Abstract
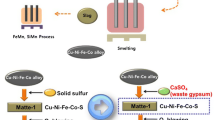
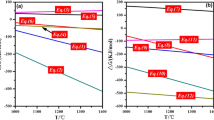
In the present work, a novel method of producing Ni–Fe–S matte from saprolite type laterite (SL) ores was proposed using CaS as the sulfurization agent at 1500 °C under Ar atmosphere. The prepared Ni–Fe–S matte could be used as the raw material for the preparation of metallic nickel. CaS was prepared by reducing CaSO4 with graphite under Ar atmosphere. It was found that as increasing CaS addition, the recovery extents of Ni increases, while the Ni grade of Ni–Fe–S matte decreases. When the CaS addition was 6 times of the stoichiometric quantity required for the sulfurization of NiO, the recovery extents of Ni, Ni grade and CaS utilization degree reach to 93.63 pct, 8.84 wt pct and 92.10 pct, respectively. Compared with the reported method of using S2 as the sulfurization agent, the use of CaS in the present work can reduce the SO2 emission and improve the recovery extent of Ni. Besides, the feasibility of using CaSO4 as the sulfurization agent was also investigated at 1500 °C. Unfortunately, the carbon, which was used to reduce the CaSO4, could also react with NiO and Fe2O3 to produce the Ni–Fe alloy at 1500 °C. Accordingly, CaS is a better sulfurization agent than CaSO4.











Similar content being viewed by others
References
A.D. Dalvi, W.G. Bacon, and R.C. Osborne: in: PDAC 2004 International Convention., 2004, March 7–10, pp. 1–27.
F.K. Crundwell, M.S. Moats, V. Ramachandran, T.G. Robinson, and W.G. Davenport: Extractive Metallurgy of Nickel, Cobalt and Platinum Group Metals, 1st ed. Elsevier, Amsterdam, 2011, pp. 4–9.
F.K. Crundwell, M.S. Moats, V. Ramachandran, T.G. Robinson, and W.G. Davenport: Extractive Metallurgy of Nickel, Cobalt and Platinum Group Metals, 1st ed. Elsevier, Amsterdam, 2011, p. 207.
F.K. Crundwell, M.S. Moats, V. Ramachandran, T.G. Robinson, and W.G. Davenport: Extractive Metallurgy of Nickel, Cobalt and Platinum Group Metals, 1st ed. Elsevier, Amsterdam, 2011, p. 236.
A.E.M. Warner, C.M. Díaz, A.D. Dalvi, P.J. Mackey, and A.V. Tarasov: JOM., 2006, vol. 58(4), pp. 11–20.
F.K. Crundwell, M.S. Moats, V. Ramachandran, T.G. Robinson, and W.G. Davenport: Extractive Metallurgy of Nickel, Cobalt and Platinum Group Metals, 1st ed. Elsevier, Amsterdam, 2011, p. 118.
E.H. Jeong, C.W. Nam, K.H. Park, and J.H. Park: Metall. Mater. Trans. B., 2016, vol. 4B7(2), pp. 1103–12.
C. Doyle: in: The Minerals, Metals amp; Materials Society Annual Meeting and International Laterite Nickel Symposium, 2004, pp. 670–84
C.T. Harris, J.G. Peacey, and C.A. Pickles: Miner. Eng., 2011, vol. 24(7), pp. 651–60.
C.T. Harris, J.G. Peacey, and C.A. Pickles: Miner. Eng., 2013, vol. 54, pp. 21–31.
B.B. Kale, A.R. Pande, and A.N. Gokarn: Metall. Mater. Trans. B., 1992, vol. 23B(5), pp. 567–72.
D. Zheng, H. Lu, X. Sun, X. Liu, W. Han, and L. Wang: Thermochim Acta., 2013, vol. 559, pp. 23–31.
T.D. Wheelock and D.R. Boylan: Ind. Eng. Chem., 1960, vol. 52(3), pp. 215–28.
L.M. Diaz, S.E. Squier, and A.H. Pulsifer: Chem. Eng. Sci., 1985, vol. 40(3), pp. 319–24.
L. Ma, X. Niu, J. Hou, S. Zheng, and W. Xu: Thermochim Acta., 2011, vol. 526(1–2), pp. 163–68.
E.M. Merwe, C.A. Strydom, and J.H. Potgieter: Thermochim. Acta., 1999, vol. 340, pp. 431–37.
T. Kato, K. Murakami, and K. Sugawara: Chem. Eng. Trans., 2012, vol. 29, pp. 805–10.
D.Q. Zhu, Y. Cui, K. Vining, S. Hapugoda, J. Douglas, J. Pan, and G.L. Zheng: Int J Miner Process., 2012, vol. 106–109, pp. 1–7.
YS/T 341.1-2006: Methods for chemical analysis of nickel concentrates-dimethylglyoxime precipitate separation-EDTA titration method (Non-ferrous metal industry standards of the People’s Republic of China) http://www.cnsmq.com/mulu.shtml?bzh1=YS&bzh2=341.1-2006&dosubmit.
H. Su, H.B. Zuo, and J. Zhao: Inorg Chem. Ind., 2019, vol. 51(7), pp. 68–73.
Y. Hara and A. Jha: Energy Technol. 2015, pp. 105–13.
F.K. Crundwell, M.S. Moats, V. Ramachandran, T.G. Robinson, and W.G. Davenport: Extractive Metallurgy of Nickel, Cobalt and Platinum Group Metals, 1st ed. Elsevier, Amsterdam, 2011, p. 260.
S. Wu, Y. Yao, X. Yao, C. Ren, and W. Wang: J. Cleaner Prod., 2020, vol. 265, p. 121801.
Acknowledgments
This work was supported by the National Natural Science Foundation of China (51734002).
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, HY., Hou, Y., Chang, HQ. et al. Preparation of Ni–Fe–S Matte from Nickeliferous Laterite Ore Using CaS as the Sulfurization Agent. Metall Mater Trans B 53, 1136–1147 (2022). https://doi.org/10.1007/s11663-022-02444-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11663-022-02444-2