycling (Paz-Ferreiro and Fu 2016; Memoli et al. 2021). It is generally considered that soil extracellular enzyme activity and microbial biomass decrease immediately after fire and remained low for several years after fire (Holden et al. 2013; Ribeiro-Kumara et al. 2020). Our study also showed that fire significantly reduced C, N, P acquisition enzyme activity and MBN (Figs. 3, 4). This may be mainly due to three aspects: (1) fire-induced high temperatures reduce microbial biomass, resulting in a decrease in soil extracellular enzyme activity (Dooley and Treseder 2012), and once the rising temperature decreases and ash deposition occurs, extracellular enzyme activity may increase (Hu et al. 2023); (2) fire alters the quantity and quality of organic matter inputs, leading to substrate limitation of microbial populations (Pellegrini et al. 2020); and (3) the feedback effect of fire-induced changes in soil nutrient availability on extracellular enzymes (Holden et al. 2013; Ribeiro-Kumara et al. 2020). However, it is not clear how soil nutrient availability mediates extracellular enzyme activity after fire. Studies at a global scale have shown that N-acquiring enzymes are positively correlated with soil N availability after fire, and P-acquiring enzymes are negatively correlated with P availability (Zhou et al. 2022). Other studies did not find a direct relationship between N-acquiring enzymes and N availability or P-acquiring enzymes and P availability after fire but found that N-acquiring enzymes were positively correlated with P availability (Hu et al. 2023). In this study, soil N-acquiring enzymes were significantly correlated with both N and P availability, while soil P-acquiring enzymes were not significantly correlated with either (Supplementary Table S3). This may be due to the fact that previous studies were carried out in non-rhizosphere soils, and there are different scaling relationships of N- and P-acquiring enzymes between rhizosphere and non-rhizosphere soils (Tapia-Torres et al. 2015). In addition, plant roots produce acid phosphatase (Staddon et al. 1998), resulting in significant differences in ACP activity between rhizosphere and non-rhizosphere soils.
Carbon, N, and P-acquiring enzymes showed differences with increasing time since fire. C-acquiring enzymes in rhizosphere soil completely recovered to unburnt levels 4 years after fire, while N and P-acquiring enzymes were not restored 30 years after fire (Figs. 3, 4). López-Poma and Bautista (2014) also reported differences in soil extracellular enzymes during post-fire restoration, and found that P- and C-acquiring enzymes were in contrast, with C-acquiring enzymes recovering faster than P-acquiring enzymes. This was attributed to the long-term decline in acid phosphatase to the combined effect of availability and post-fire drought. Our results show that the effect of fire on soil extracellular enzymes is temporary, and that C-acquiring enzymes have better fire resistance than N- and P-acquiring enzymes.
Factors leading to changes in plant stoichiometry at different times since fire
Ecological stoichiometry typically uses mixed non-rhizosphere soils to explore the relationships between different components at an ecosystem scale, thus overcoming small-scale plant heterogeneity (Sterner and Elser 2002; Yang et al. 2018). We selected the rhizosphere soil of a single tree species to assess ecological stoichiometric characteristics at a finer spatial scale. We found that plant stoichiometry was correlated with soil properties, extracellular enzymes and microbial biomass (Supplementary Fig. S2), with stronger correlations between extracellular enzyme stoichiometry and the stoichiometry of different plant organs (leaves, branches, fine roots), suggesting that plant stoichiometric characteristics may be more influenced by soil extracellular enzyme stoichiometry. Similarly, there were more significant correlations between soil extracellular enzyme stoichiometry ratios and plant stoichiometry ratios than between soil properties and microbial biomass (Supplementary Fig. S3). The results suggest that soil extracellular enzyme stoichiometry could predict changes in plant stoichiometric characteristics during post-fire restoration, which is in contrast with our hypothesis that soil properties are the primary regulator of plant stoichiometry. This difference may be due to plants relying more on internal element regulation mechanisms after fire, making them less dependent on changes in nutrient availability under unpredictable environmental conditions (Di Palo and Fornara 2017). However, most current studies have focused on the effects of plants on soil extracellular enzymes. For example, Cui et al. (2018) reported a greater effect of plants on the stoichiometry characteristics of extracellular enzymes than on soil properties. Peng and Wang (2016) also found that plants can better reflect the stoichiometric of soil extracellular enzymes. However, the relative contribution of rhizospheric nutrients and extracellular enzyme stoichiometry to plant stoichiometry during post-fire restoration still requires further study.
RDA results also showed that soil N-acquiring enzymes, MBC, and MBN, had more effects on plant stoichiometry in different organs than soil properties in different post-fire restoration years (Supplementary Table S1). This further indicates that plant stoichiometry was more influenced by soil extracellular enzymes and microbial biomass than by soil properties during post-fire restoration. Soil enzymes and microbial biomass may serve as sensitive indicators in the process of plant restoration in burnt areas (Raiesi and Salek-Gilani 2018).
Conclusion
This study analyzed the drivers of ecological stoichiometry of the pioneer species B. platyphlla in burnt areas. First, there were relatively long-term effects of fire on plant, rhizosphere soil, and microbial stoichiometry. Secondly, there were significant variations in C, N, P stoichiometry of different plant organs at different times since fire. Plants altered their nutrient allocation strategies to adapt to environmental changes. Thirdly, there was a strong correlation between plant–soil–microbial systems in burned areas, and soil extracellular enzymes and microbial biomass are better suited to explain changes in plant stoichiometry during post-fire recovery than soil properties. These results suggest that fire alters the biogeochemical cycling of C, N, and P and that there are complex plant–soil–microbial interactions during post-fire restoration. In addition, soil extracellular enzymes and microbial biomass may serve as sensitive indicators of plant restoration in burned areas. In future, the relative contributions of soil nutrients and extracellular enzymes, and microbial biomass to plant nutrient stoichiometry at different times since fire need to be studied.
References
Adkins J, Serman J, Miesel J (2019) Soil carbon pools and fluxes vary across a burn severity gradient three years after wildfire in Sierra Nevada mixed-conifer forest. Geoderma 333:10–22. https://doi.org/10.1016/j.geoderma.2018.07.009
Bai YF, Wu JG, Clark CM, Pan QM, Zhang LX, Chen SP, Wang QB, Han XG (2012) Grazing alters ecosystem functioning and C: N: P stoichiometry of grasslands along a regional precipitation gradient. J Appl Ecol 49(6):1204–1215. https://doi.org/10.1111/j.1365-2664.2012.02205.x
Bell C, Carrillo Y, Boot CM, Rocca JD, Pendall E, Wallenstein MD (2014) Rhizosphere stoichiometry: are C: N: P ratios of plants, soils, and enzymes conserved at the plant species-level? New Phytol 201(2):505–517. https://doi.org/10.1111/nph.12531
Bodí MB, Doerr SH, Cerdà A, Mataix-Solera J (2012) Hydrological effects of a layer of vegetation ash on underlying wettable and water repellent soil. Geoderma 191:14–23. https://doi.org/10.1016/j.geoderma.2012.01.006
Bond-Lamberty B, Peckham SD, Ahl DE, Gower ST (2007) Fire as the dominant driver of central Canadian boreal forest carbon balance. Nature 450:89–92. https://doi.org/10.1038/nature06272
Boring LR, Hendricks JJ, Wilson CA, Mitchell RJ (2004) Season of burn and nutrient losses in a longleaf pine ecosystem. Int J Wildland Fire 13(4):443–453. https://doi.org/10.1071/WF03060
Carrillo Y, Bell C, Koyama A, Canarini A, Boot CM, Wallenstein M, Pendall E (2017) Plant traits, stoichiometry and microbes as drivers of decomposition in the rhizosphere in a temperate grassland. J Ecol 105(6):1750–1765. https://doi.org/10.1111/1365-2745.12772
Cavard X, Bergeron Y, Paré D, Nilsson MC, Wardle DA (2019) Disentangling effects of time since fire, overstory composition and organic layer thickness on nutrient availability in Canadian boreal forest. Ecosystems 22:33–48. https://doi.org/10.1007/s10021-018-0251-3
Chen YL, Chen LY, Peng YF, Ding JZ, Li F, Yang GB, Kou D, Liu L, Fang K, Zhang BB, Wang J, Yang YH (2016) Linking microbial C: N: P stoichiometry to microbial community and abiotic factors along a 3500-km grassland transect on the Tibetan plateau. Glob Ecol Biogeogr 25(12):1416–1427. https://doi.org/10.1111/geb.12500
Cheng W, Kuzyakov Y (2005) Root effects on soil organic matter decomposition. In: Zobel RW, Wright SF (eds) Roots and soil management: interactions between roots and the soil. ASA-SSSA, Madison, pp 19–143. https://doi.org/10.2134/agronmonogr48.c7
Cui Q, Lu XT, Wang QB, Han XG (2010) Nitrogen fertilization and fire act independently on foliar stoichiometry in a temperate steppe. Plant Soil 334:209–219. https://doi.org/10.1007/s11104-010-0375-5
Cui YX, Fang LC, Guo XB, Wang X, Zhang YJ, Li PF, Zhang XC (2018) Ecoenzymatic stoichiometry and microbial nutrient limitation in rhizosphere soil in the arid area of the northern Loess Plateau, China. Soil Biol Biochem 116:11–21. https://doi.org/10.1016/j.soilbio.2017.09.025
Di Palo F, Fornara DA (2017) Plant and soil nutrient stoichiometry along primary ecological successions: Is there any link? PLoS ONE 12(8):e0182569. https://doi.org/10.1371/journal.pone.0182569
Dijkstra FA, Adams MA (2015) Fire eases imbalances of nitrogen and phosphorus in woody plants. Ecosystems 18:769–779. https://doi.org/10.1007/s10021-015-9861-1
Dooley SR, Treseder KK (2012) The effect of fire on microbial biomass: a meta-analysis of field studies. Biogeochemistry 109:49–61. https://doi.org/10.1007/s10533-011-9633-8
Francos M, Úbeda X, Pereira P, Alcañiz M (2018) Long-term impact of wildfire on soils exposed to different fire severities. A case study in Cadiretes Massif (NE Iberian Peninsula). Sci Total Environ 615:664–671. https://doi.org/10.1016/j.scitotenv.2017.09.311
Holden SR, Gutierrez A, Treseder KK (2013) Changes in soil fungal communities, extracellular enzyme activities, and litter decomposition across a fire chronosequence in Alaskan boreal forests. Ecosystems 16:34–46. https://doi.org/10.1007/s10021-012-9594-3
Hu XY, Zhu JX, Wang CK, Zheng TL, Wu QQ, Yao H, Fang JY (2016) Impacts of fire severity and post-fire reforestation on carbon pools in boreal larch forests in Northeast China. J Plant Ecol 9(1):1–9. https://doi.org/10.1093/jpe/rtv036
Hu MJ, Wang JL, Lu LL, Shao PS, Zhou ZX, Wang D, Han SJ, Osborne B, Chen J (2023) Post-fire soil extracellular enzyme activities in subtropical–warm temperate climate transitional forests. Land Degrad Dev 34(7):1973–1983. https://doi.org/10.1002/ldr.4582
Joergensen RG, Mueller T (1996) The fumigation-extraction method to estimate soil microbial biomass: calibration of the kEN value. Soil Biol Biochem 28(1):33–37. https://doi.org/10.1016/0038-0717(95)00101-8
Keeley JE (2009) Fire intensity, fire severity and burn severity: a brief review and suggested usage. Int J Wildland Fire 18(1):116–126. https://doi.org/10.1071/WF07049
Koerselman W, Meuleman A (1996) The vegetation N: P ratio: a new tool to detect the nature of nutrient limitation. J Appl Ecol 33:1441–1450. https://doi.org/10.2307/2404783
Kong JJ, Xiang XJ, Yang J (2022) Wildfire alters the linkage between total and available soil C:N: P ratios and the stoichiometric effects on fine root growth in a Chinese boreal larch forest. Plant Soil 471:211–225. https://doi.org/10.1007/s11104-021-05215-1
Liu RT, Zhao HL, Zhao XY, Drake S (2011) Facilitative effects of shrubs in shifting sand on soil macro-faunal community in Horqin Sand Land of Inner Mongolia, Northern China. Eur J Soil Biol 47(5):316–321. https://doi.org/10.1016/j.ejsobi.2011.07.006
López-Poma R, Bautista S (2014) Plant regeneration functional groups modulate the response to fire of soil enzyme activities in a Mediterranean shrub land. Soil Biol Biochem 79:5–13. https://doi.org/10.1016/j.soilbio.2014.08.016
Memoli V, Santorufo L, Santini G, Musella P, Barile R, De Marco A, Di Natale G, Trifuoggi M, Maisto G (2021) Role of seasonality and fire in regulating the enzymatic activities in soils covered by different vegetation in a Mediterranean area. Appl Sci 11(18):8342. https://doi.org/10.3390/app11188342
Muqaddas B, Zhou XQ, Lewis T, Wild C, Chen CR (2015) Long-term frequent prescribed fire decreases surface soil carbon and nitrogen pools in a wet sclerophyll forest of Southeast Queensland, Australia. Sci Total Environ 536:39–47. https://doi.org/10.1016/j.scitotenv.2015.07.023
Murphy JD, Johnson DW, Miller WW, Walker RF, Carroll EF, Blank RR (2006) Wildfire effects on soil nutrients and leaching in a Tahoe Basin watershed. J Environ Qual 35(2):479–489. https://doi.org/10.2134/jeq2005.0144
Ordoñez JC, Van Bodegom PM, Witte JPM, Wright IJ, Reich PB, Aerts R (2009) A global study of relationships between leaf traits, climate and soil measures of nutrient fertility. Glob Ecol Biogeogr 18(2):137–149. https://doi.org/10.1111/j.1466-8238.2008.00441.x
Paz-Ferreiro J, Fu SL (2016) Biological indices for soil quality evaluation: perspectives and limitations. Land Degrad Dev 27(1):14–25. https://doi.org/10.1002/ldr.2262
Pellegrini AFA, Hedin LO, Staver AC, Govender N (2015) Fire alters ecosystem carbon and nutrients but not plant nutrient stoichiometry or composition in tropical savanna. Ecology 96(5):1275–1285. https://doi.org/10.1890/14-1158.1
Pellegrini AFA, Hobbie SE, Reich PB, Jumpponen A, Brookshire ENJ, Caprio AC, Coetsee C, Jackson RB (2020) Repeated fire shifts carbon and nitrogen cycling by changing plant inputs and soil decomposition across ecosystems. Ecol Monogr 90(4):e01409. https://doi.org/10.1002/ecm.1409
Peng XQ, Wang W (2016) Stoichiometry of soil extracellular enzyme activity along a climatic transect in temperate grasslands of northern China. Soil Biol Biochem 98:74–84. https://doi.org/10.1016/j.soilbio.2016.04.008
R Core Team (2021) R: A language and environment for statistical computing The R Project for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Raiesi F, Salek-Gilani S (2018) The potential activity of soil extracellular enzymes as an indicator for ecological restoration of rangeland soils after agricultural abandonment. Appl Soil Ecol 126:140–147. https://doi.org/10.1016/j.apsoil.2018.02.022
Ribeiro-Kumara C, Pumpanen J, Heinonsalo J, Metslaid M, Orumaa A, Jõgiste K, Berninger F, Köster K (2020) Long-term effects of forest fires on soil greenhouse gas emissions and extracellular enzyme activities in a hemiboreal forest. Sci Total Environ 718:135291. https://doi.org/10.1016/j.scitotenv.2019.135291
Schaller J, Tischer A, Struyf E, Bremer M, Belmonte DU, Potthast K (2015) Fire enhances phosphorus availability in topsoils depending on binding properties. Ecology 96(6):1598–1606. https://doi.org/10.1890/14-1311.1
Schoenholtz SH, Van Miegroet H, Burger JA (2000) A review of chemical and physical properties as indicators of forest soil quality: challenges and opportunities. For Ecol Manage 138(1–3):335–356. https://doi.org/10.1016/S0378-1127(00)00423-0
Schreeg LA, Santiago LS, Wright SJ, Turner BL (2014) Stem, root, and older leaf N: P ratios are more responsive indicators of soil nutrient availability than new foliage. Ecology 95(8):2062–2068. https://doi.org/10.1890/13-1671.1
Scoffoni C, Rawls M, Mckown A, Cochard H, Sack L (2011) Decline of leaf hydraulic conductance with dehydration: relationship to leaf size and venation architecture. Plant Physiol 156(2):832–843. https://doi.org/10.1104/pp.111.173856
Sinsabaugh RL, Hill BH, Follstad Shah JJ (2009) Ecoenzymatic stoichiometry of microbial organic nutrient acquisition in soil and sediment. Nature 462:795–798. https://doi.org/10.1038/nature08632
Song ZP, Liu YH (2019) Fire intensity affects the relationship between species diversity and the N utilization stability of dominant species. Forests 10(3):207. https://doi.org/10.3390/f10030207
Staddon WJ, Duchesne LC, Trevors JT (1998) Acid phosphatase, alkaline phosphatase and arylsulfatase activities in soils from a jack pine (Pinus banksiana Lamb.) ecosystem after clear-cutting, prescribed burning, and scarification. Biol Fertil Soils 27:1–4. https://doi.org/10.1007/s003740050390
Sterner RW, Elser JJ (2002) Ecological stoichiometry: the biology of elements from molecules to the biosphere. The University of Chicago Press Princeton, New Jersey. https://doi.org/10.1515/9781400885695
Tang ZY, Xu WT, Zhou GY, Xie ZQ (2018) Patterns of plant carbon, nitrogen, and phosphorus concentration in relation to productivity in China’s terrestrial ecosystems. Proc Natl Acad Sci 115(16):4033–4038. https://doi.org/10.1073/pnas.170029511
Tapia-Torres Y, Elser JJ, Souza V, García-Oliva F (2015) Ecoenzymatic stoichiometry at the extremes: how microbes cope in an ultra-oligotrophic desert soil. Soil Biol Biochem 87:34–42. https://doi.org/10.1016/j.soilbio.2015.04.007
Toberman H, Chen C, Lewis T, Elser JJ (2014) High-frequency fire alters C:N: P stoichiometry in forest litter. Glob Change Biol 20(7):2321–2331. https://doi.org/10.1111/gcb.12432
Vance ED, Brookes PC, Jenkinson DS (1987) An extraction method for measuring soil microbial biomass C. Soil Biol Biochem 19(6):703–707. https://doi.org/10.1016/0038-0717(87)90052-6
Wei YM, Hu HQ, Sun JB, Yuan Q, Sun L, Liu HF (2016) Effect of fire intensity on active organic and total soil carbon in a Larix gmelinii forest in the Daxing’anling Mountains, Northeastern China. J for Res 27:1351–1359. https://doi.org/10.1007/s11676-016-0251-0
Wright JP, Sutton-Grier A (2012) Does the leaf economic spectrum hold within local species pools across varying environmental conditions? Funct Ecol 26(6):1390–1398. https://doi.org/10.1111/1365-2435.12001
Wu ZW, He HS, Liang Y, Cai LY, Lewis BJ (2013) Determining relative contributions of vegetation and topography to burn severity from LANDSAT imagery. Environ Manage 52:821–836. https://doi.org/10.1007/s00267-013-0128-3
Xu YH, Jian S, Lin Q, Jing M, Shi YW, Lou K (2012) Effects of a surface wildfire on soil nutrient and microbial functional diversity in a shrubbery. Acta Ecol Sin 32(5):258–264. https://doi.org/10.1016/j.chnaes.2012.07.007
Yang XJ, Huang ZY, Zhang KL, Cornelissen JHC (2015) C:N:P stoichiometry of Artemisia species and close relatives across northern China: unravelling effects of climate, soil and taxonomy. J Ecol 103(4):1020–1031. https://doi.org/10.1111/1365-2745.12409
Yang Y, Liu BR, An SS (2018) Ecological stoichiometry in leaves, roots, litters and soil among different plant communities in a desertified region of Northern China. CATENA 166:328–338. https://doi.org/10.1016/j.catena.2018.04.018
Zechmeister-Boltenstern S, Keiblinger KM, Mooshammer M, Penuelas J, Richter A, Sardans J, Wanek W (2015) The application of ecological stoichiometry to plant microbial-soil organic matter transformations. Ecol Monogr 85(2):133–155. https://doi.org/10.1890/14-0777.1
Zhang FS, Cao YP (1992) Rhizosphere dynamic processes and plant nutrition. Acta Pedol Sin 29(3):239–250 (in Chinese)
Zhang L, Liu L, Pan K, Li W, Wang Y, Deng M, Yang X (2015) Post-wildfire soil and plant foliar nutrient ratios and soil fungi: bacterial ratios in alpine meadows on the southeastern Qinghai-Tibet Plateau. Int J Wildland Fire 24(7):933–939. https://doi.org/10.1071/WF14147
Zhou Y, Biro A, Wong MY, Batterman SA, Staver AC (2022) Fire decreases soil enzyme activities and reorganizes microbially mediated nutrient cycles: a meta-analysis. Ecology 103(11):e3807. https://doi.org/10.1002/ecy.3807
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Project funding: This study was supported by National Natural Science Foundation of China (No. 32071777), and the Fundamental Research Funds for the Central Universities (2572021BA04).
The online version is available at http://www.springerlink.com.
Corresponding editor: Lei Yu.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Cai, H., Lin, Y., Liang, Y. et al. Time since fire affects ecological stoichiometry of plant–soil–microbial systems of Betula platyphylla, a pioneer species in burnt areas of China’s boreal forest. J. For. Res. 35, 105 (2024). https://doi.org/10.1007/s11676-024-01753-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11676-024-01753-3