Abstract
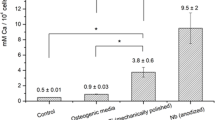
The cold plasma sterilization is an alternative sterilization process that does not modify the morphological properties of nanostructured surfaces of titanium and its alloys. This study aims to evidence the effect of surface morphology of the cold-plasma-sterilized nanostructured Ti6Al4V on the osteogenic differentiation of bone marrow-derived mesenchymal stem cells (BM-MSCs). The surface treatments on Ti6Al4V used were sanding and electropolishing in a H2SO4/HF/glycerin solution. The samples were characterized by AFM, optical interferometry, and wettability. BM-MSCs were cultured for 14 days and tested for cell adhesion, metabolic activity, ALP activity, and mineralization. Results demonstrated that the nanostructured morphology of Ti6Al4V remained intact after the sterilization and promoted a more hydrophilic surface, which contributed to the increase in the metabolic activity, and to osteogenesis of BM-MSCs, as well as to the extracellular matrix mineralization. The bacteriological and mycological tests showed that bacteria, fungi, and yeasts were not detected after cold plasma sterilization.
Graphical abstract






Similar content being viewed by others
References
X.L. Liu, W.R. Zhou, Y.H. Wu, Y. Cheng and Y.F. Zheng, Effect of Sterilization Process on Surface Characteristics and Biocompatibility of Pure Mg and MgCa Alloys, Mat. Sci. Eng. C, 2013, 33, p 4144–4154. https://doi.org/10.1016/j.msec.2013.06.004
Y. Zhao, B. Zhu, Y. Wang, C. Liu and C. Shen, Effect of Different Sterilization Methods on the Properties of Commercial Biodegradable Polyesters for Single-Use, Disposable Medical Devices, Mat. Sci. Eng. C, 2019, 105, p 110041–110049. https://doi.org/10.1016/j.msec.2019.110041
S.A. Smith, B. Gause, D. Plumley and M.J. Drexel, Irradiation-Assisted Stress-Corrosion Cracking of Nitinol During eBeam Sterilization, J. Mater. Eng. Perform., 2012, 21, p 2638–2642. https://doi.org/10.1007/s11665-012-0396-8
L.M. Antonini, C.F. Malfatti, G.C. Reilly, R. Owen and A.S. Takimi, Effect of Sterilization Processes on Nanostructured Ti6Al4V Surfaces Obtained by Electropolishing, J. Mater. Res., 2019, 34(8), p 1439–1446. https://doi.org/10.1557/jmr.2019.28
M.M. Rodrigues, C.P. Fontoura, C.S.C. Garcia, S.T. Martins, J.A.P. Henriques, C.A. Figueroa, M.R. Ely and C. Aguzzoli, Investigation of Plasma Treatment on UHMWPE Surfaces: Impact on Physicochemical Properties, Sterilization and Fibroblastic Adhesion, Mat. Sci. Eng. C, 2009, 102, p 264–275. https://doi.org/10.1016/j.msec.2019.04.048
I. Junkar, M. Kulkarni, B. Drasler, N. Rugelj, A. Mazare, A. Flasker, D. Drobne, P. Humpolícek, M. Resnik, P. Schmuki, M. Mozetic and A. Iglic, Influence of Various Sterilization Procedures on TiO2 Nanotubes used for Biomedical Devices, Bioelectrochemistry, 2016, 109, p 79–86. https://doi.org/10.1016/j.bioelechem.2016.02.001
M. Ueno, W.M.I. Urruchi, A.O.C. Jorge, C. Otani and H.S. Maciel, Esterilização de Limas Endodônticas com Plasma de Oxigênio, Pesqui. Odontol. Bras., 2000, 14(3), p 205–208. https://doi.org/10.1590/S1517-74912000000300003
K. Lee, K. Paek, W. Tae and Y. Lee, Sterilization of Bacteria, Yeast, and Bacterial Endospores by Atmospheric-Pressure Cold Plasma using Helium and Oxygen, J. Microbiol., 2006, 44(3), p 269–275.
Q.S. Yu, C. Huang, F.H. Hsieh, H. Huff and Y. Duan, Sterilization Effects of Atmospheric Cold Plasma Brush, Appl. Phys. Lett., 2006, 88(3), p 013903-1-013903–3. https://doi.org/10.1063/1.2161807
L.M. Antonini, V. Kothe, G.C. Reilly, R. Owen, J.S. Marcuzzo and C.F. Malfatti, Effect of Ti6Al4V Surface Morphology on the Osteogenic Differentiation of Human Embryonic Stem Cells, J. Mater. Res., 2017, 32(20), p 3811–3821. https://doi.org/10.1557/jmr.2017.392
A. Tavangar, B. Tan and K. Venkatakrishnan, Synthesis of Bio-Functionalized Three-Dimensional Titania Nanofibrous Structures Using Femtosecond Laser Ablation, Acta Biomater., 2011, 7(6), p 2726–2732. https://doi.org/10.1016/j.actbio.2011.02.020
B.-S. Moon, S. Kim, H.-E. Kim and T.-S. Jang, Hierarchical Micro-Nano Structured Ti6Al4V Surface Topography via Two-Step Etching Process for Enhanced Hydrophilicity and Osteoblastic Responses, Mat. Sci. Eng. C, 2017, 73, p 90–98. https://doi.org/10.1016/j.msec.2016.12.064
X. Liu and S. Wang, Three-Dimensional Nano-Biointerface as a New Platform for Guiding Cell Fate, Chem. Soc. Rev., 2014, 43(8), p 2385–2401. https://doi.org/10.1039/C3CS60419E
E. Filova, J. Fojt, M. Kryslova, H. Moravec, L. Joska and L. Bacakova, The Diameter of Nanotubes Formed on Ti-6Al-4V Alloy Controls the Adhesion and Differentiation of Saos-2 Cells, Int. J. Nanomed., 2015, 10, p 7145–7163. https://doi.org/10.2147/IJN.S87474
J. Park, S. Bauer, A. Pittrof, M.S. Killian, P. Schmuki and K. von der Mark, Synergistic Control of Mesenchymal Stem Cell Differentiation by Nanoscale Surface Geometry and Immobilized Growth Factors on TiO2 Nanotubes, Small, 2012, 8(1), p 98–107. https://doi.org/10.1002/smll.201100790
J. Qiu, J. Li, S. Wang, B. Ma, S. Zhang, W. Guo, X. Zhang, W. Tang, Y. Sang and H. Liu, TiO2 Nanorod Array Constructed Nanotopography for Regulation of Mesenchymal Stem Cells Fate and the Realization of Location-Committed Stem Cell Differentiation, Small, 2016, 12(13), p 1770–1778. https://doi.org/10.1002/smll.201503946
Y. Kutes, V. Vyas and B.D. Huey, Nano and Micro Scale Analysis of Dentin with in vitro and High Speed Atomic Force Microscopy, J. Mater. Res., 2013, 28(17), p 2300–2307. https://doi.org/10.1557/jmr.2013.159
M. Pidwirny, Atmospheric composition: fundamentals of physical geography, 2nd ed., National Council of Educational Research and Training, 2006 (New Delhi).
R.K.A. Feltham and G.I. Barrow, Cowan and Steel’s Manual for the Identification of Medical Bacteria, 2nd ed. Cambridge University Press, Cambridge, 1993.
C.D. Hoemann, H. El-Gabalawy and M.D. McKee, In vitro Osteogenesis Assays: Influence of the Primary Cell Source on Alcaline Phosphatase Activity and Mineralization, Pathol. Biol., 2009, 57(4), p 318–323. https://doi.org/10.1016/j.patbio.2008.06.004
J.C. Keller, G.B. Schneider, C.M. Stanford and B. Kellogg, Effects of Implant Microtopography on Osteoblast Cell Attachment, Implant Dent., 2003, 12(2), p 175–181. https://doi.org/10.1097/01.ID.0000058309.77613.87
X. Zhu, J. Chen, L. Scheideler, R. Reichl and J. Geis-Gerstorfer, Effects of Topography and Composition of Titanium Surface Oxides on Osteoblast Respondes, Biomaterials, 2004, 25, p 4087–4103. https://doi.org/10.1016/j.biomaterials.2003.11.011
X. Zhu, J. Chen, L. Scheideler, T. Altebaeumer, J. Geis-Gerstorfer and D. Kem, Cellular Reactions of Osteoblasts to Micron- and Submicron-Scale Porous Structures of Titanium Surfaces, Cells Tissues Organs, 2004, 178(1), p 13–22. https://doi.org/10.1159/000081089
L. Bacakova, E. Filova, M. Pariek, T. Ruml and V. Svorcik, Modulation of Cell Adhesion, Proliferation and Differentiation on Materials Designed for Body Implants, Biotechnol. Adv., 2011, 29(6), p 739–767. https://doi.org/10.1016/j.biotechadv.2011.06.004
H.F. Li, Y.B. Wang, Y.F. Zheng and J.P. Lin, Osteoblast Response on Ti- and Zr-based Bulk Metallic Glass Surfaces after Sand Blasting Modification, J. Biomed. Mater. Res. B., 2012, 100(7), p 1721–1728. https://doi.org/10.1002/jbm.b.32738
Z. Deng, B. Yin, W. Li, J. Liu, J. Yang, T. Zheng, D. Zhang, H. Yu, X. Liu and J. Ma, Surface Characteristics of and in vitro Behavior of Osteoblast-Like Cells on Titanium with Nanotopography Prepared by High-Energy Shot Peening, Int. J. Nanomedicine, 2014, 9(1), p 5565–5573. https://doi.org/10.2147/IJN.S71625
F. Zhou, L. Yuan, H. Huang and H. Chen, Phenomenon of “Contact Guidance” on the Surface with Nano-Micro-Groove-Like Pattern and Cell Physiological Effects, Chinese Sci. Bull., 2009, 54, p 3200–3205. https://doi.org/10.1007/s11434-009-0366-1
S. Minagar, J. Wang, C.C. Berndt, E.P. Ivanova and C. Wen, Cell Response of Anodized Nanotubes on Titanium and Titanium Alloys, J. Biomed. Mater. Res. A, 2013, 101(9), p 2726–2739. https://doi.org/10.1002/jbm.a.34575
W.E. Yang and H.H. Huang, Improving the Biocompatibility of Titanium Surface Through Formation of a TiO2 Nano-mesh Layer, Thin Solid Films, 2010, 518(24), p 7545–7550. https://doi.org/10.1016/j.tsf.2010.05.045
M.J. Dalby, M.O. Riehle, D.S. Sutherland, H. Agheli and A.S.G. Curtis, Changes in Fibroblast Morphology in Response to Nano-Columns Produced by Colloidal Lithography, Biomaterials, 2004, 25(23), p 5415–5422. https://doi.org/10.1016/j.biomaterials.2003.12.049
R. Olivares-Navarrete, S.L. Hyzy, M.E. Berg, J.M. Schneider, K. Hotchkiss, Z. Schwartz and B.D. Boyan, Osteoblast Lineage Cells can Discriminate Microscale Topographic Features on Titanium-Aluminum-Vanadium Surfaces, Ann. Biomed. Eng., 2014, 42(12), p 2551–2561. https://doi.org/10.1007/s10439-014-1108-3
R.A. Gittens, R. Olivares-Navarrete, T. McLachlan, Y. Cai, S.L. Hyzy, J.M. Schneides, Z. Schwartz, K.H. Sandhage and B.D. Boyan, Differential Responses of Osteoblast Lineage Cells to Nanotopographically-Modified, Microroughened Titanium–Aluminum–Vanadium Alloy Surfaces, Biomaterials, 2012, 33(35), p 8986–8994. https://doi.org/10.1016/j.biomaterials.2012.08.059
L.M. Antonini, A.G.S. Junior, G. Reilly and C.F. Malfatti, Human Embryonic Stem Cell-Derived Mesenchymal Progenitor (hESCs-MP) Growth on Nanostructured Ti6Al4V Surfaces, Mater. Res., 2018, 21(5), p 1–10. https://doi.org/10.1590/1980-5373-mr-2017-1081
L. Bárdos and H. Baránková, Cold Atmospheric Plasma: Sources, Processes, and Applications, Thin Solid Films, 2010, 518, p 6705–6713. https://doi.org/10.1016/j.tsf.2010.07.044
A. Cunha, A.P. Serro, V. Oliveira, A. Almeida, R. Vilar and M.-C. Durrieu, Wetting Behaviour of Femtosecond Laser Textured Ti-6Al-4V Surfaces, Appl. Surf. Sci., 2013, 265, p 688–696. https://doi.org/10.1016/j.apsusc.2012.11.085
D.-Z. Cui, K.-D. Park, K.-K. Lee, Y.-S. Jung, B.-A. Lee, Y.-J. Lee, O.-S. Kim, H. Chung and Y.-J. Kim, Surface Characteristics and Osteoblastic Cell Response to Titanium–8tantalum–3neobium Alloy, Appl. Surf. Sci., 2012, 262, p 107–109. https://doi.org/10.1016/j.apsusc.2012.02.112
M.L. Wang, R. Tuli, P.A. Manner, P.F. Sharkey, D.J. Hall and R.S. Tuan, Direct and Indirect Induction of Apoptosis in Human Mesenchymal Stem Cells in Response to Titanium Particles, J. Orthop. Res., 2003, 21(4), p 697–707. https://doi.org/10.1016/S0736-0266(02)00241-3
M.L. Wang, L.J. Nesti, R. Tuli, J. Lazatin, K.G. Danielson and P.F. Sharkey, Titanium Particles Suppress Expression of Osteoblastic Phenotype in Human Mesenchymal Stem Cells, J. Orthop. Res., 2002, 20(6), p 1175–1184. https://doi.org/10.1016/S0736-0266(02)00076-1
E.V. Ortega, A. Jos, A.M. Cameán, J.P. Mourelo and J.J.S. Egea, In vitro Evaluation of Cytotoxicity and Genotoxicity of a Commercial Titanium Alloy for Dental Implantology, Mutat. Res., 2010, 702(1), p 17–23. https://doi.org/10.1016/j.mrgentox.2010.06.013
T. Rae, The Toxicity of Metals used in Orthopaedic Prostheses. An Experimental Study Using Cultured Human Synovial Fibroblasts, J. Bone Joint Surg. Br., 1981, 63-B(3), p 435–440. https://doi.org/10.1302/0301-620X.63B3.7263760
M. Esposito, J.M. Hirsch, U. Lekholm and P. Thomsen, Biological Factors Contributing to Failures of Osseointegrated Oral Implants (II) Etiopathogenesis, Eur. J. Oral Sci., 1998, 106(3), p 721–764. https://doi.org/10.1046/j.0909-8836.t01-6-.x
Y. Okazaki, S. Rao, I. Yoshumasa and T. Tateishi, Corrosion Resistance, Mechanical Properties, Corrosion Fatigue Strenght and Cytocompatibility of New Ti Alloys Without Al and V, Biomaterials, 1998, 19(13), p 1197–1215. https://doi.org/10.1016/S0142-9612(97)00235-4
H.-H. Huang, C.-P. Wu, Y.-S. Sun and T.-H. Lee, Improvements in the Corrosion Resistance and Biocompatibility of Biomedical Ti-6Al-7Nb Alloy using an Electrochemical Anodization Treatment, Thin Solid Films, 2013, 528, p 157–162. https://doi.org/10.1016/j.tsf.2012.08.063
S. Sista, A. Nouri, Y. Li, C. Wen, P.D. Hodgson and G. Pande, Cell Biological Responses of Osteoblasts on Anodized Nanotubular Surface of a Titanium-Zirconium Alloy, J. Biomed. Mater. Res. A, 2013, 101(12), p 3416–3430. https://doi.org/10.1002/jbm.a.34638
E.B. Partida, A.M. Ulloa, B.V. Salas, C. Velasquillo, M. Carrillo, A. Escamilla, E. Valdez and F. Villarreal, Improved Osteoblast and Chondrocyte Adhesion and Viability by Surface-Modified Ti6Al4V Alloy with Anodized TiO2 Nanotubes Using a Super-Oxidative Solution, Materials, 2015, 8(3), p 867–883. https://doi.org/10.3390/ma8030867
M.S. Stan, I. Memet, C. Fratila, E. Krasicka-Cydzik, I. Roman and A. Dinischiotu, Effects of Titanium-Based Nanotube Films on Osteoblast Behavior in vitro, J. Biomed. Mater. Res. A, 2015, 103(1), p 48–56. https://doi.org/10.1002/jbm.a.35148
I. Han, B. Vagaska, H.J. Seo, J.K. Kang, B.J. Kwon, M.H. Lee and J.C. Park, Promoted Cell and Material Interaction on Atmospheric Pressure Plasma Treated Titanium, Appl. Surf. Sci., 2012, 258, p 4718–4723. https://doi.org/10.1016/j.apsusc.2012.01.065
D.F. Williams, E.J.C. Kellar, D.A. Jesson and J.F. Watts, Surface Analysis of 316 Stainless Steel Treated with Cold Atmospheric Plasma, Appl. Surf. Sci., 2017, 403, p 240–247. https://doi.org/10.1016/j.apsusc.2017.01.150
A.J. Moreira, R.D. Mansano, T.J.A. Pinto, R. Ruas, L.S. Zambon, M.V. Silva and P.B. Verdonck, Sterilization by Oxygen Plasma, Appl. Surf. Sci., 2004, 235, p 151–155. https://doi.org/10.1016/j.apsusc.2004.05.128
H. Wang, B. Zhao, C. Liu, C. Wang, X. Tan and M.A. Hu, A Comparasion of Biocompatibility of a Titanium Alloy Fabricated by Electron Beam Melting and Selective Laser Melting, PLoS ONE, 2016, 11(7), p 1–19. https://doi.org/10.1371/journal.pone.0158513
T. Han, B. Chang, X. Ding, G. Yue, W. Song, H. Tang, L. Jia, L. Zhao and Y. Zhang, Improved Bone Formation and Ingrowth for the Additively Manufactured Porous Ti6Al4V Bone Implants with Strontium Laden Nanotube Array Coating, RSC Adv., 2016, 6(17), p 13686–13697. https://doi.org/10.1039/C5RA20370H
G.S. Stein, J.B. Lian, A.J. Van Wijnen, J.L. Stein, M. Montecino, A. Javed, S.K. Zaidi, D.W. Young, J. Choi and S.M. Pockwinse, Runx2 Control of Organization, Assembly and Activity of the Regulatory Machinery for Skeletal Gene Expression, Oncogene, 2004, 23(24), p 4315–4329. https://doi.org/10.1038/sj.onc.1207676
M. Mozetic, A. Vesel, G. Primc, C.E. Sittner, J. Bauer, A. Eder, G.H.S. Schmid, D.N. Ruzic, Z. Ahmed, D. Barker, K.O. Douglass, S. Eckel, J.A. Fedchak, J. Hendricks, N. Klimov, J. Ricker, J. Scherschligt, J. Stone, G. Strouse, I. Capan, M. Buljan, S. Milosevic, C. Teichnert, S.R. Cohen, A.G. Silva, M. Lehocky, P. Humpolicek, C. Rodrigues, J.H. Montelongo, D. Mercier, M.M. Silvan, G. Geccone, A. Galtayries, K.S. Kleinschek, I. Petrov, J.E. Greene, J. Avila, C.Y. Chen, B.C. Munoz, H. Yi, A. Boury, S. Lorcy, M.C. Asensio, J. Bredin, T. Gans, D.O. Connell, J. Brendin, F. Reniers, A. Vincze, M. Anderle and L. Montelius, Recent Developments in Surface Science and Engineering, Thin Films, Nanoscience, Biomaterials, Plasma Science, and Vacuum Technology, Thin Solid Films, 2018, 660, p 120–160. https://doi.org/10.1016/j.tsf.2018.05.046
K.S. Brammer, S. Oh, C.J. Cobb, L.M. Bjursten, H. Van De Heyde and S. Jin, Improved Bone-Forming Functionality on Diameter-Controlled TiO2 Nanotube Surface, Acta Biomater., 2009, 5(8), p 3215–3223. https://doi.org/10.1016/j.actbio.2009.05.008
B.D. Boyan, R. Batzer, K. Kieswetter, Y. Liu, D.L. Cochran, S. Szmuckler-Monclers, D.D. Dean and Z. Schwartz, Titanium Surface Roughness Alters Responsiveness of MG63 Osteoblastic-like Cells to 1alpha,25-(OH)2D3, J. Biomed. Mater. Res., 1998, 39(1), p 77–85. https://doi.org/10.1002/(sici)1097-4636(199801)39:1%3c77::aid-jbm10%3e3.0.co;2-l
B.D. Boyan, S. Lossdorfer, L. Wang, G. Zhao, C.H. Lohmann, D.L. Cochran and Z. Schwartz, Osteoblasts Generate an Osteogenic Microenvironment when Grown on Surfaces with Rough Microtopographies, Eur. Cell Mater., 2003, 6(22), p 22–27. https://doi.org/10.22203/ecm.v006a03
Acknowledgments
The present work was carried out with the support of CAPES (Brazilian Coordination for the Improvement of Higher Education Personnel) (CAPES - PROEX Process 23038.000341/2019-71), CNPq (National Council for Scientific and Technological Development) [Grants No. 408366/2018-4] and FAPERGS [Grants Nos. 19/2551-0002280-8 and 19/2551-0000699-3]. Célia de Fraga Malfatti acknowledges CNPq (Grant 307723/ 2018-6). L.M. Antonini thanks for the postdoctorate scholarship CAPES PNPD (Grant PNPD20132547). The authors would like to thank Jane Brazil (from Universidade Luterana do Brasil - ULBRA, Brazil), for her technical services related to cell culture. INCT-Regenera. All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest or non-financial interest in the subject matter or materials discussed in this manuscript.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
LMA contributed to investigation, writing, methodology—original draft. VPA contributed to methodology, investigation, writing—review. MC contributed to writing—review. AST contributed to conceptualization, formal analysis. CFM contributed to conceptualization, supervision, formal analysis.
Corresponding author
Ethics declarations
Conflict of interest
The authors indicated no potential conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Antonini, L.M., Takimi, A.S., Amaral, V.P. et al. Cold-Plasma-Sterilized Nanostructured Ti6Al4V: Effect on Nanostructured Surface Morphology and Osteogenic Differentiation of Bone-Marrow-Derived Mesenchymal Stem Cells. J. of Materi Eng and Perform 30, 7236–7246 (2021). https://doi.org/10.1007/s11665-021-05903-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11665-021-05903-0