Abstract
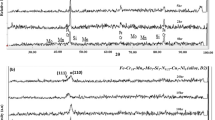
The corrosion behavior and characteristics of 3Cr steel in coexisting H2S- and CO2-containing solution at temperature from 25 to 65 °C were investigated by potentiodynamic polarization sweep, electrochemical impedance spectroscopy (EIS) and scanning electron microscopy (SEM) with energy-dispersive x-ray (EDX). The EIS data fitted by ZsimpWin software were analyzed and discussed. The experimental results indicated that corrosion potential (Ecorr) became more negative and that corrosion current density (icorr) decreased as the Na2S·9H2O concentration (i.e., H2S/HS−) increased from 0 to 1.6%, with the obvious passivation characteristics occurring in anodic polarization branch curve in 1.6% Na2S·9H2O case. Warburg impedance characteristics of diffusion process in low-frequency region following a depressed capacitive semicircle in the intermediate frequency region were observed with the addition of certain Na2S·9H2O concentration or temperature, with the phase angle peaks for H2S-containing cases in the intermediate frequency region shifting to low-frequency direction and raising toward −90° significantly. Severe corrosion pits or cavities or river-liking platform could be observed after removing the corrosion products. The contents of S element in the “first deposition” black corrosion product layer were higher than those in the “second deposition” gray corrosion product crystals. All result displayed the inhibition effect with the addition of Na2S·9H2O. In addition, one four-stage corrosion model involving H2S and CO2 corrosion was proposed.















Similar content being viewed by others
References
F.X. Shi, L. Zhang, J.W. Yang, M.X. Lu, J.H. Ding, and H. Li, Polymorphous FeS Corrosion Products of Pipeline Steel under Highly Sour Conditions, Corros. Sci., 2016, 102, p 103–113
Q. Liu, Z. Li, Z.Y. Liu, X.G. Li, and S.Q. Wang, Effects of H2S/HS-, on Stress Corrosion Cracking Behavior of X100 Pipeline Steel Under Simulated Sulfate-Reducing Bacteria Metabolite Conditions, J. Mater. Eng. Perform., 2017, 26, p 2763–2775
J. Bana’s, U. Lelek-Borkowska, B. Mazurkiewicz, and W. Solarski, Effect of CO2 and H2S on the Composition and Stability of Passive Film on Iron Alloys in Geothermal Water, Electrochim. Acta, 2007, 52, p 5704–5714
P.P. Bai, S.Q. Zheng, and C.F. Chen, Electrochemical Characteristics of the Early Corrosion Stages of API, X52 Steel Exposed to H2S Environments, Mater. Chem. Phys., 2015, 295, p 149–150
T.L. Zhao, Z.Y. Liu, S.S. Hu, C.W. Du, and X.G. Li, Effect of Hydrogen Charging on the Stress Corrosion Behavior of 2205 Duplex Stainless Steel Under 35% NaCl Thin Electrolyte Layer, J. Mater. Eng. Perform., 2017, 26, p 2837–2846
D. Hardie, E.A. Charles, and A.H. Lopez, Hydrogen Embrittlement of High Strength Pipeline Steels, Corros. Sci., 2006, 48, p 4378–4385
P. Roffey and E.H. Davies, The Generation of Corrosion under Insulation and Stress Corrosion Cracking due to Sulphide Stress Cracking in an Austenitic Stainless Steel for Hydrocarbon Gas Pipeline, Eng. Fail. Anal., 2014, 44, p 148
J.B. Sardisco and R.E. Pitts, Corrosion of Iron in an H2S-CO2-H2O System, Corrosion, 1965, 21, p 350–354
D.W. Shoesmith, P. Taylor, M. Grant Bailey, and D.G. Owen, The Formation of Ferrous Monosulfide Polymorphs during the Corrosion of Iron by Aqueous Hydrogen Sulfide at 21°C, J. Electrochem. Soc., 1980, 127, p 1007–1015
X.L. Cheng, H.Y. Ma, J.P. Zhang, X. Chen, S.H. Chen, and H.Q. Yang, Corrosion of Iron in Acid Solution with Hydrogen Sulfide, Corrosion, 1998, 54, p 369–376
H.Y. Ma, X.L. Cheng, S.H. Chen, G.Q. Li, X. Chen, S.B. Lei, and H.Q. Yang, Theoretical Interpretation on Impedance Spectra for Anodic Iron Dissolution in Acidic Solutions Containing Hydrogen Sulfide, Corrosion, 1998, 54, p 634–640
K. Masamura, S. Hashizume, and J. Sakai, Polarization Behavior of High-alloy OCTG in CO2 Environment as Affected by Chlorides and Sulfides, Corrosion, 1987, 6, p 359–368
Z.Y. Liu, X.Z. Wang, R.K. Liu, C.W. Du, and X.G. Li, Electrochemical and Sulfide Stress Corrosion Cracking Behaviors of Tubing Steels in a H2S/CO2 Annular Environment, J. Mater. Eng. Perform., 2014, 23, p 1279–1287
S. Arzola and J. Genesca, The Effect of H2S Concentration on the Corrosion Behavior of API, 5L X-70 steel, J. Solid State Electrochem., 2005, 9, p 197–200
Standard Technical Method NACE TM 0177, Laboratory Testing of Metals for Resistance to Specific Forms of Environmental Cracking in H2S, National Association of Corrosion Engineers, 1996.
J.L. Crolet, N. Thevenot, and S. Nesic, Role of Conductive Corrosion Products in the Protectiveness of Corrosion Layers, Corrosion, 1998, 54, p 194–203
M.M. Singh and A. Gupta, Corrosion Behavior of Mild Steel in Acetic Acid Solutions, Corrosion, 2000, 56, p 371–379
D.D. Macdonald, The Point Defect Model for the Passive state, J. Electrochem. Soc., 1992, 139, p 3434–3449
Y. Choi, S. Nesic, and S. Ling, Effect of H2S on the CO2 Corrosion of Carbon Steel in Acidic Solutions, Electrochim. Acta, 2011, 56, p 1752–1760
M.A. Veloz and I. Gonzalez, Electrochemical Study of Carbon Steel Corrosion in Buffered Acetic Acid Solutions with Chlorides and H2S, Electrochim. Acta, 2002, 48, p 135–144
J. Tang, Y. Shao, J. Guo, T. Zhang, G. Meng, and F. Wang, The Effect of H2S Concentration on the Corrosion Behavior of Carbon Steel at 90°C, Corros. Sci., 2010, 52, p 2050–2058
H.Y. Ma, X.L. Cheng, G.Q. Li, S.H. Chen, Z.L. Quan, S.Y. Zhao, and L. Niu, The Influence of Hydrogen Sulfide on Corrosion of Iron under Different Conditions, Corros. Sci., 2000, 42, p 1669–1682
E. McCafferty, On the Determination of Distributed Double-layer Capacitances from Cole-cole Plots, Corros. Sci., 1997, 39, p 243–254
D.C. Silverman and J.E. Carrico, Electrochemical Impedance Technique—a Practical Tool for Corrosion Prediction, Corrosion, 1988, 44, p 280–287
G. Walter, A Review of Impedance Plot Methods Used for Corrosion Performance Analysis of Painted Metals, Corros. Sci., 1986, 26, p 681–703
Y. Chen and W.P. Jepson, EIS Measurement for Corrosion monitoring under multiphase flow conditions, Electrochim. Acta, 1999, 44, p 4453–4464
C.N. Cao and J.Q. Zhang, An Introduction to Electrochemical Impedance Spectroscopy, Science Press, Beijing, 2002
S. Walkner and A.W. Hassel, Combined Chemical and EIS Study of the Reaction of Zinc Coatings under Alkaline Conditions, Electrochim. Acta, 2014, 131, p 130–136
B. Wang, C. Cui, and S. Zhang, Electrochemical Corrosion Behavior of Fe3Al in H2S-CO2-Cl− Environment, Mater. Chem. Phys., 2017, 198, p 7–15
C.N. Cao, Principles of Electrochemistry of Corrosion, 3rd ed., Chemical Industry Press, Beijing, 2008
E. Barsoukov and J.R. Macdonald, Impedance Spectroscopy Theory, Experiment, and Applications, 2nd ed., Wiley, Hoboken, NJ, 2005
C.F. Chen, M.X. Lu, D.B. Sun, Z.H. Zhang, and W. Chang, Effect of Chromium on the Pitting Resistance of Oil tube Steel in a Carbon Dioxide Corrosion System, Corrosion, 2005, 61, p 594–601
D.S. Carvalho, C.J.B. Joia, and O.R. Mattos, Corrosion Rate of Iron and Iron-chromium Alloys in CO2 Medium, Corros. Sci., 2005, 47, p 2974–2986
G.S. Eklund, Initiation of Pitting at Sulfide Inclusions in Stainless Steel, J. Electrochem. Soc., 1974, 121, p 467–473
S.E. Lott and R.C. Alkire, The Role of Inclusions on Initiation of Crevice Corrosion of Stainless Steel: I. Experimental Studies, J. Electrochem. Soc., 1989, 136, p 973–979
H.H. Huang, W.T. Tsai, and J.T. Lee, Electrochemical Behavior of the Simulated Heat Affected Zone of A516 Carbon Steel in H2S Solution, Electrochim. Acta, 1996, 41, p 1191–1199
H.H. Uhlig and R.W. Revie, Corrosion and Corrosion Control, 3rd ed., Wiley, New York, 1991
Z.F. Yin, W.Z. Zhao, Y.R. Feng, and S.D. Zhu, Characterisation of CO2 Corrosion Scale in Simulated Solution with Cl− Ion under Turbulent Flow Conditions, Corros. Eng. Sci. Technol., 2009, 44, p 453–461
J.O.M. Bockris, D. Drazic, and A.R. Despic, The Electrode Kinetics of the Deposition and Dissolution of Iron, Electrochim. Acta, 1961, 4, p 325–361
S. Nesic, N. Thevenot, J.L. Crolet, and D. Drazic, Electrochemical Properties of Iron Dissolution in the Presence of CO2, paper no. 3, NACE, Houston, TX, 1996
Z.F. Yin, W.Z. Zhao, W.Y. Lai, C.X. Yin, and S.D. Zhu, Film Characteristics of Carbon Steel in Simulant Solution with the Effect of Acetic Acid and CO2, J. Mater. Eng. Perform., 2010, 19, p 693–699
J.L. Crolet, N. Thevenot, and A. Dugstad, Role of Free Acetic Acid on the CO2 Corrosion of Steels, Corrosion/99, paper no. 24, NACE International, Houston, TX, 1999
F.W. Song, A Comprehensive Model for Predicting CO2 Corrosion Rate in Oil and Gas Production and Transportation Systems, Electrochim. Acta, 2010, 55, p 689–700
W. Sun, S. Nesic, and D. Young, Equilibrium Expressions Related to the Solubility of the Sour Corrosion Product Mackinawite, Ind. Eng. Chem. Res., 2008, 47, p 1738–1742
Acknowledgments
The authors kindly thank the support to this work for experimental setup supplied by Shaanxi Yanchang Petroleum (Group) Co. LTD, and financial support from Special Scientific Research Plan of Shaanxi Provincial Education Department (18JS099) and Xi’An Science and Technology Plan Projects (2017CGWL09).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yin, Z.F., Zhang, Y.L., Chang, G.R. et al. Corrosion Behavior and Characteristics of 3Cr Steel in Coexisting H2S- and CO2-Containing Solutions. J. of Materi Eng and Perform 29, 5442–5457 (2020). https://doi.org/10.1007/s11665-020-05023-1
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11665-020-05023-1