Abstract
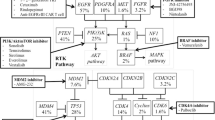
This review evaluates the role of Glivec in the treatment of chronic myelogenous leukemia and other malignant tumors. Preclinical and clinical evidence showed that Glivec demonstrated a potent and specific inhibition on BCR-ABL positive leukemias and other malignant tumors in which overexpression of c-kit and PDGFR-β played a major role in their pathogenesis. Glivec has induced complete hematologic responses in up to 98% of patients evaluated in clinical trials. It’s a very successful drug that supported the idea of targeted therapy through inhibition of tyrosine kinases. Although it’s still in the early stages of clinical development and the resistance to Glivec remains to be a problem needed further study, a great deal has been learned from these research and observation. And with the increasing data, molecular targeting therapy will play much more important role in the treatment of malignant tumors. With the better understanding of the pathogenesis of malignant tumors, well-designed drugs targeting the specific molecular abnormalities with higher efficacy and lower side effect will benefit numerous patients with malignant tumors.
Similar content being viewed by others
References
Carlo-Stella C, Regazzi E, Sammarelli G, et al. Effects of the tyrosine kinase inhibitor AG957 and an Anti-Fas receptor antibody on CD34(+) chronic myelogenous leukemia progenitor cells [J]. Blood 1999; 93:3973.
Sun XM, Layton JE, Elefanty A, et al. Comparison of effects of the tyrosine kinase inhibitors AG957, AG490, and STI571 on BCR-ABL-expressing cells, demonstrating synergy between AG490 and STI571 [J]. Blood 2001; 97:2008.
Druker BJ, Tamura S, Buchdunger E, et al. Effects of a selective inhibitor of the Abl tyrosine kinase on the growth of Bcr-Abl positive cells [J]. Nat Med 1996; 2:561.
Carroll M, Ohno-Jones S, Tamura S, et al. CGP 57148, a tyrosine kinase inhibitor, inhibits the growth of cells expressing BCR-ABL, TEL-ABL, and TEL-PDGFR fusion proteins [J]. Blood 1997; 90:4947.
Beran M, Cao X, Estrov Z, et al. Selective inhibition of cell proliferation and BCR-ABL phosphorylation in acute lymphoblastic leukemia cells expressing Mr 190,000 BCR-ABL protein by a tyrosine kinase inhibitor (CGP-57148) [J]. Clin Cancer Res 1998; 4:1661.
Gambacorti-Passerini C, Barni R, le Coutre P, et al. Role of alphal acid glycoprotein in the in vivo resistance of human BCR-ABL(+) leukemic cells to the abl inhibitor ST1571 [J], J Natl Cancer Inst 2000; 92:1641.
Deininger MW, Goldman JM, Lydon N, et al. The tyrosine kinase inhibitor CGP57148B selectively inhibits the growth of BCR-ABL-positive cells [J]. Blood 1997; 90:3691.
Le Coutre P, Mologni L, Cleris L, et al. In vivo eradication of human BCR/ABL-positive leukemia cells with an ABL kinase inhibitor [J]. J Natl Cancer Inst 1999; 91:163.
Druker B, Talpaz M, Resta DJ, et al. Efficacy and safety of the BCR-ABL tyrosine kinase in chronic myeloid leukemia [J]. N Engl J Med 2001; 344:1031.
Druker BJ, Sawyers CL, Kantarjian H, et al. Activity of a specific inhibitor of the Bcr-Abl tyrosine kinase in the blast crisis of chronic myeloid leukemia and acute lymphoblastic leukemia with the Philadelphia chromosome [J]. N Engl J Med 2001; 344:1038.
Kantarjian H, Sawyers C, Hochhaus A, et al. Hematologic and cytogenetic responses to imatinib mesylate in chronic myelogenous leukemia [J]. New Engl J Med 2002; 346:645.
Talpaz M, Silver RT, Druker BJ, et al. Imatinib induces durable hematologic and cytogenetic responses in patients with accelerated phase chronic myeloid leukemia: results of a phase 2 study [J]. Blood 2002; 99:1928.
Sawyers CL, Hochhaus A, Feldman E, et al. Imatinib induces hematologic and cytogenetic responses in patients with chronic myelogenous leukemia in myeloid blast crisis: results of a phase II study [J]. Blood 2002; 99:3530.
Kantarjian HM, O’Brien S, Cortes JE, et al. Imatinib mesylate therapy for relapse after allogeneic stem cell transplantation for chronic myelogenous leukemia [J]. Blood 2002; 100:1590.
Topaly J, Zeller WJ, Fruehauf S. Combination therapy with imatinib mesylate (ST1571): synopsis of in vitro studies [J]. Br J Haematol 2002; 119:3.
Weisberg E, Griffin JD. Mechanism of resistance to the ABL tyrosine kinase inhibitor ST1571 in BCR/ABL-transformed hematopoietic cell lines [J]. Blood 2000; 95:3498.
Mahon FX, Deininger MW, Schultheis B, et al. Selection and characterization of BCR-ABL positive cell lines with differential sensitivity to the tyrosine kinase inhibitor ST1571: diverse mechanisms of resistance [J]. Blood 2000; 96:1070.
Le Coutre P, Tassi E, Varella-Garcia M, et al. Induction of resistance to the Abelson inhibitor ST1571 in human leukemic cells through gene amplification [J]. Blood 2000; 95:1758.
Gorre ME, Mohammed M, Ellwood K, et al. Clinical resistance to STI-571 cancer therapy caused by BCR-ABL gene mutation or amplification [J]. Science 2001; 293:876.
Hirota S, Isozaki K, Moriyanma Y, et al. Gain-of-function mutations of c-kit in human gastrointestinal stromal tumors [J]. Science 1998; 279:577.
Rubin BP, Singer S, Tsao C, et al. KIT activation is a ubiquitous feature of gastrointestinal stromal tumors [J]. Cancer Res 2001; 61:8118.
Heinrich MC, Griffith DJ, Druker BJ, et al. Inhibition of c-kit receptor tyrosine kinase activity by STI 571, a selective tyrosine kinase inhibitor [J]. Blood 2000; 96:925.
Tuveson DA, Willis NA, Jacks T, et al. STI571 inactivation of the gastrointestinal stromal tumor c-KIT oncoprotein: biological and clinical implications [J]. Oncogene 2001; 20:5054.
Van Oosterom AT, Judson I, Verweij J, et al. Safety and efficacy of imatinib (STI571) in metastatic gastrointestinal stromal tumors: a phase I study [J]. Lancet 2001; 358:1421.
Heinrich MC, Blanke CD, Druker BJ, et al. Inhibition of KIT tyrosine kinase activity: a novel molecular approach to the treatment of KIT-positive malignancies [J]. J Clin Oncol 2002; 20:1692.
Scappini B, Onida F, Kantarjian HM, et al. Effects of signal transduction inhibitor 571 in acute myelogenous leukemia cells [J]. Clin Cancer Res 2001; 7:3884.
Sjoblom T, Boureux A, Ronnstrand L, et al. Characterization of the chronic myelomonocytic leukemia associated TEL-PDGF beta R fusion protein [J]. Oncogene 1999; 18:7055.
Kilic T, Alberta JA, Zdunek PR, et al. Intracranial inhibition of platelet-derived growth factor-mediated glioblastoma cell growth by an orally active kinase inhibitor of the 2-phenylaminopyrimidine class [J]. Cancer Res 2000; 60:5143.
Tomasson MH, Williams IR, Hasserjian R, et al. TEL/PDGFbetaR induces hematologic malignancies in mice that respond to a specific tyrosine kinase inhibitor [J]. Blood 1999; 93:1707.
Apperley JF, Gardembas M, Melo JV, et al. Response to imatinib mesylate in patients with chronic myeloproliferative diseases with rearrangements of the platelet-derived growth factor receptor beta [J]. N Engl J Med 2002; 347:481.
Pietras K, Ostman A, Sjoquist M, et al. Inhibition of platelet-derived growth factor receptors reduces interstitial hypertension and increases transcapillary transport in tumors [J]. Cancer Res 2001; 61:2929.
Author information
Authors and Affiliations
Corresponding author
Additional information
Foundation item: This work was supported by a grant from Nanjing Science & Technology Foundation.
Biography: SUN Xue-mei (1964-), female, associate professor, master of medicine, Nanjing Drum Tower Hospital, Nanjing University Medical College, majors in hematology, and as a scholar worked at Ludwig Institute for Cancer Research in Australia in 1999–2000.
Rights and permissions
About this article
Cite this article
Sun, Xm., Brady, B. New drug targeting treatment — Glivec. Chin J Cancer Res 15, 235–239 (2003). https://doi.org/10.1007/s11670-003-0034-y
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11670-003-0034-y