Abstract
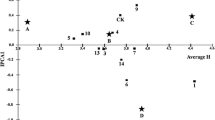
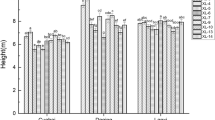
White birch (Betula platyphylla) is precious material for pulpwood and widely distributed in 14 provinces of China. Previous study indicated that inhibited expression of a gene encoding an auxin amide synthase, BpGH3.5, in transgenic plants reduced the level of IAA–amino acid conjugation, resulting in more free IAA, thereby better growth of birch. Utilizing transgenic-BpGH3.5 lines to increase wood production in a wide range of environments is the goal for breeders. In three field trials here, we measured tree height, diameter at breast height, and volume of 16 BpGH3.5-transgenic 7-year-old white birch lines (including 12 antisense strand lines and 4 overexpression lines) and a wild-type white birch line from three sites that varied greatly in their environmental conditions. To select elite BpGH3.5-transgenic lines for each target environment, we used an additive main effects and multiplicative interaction model to analyze genotype by environment interaction, growth adaptability and stability. The selection criteria for elite transgenic lines were set as the average volume plus 0.75 times the standard deviation for the tested lines at each test site. Results showed that the effect of line and site for height was highly significant (P < 0.01), and the effect of line × site was significant (P < 0.05); selected as the elite lines were FG12, FG13 and FG27 at the Maoershan Experimental Forest Farm, FG13 and FG32 at the Shidaohe Forest Farm, and FG3 and FG31 at the Ecological Experiment Forest Farm. These seven high-yield, stable lines can now be tested in production trials or adjacent trial areas with similar environmental conditions, while the high-yield, unstable lines should be tested in production trials in areas deemed suitable for their growth. These results provide guidance on which released transgenic elite lines will grow best in a wide range of conditions.



Similar content being viewed by others
References
Bose LK, Jambhulkar NN, Pande K, Singh ON (2014) Use of AMMI and other stability statistics in the simultaneous selection of rice genotypes for yield and stability under direct-seeded conditions. Chilean J Agri Res 74(1):3–9. https://doi.org/10.4067/S0718-58392014000100001
Chen J, Liu C, Wang S, Xu W, Huang H, Jiang J (2018) A preliminary study on function of BpTOPP1 gene in Betula platyphylla × B. pendula. J Northeast For Univ 46(8):13–19. https://doi.org/10.13759/j.cnki.dlxb.2018.08.003 (in Chinese)
Chen S, Wang Y, Yu L, Zheng T, Wang S, Yue Z, Jiang J, Kumari S, Zheng C, Tang H, Li J, Li Y, Chen J, Zhang W, Kuang H, Jon SR, Patric XZ, Li H, Shu S, Yordan SY, Huang H, David MG, Gai Y, Qi Q, Min J, Xu C, Wang S, Qu G, Andrew HP, David S, Wei H, Liu G, Yang C (2021) Genome sequence and evolution of Betula platyphylla. Horticulture Res 8(1):37. https://doi.org/10.1038/s41438-021-00481-7
Chen M (2017) Overexpressing early auxin-responsive gene CsGH3 enhances canker resistance in citrus. Master’s thesis. Southwest University. Chongqing, China
Ding X, Cao Y, Huang L, Zhao J, Xu C, Li X, Wang S (2008) Activation of the Indole-3-acetic acid-amido synthetase GH3-8 suppresses expansin expression and promotes salicylate- and jasmonate-independent basal immunity in rice. Plant Cell 20(1):228–240. https://doi.org/10.1105/tpc.107.055657
Du H, Wu N, Fu J, Wang S, Li X, Xiao J, Xiong L (2012) A GH3 family member, OsGH3-2, modulates auxin and abscisic acid levels and differentially affects drought and cold tolerance in rice. J Exp Bot 63(18):6467–6480. https://doi.org/10.1093/jxb/ers300
Fang Z, Jiang J, Wang F, Lv D, Gu C, Li T (2018) Overexpression of BpCHS confers changes of pigment content in leaves and phloem and other phenotypic traits in transgenic birch. J Northeast For Univ 46(6):8–13. https://doi.org/10.13759/j.cnki.dlxb.2018.06.002 (in Chinese)
Gang H, Li R, Zhao Y, Liu G, Chen S, Jiang J (2019a) Loss of GLK1 transcription factor function reveals new insights in chlorophyll biosynthesis and chloroplast development. J Exp Bot 70(12):3125–3138. https://doi.org/10.1093/jxb/erz128
Gang H, Liu G, Zhang M, Zhao Y, Jiang J, Chen S (2019b) Comprehensive characterization of T-DNA integration induced chromosomal rearrangement in a birch T-DNA mutant. BMC Genomics 20(1):1–11. https://doi.org/10.1186/s12864-019-5636-y
Guilfoyle TJ, Ulmasov T, Hagen G (1998) The ARF family of transcription factors and their role in plant hormone-responsive transcription. Cell Mol Life Sci (CMLS) 54(7):619–627. https://doi.org/10.1007/s000180050190
Huang H, Wang S, Jiang J, Liu G, Li H, Chen S, Xu H (2013) Overexpression of BpAP1 induces early flowering and produces dwarfism in Betula platyphylla × B. pendula. Physiol Plantarum 151(4):495–506. https://doi.org/10.1111/ppl.12123
Li X, Zhu Z (1998) Selection of better combinations of genotypes and sites of forest trees. J Beijing For Univ 20(3):15–18. https://doi.org/10.13332/j.1000-1522.1998.03.003 (in Chinese)
Li B, Gu W, Xia L, Li X, Gan S (2001) Genetic variation and provenance selection of Chinese tuliptree. For Res 03:237–241. https://doi.org/10.3321/j.issn:1001-1498.2001.03.001
Li Y, Zuo KJ, Tang K (2008) A survey of functional studies of the GH3 gene family in plants. Chin Bull Botany 25(5):507–515. https://doi.org/10.3724/SP.J.1005.2008.01083 (in Chinese)
Li Y, Yang G, Wei R, Sun Y, Guo Y, Zhang R, Liu G (2013a) TabZIP transferred Betula platyphylla generation and salt tolerance analysis. J Nanjing For Univ (Nat Sci Edition) 37(05):6–12 (in Chinese)
Li Z, Zhao X, Yang C, Wang G, Wang F, Zhang L (2013) Variation and growth adaptability analysis of transgenic Populus simonii × P. nigra lines carrying TaLEA gene. J Beijing For Univ 35(2):57–62. https://doi.org/10.13332/j.1000-1522.2013.02.024 (in Chinese)
Li R, Chen S, Liu G, Han R, Jiang J (2017) Characterization and identification of a woody lesion mimic mutant lmd, showing defence response and resistance to Alternaria alternate in birch. Sci Rep 7(1):11308. https://doi.org/10.1038/s41598-017-11748-2
Liscum E, Reed JW (2002) Genetics of Aux/IAA and ARF action in plant growth and development. Plant Mol Biol 49(3–4):387–400. https://doi.org/10.1023/a:1015255030047
Liu M, Yin S, Si D, Shao L, Li Y, Zheng M, Wang F, Li S, Liu G, Zhao X (2015) Variation and genetic stability analyses of transgenic TaLEA poplar clones from four different sites in China. Euphytica 206(2):331–342. https://doi.org/10.1007/s10681-015-1471-7
Mellor N, Band LR, Pencik A, Novak O, Rashed A, Holman T, Wilson MH, Voss U, Bishopp A, King JR, Ljung K, Bennett MJ, Owen MR (2016) Dynamic regulation of auxin oxidase and conjugating enzymes AtDAO1 and GH3 modulates auxin homeostasis. Proc Natl Acad Sci 113(39):201604458. https://doi.org/10.1073/pnas.1604458113
Nagamitsu T, Nagasaka K, Yoshimaru H, Tsumura Y (2014) Provenance tests for survival and growth of 50-year-old Japanese larch (Larix kaempferi) trees related to climatic conditions in central Japan. Tree Genet Genomes 10(1):87–99. https://doi.org/10.1007/s11295-013-0666-0
Nakazawa M, Yabe N, Ichikawa T, Yamamoto YY, Yoshizumi T, Hasunuma K, Matsui M (2001) DFL1, an auxin-responsive GH3 gene homologue, negatively regulates shoot cell elongation and lateral root formation, and positively regulates the light response of hypocotyl length. Plant J 25(2):213–221. https://doi.org/10.1046/j.1365-313x.2001.00957.x
Ning K, Liu X, Lin Y, Zhao X, Liu G, Yang Y (2015) Germplasm selection of the progeny genetic variation and superior pulpwood of Betula platyphylla. Bull Botanical Res 35(1):39–46. https://doi.org/10.7525/j.issn.1673-5102.2015.01.008 (in Chinese)
Shen X (2015) Selection and breeding of Pinus tabulaeformis and Larix principis-rupprechtii: Practice and Theorty, 1st edn. Science Press, Beijing, China, pp 203–207
Takase T, Nakazawa M, Ishikawa A, Kawashima M, Ichikawa T, Takahashi N, Shimada H, Manabe K, Matsui M (2004) ydk1-D, an auxin-responsive GH3 mutant that is involved in hypocotyl and root elongation. Plant J 37(4):471–483. https://doi.org/10.1046/j.1365-313X.2003.01973.x
Tang Q, Zhang C (2013) Data processing system (DPS) software with experimental design, statistical analysis and data mining developed for use in entomological research. Insect Sci 20(2):254–260
Wang G, Fang H (2002) Plant genetic engineering principles and techniques, 2nd edn. Science Press, Beijing, China, p 313
Yang G, Wei R, Wang S, Li Z, Jiang J (2011) Rooting culture without agar and efficient transplanting technique of transgenic plantlet in birch. Practical For Technol 4:33–34. https://doi.org/10.13456/j.cnki.lykt.2011.04.019 (in Chinese)
Yang G, Chen S, Jiang J (2015) Transcriptome analysis reveals the role of BpGH3.5 in root elongation of Betula platyphylla × B. pendula. Plant Cell Tissue Organ Culture 121(3):605–617. https://doi.org/10.1007/s11240-015-0731-5
Yang G, Chen S, Wang S, Liu G, Li H, Huang H, Jiang J (2015) BpGH3.5, an early auxin-response gene, regulates root elongation in Betula platyphylla × Betula pendula. Plant Cell Tissue Organ Culture 120(1):239–250. https://doi.org/10.1007/s11240-014-0599-9
Yu J, Wang C, Gong X, Li J, Jiang J, Liu G (2019) Selection of elite transgenic lines of BpGH3.5 in Betula platyphylla. J Beijing For Univ 41(09):81–89. https://doi.org/10.13332/j.1000-1522.20180434 (in Chinese)
Zhan YG, Wang YC, Wang ZY, Yang CP, Liu ZH, Li CH (2003) Genetic transformation of Betula platyphylla and insect resistance of the transgenic plants. J Plant Physiol Mol Biol 29(5):380–386. https://doi.org/10.1023/A:1022289509702
Zhang S, Li C, Cao J, Zhang Y, Zhang S, Xia Y, Sun D, Sun Y (2009) Altered architecture and enhanced drought tolerance in rice via the down-regulation of indole-3-acetic acid by TLD1/OsGH3.13 activation. Plant Physiol 151(4):1889–1901. https://doi.org/10.1104/pp.109.146803
Zhang W, Wei R, Chen S, Jiang J, Li H, Huang H, Yang G, Wang S, Wei H, Liu G (2014) Functional characterization of Cinnamoyl-CoA Reductase (CCR) in birch (Betula platyphylla × Betula pendula) through overexpression and suppression analysis. Physiol Plant 154(2):283–296. https://doi.org/10.1111/ppl.12306
Acknowledgements
The authors thank the staff and postgraduate students at State Key Laboratory of Tree Genetics and Breeding (Northeast Forestry University) for their assistance in carrying out the study (with special thanks to Prof. Xiyang Zhao for his guidance). The authors also gratefully acknowledge the support of staff at the Jilin Provincial Academy of Forestry Sciences and Liaoning Poplar Research Institute.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Project funding: This study was supported by National Key R&D Program of China during the 14th Five-year Plan Period (2021YFD2200102), and Heilongjiang Touyan Innovation Team Program (Tree Genetics and Breeding Innovation Team).
The online version is available at http://www.springerlink.com
Corresponding editor: Yu Lei.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, C., Wang, Y., Zou, J. et al. Selection of elite lines of BpGH3.5-transgenic Betula platyphylla using growth adaptability analysis. J. For. Res. 33, 1891–1901 (2022). https://doi.org/10.1007/s11676-021-01451-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11676-021-01451-4