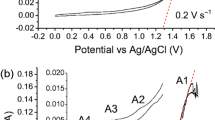
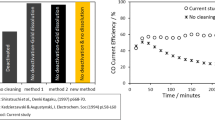
Abstract
Molten salt electrolysis is a clean and low-cost Ti production technology that allows the direct conversion of metal oxides into metals or alloys via electrochemical reduction. However, carbon contamination can always be found due to the formation of CO32− and erosion of the graphite anode during electrolysis. Herein, we explore the reduction of carbon contamination by controlling CO32− formation. By adjusting the electrolyte composition, it is possible to improve the wettability between the molten salt and the anode and reduce the O2− solubility in molten salt to reduce CO32− formation. This approach reduces carbon contamination, and the current efficiency is also increased by adding 30 wt pct KCl to pure molten CaCl2. The carbon content in the cathode product decreased by 94 pct and the current efficiency was increased by 51 pct, compared with electrolysis in pure CaCl2 molten salt. In addition, the anode corrosion decreased, and the weight loss of the graphite anode decreased by approximately 65 pct after 18 hours of electrolysis.









Similar content being viewed by others
References
1. G Z Chen, D J Fray and T W Farthing, Nature 2000, vol. 407, pp. 361-364.
2. D. Hu, A. Dolganov, M. Ma, B. Bhattacharya, M. T. Bishop and G. Z. Chen, JOM 2018, vol. 70, pp. 129-137.
3. Carsten Schwandt, Rngreg R. Doughty and Rnderek J. Fray, Key Eng. Mater. 2010, vol. 436, pp. 13-25.
4. K. Dring, R. Bhagat, M. Jackson, R. Dashwood and D. Inman, J. Alloys Compd. 2006, vol. 419, pp. 103-109.
D. Vishnu, N. Sanil, L. Shakila, G. Panneerselvam, R. Sudha, K. S. Mohandas and K. Nagarajan, Electrochim. Acta 2013, 100, 51-62.
R. O. Suzuki, H. Noguchi, Y. Haraguchi, S. Natsui and T. Kikuchi, ECS Trans. 2018, 86, 45.
7. C. Schwandt, D. T. L. Alexander and D. J. Fray, Electrochim. Acta 2009, vol. 54, pp. 3819-3829.
8. Liwen Hu, Song Yang, Jianbang Ge, Jun Zhu, Zhenchao Han and Shuqiang Jiao, Journal of Materials Chemistry A 2017, vol. 5, pp. 6219-6225.
9. L. Hu, Y. Song, S. Jiao, Y. Liu, J. Ge, H. Jiao, J. Zhu, J. Wang, H. Zhu and D. J. Fray, ChemSusChem 2016, vol. 9, pp. 588-594.
10. C. Schwandt, Transactions of the Institutions of Mining and Metallurgy, Section C: Mineral Processing and Extractive Metallurgy 2013, vol. 122, pp. 213-218.
11. K. S. Mohandas, Transactions of the Institutions of Mining and Metallurgy, Section C: Mineral Processing and Extractive Metallurgy 2013, vol. 122, pp. 195-212.
12. W. Weng, M. Wang, X. Gong, Z. Wang, D. Wang and Z. Guo, J. Electrochem. Soc. 2017, vol. 164, pp. E360-E366.
13. T. Matsuzaki, S. Natsui, T. Kikuchi and R. O. Suzuki, Mater. Trans 2017, vol. 58, pp. 371-376.
14. D. Tang, K. Zheng, H. Yin, X. Mao, D. R. Sadoway and D. Wang, Electrochim. Acta 2018, vol. 279, pp. 250-257.
15. S. Jiao and D. J. Fray, Metallurgical and Materials Transactions B: Process Metallurgy and Materials Processing Science 2010, vol. 41, pp. 74-79.
16. R. Barnett, K. T. Kilby and D. J. Fray, Metallurgical and Materials Transactions B: Process Metallurgy and Materials Processing Science 2009, vol. 40, pp. 150-157.
17. M. Alzamani and K. Jafarzadeh, Oxid. Met. 2018, vol. 89, pp. 623-640.
18. Y. Sakamura and M. Iizuka, Electrochim. Acta 2016, vol. 189, pp. 74-82.
19. K. T. Kilby, S. Jiao and D. J. Fray, Electrochim. Acta 2010, vol. 55, pp. 7126-7133.
20. Subrat Das, Lanka Dinushke Weerasiri and William Yang, Colloids Surf. A 2017, vol. 516, pp. 23-31.
21. Zhibin Zhao, Bingliang Gao, Yuqing Feng, Yipeng Huang, Zhaowen Wang, Zhongning Shi and Xianwei Hu, JOM 2017, vol. 69, pp. 281-291.
22. Laurent Cassayre, Torstein A. Utigard and Sylvie Bouvet, JOM 2002, vol. 54, pp. 41-45.
23. V. Tomkute, A. Solheim and E. Olsen, Energy Fuels 2014, vol. 28, pp. 5345-5353.
24. Dihua Wang, Bowen Deng, Zhigang Chen, Muxing Gao and Xuhui Mao, Faraday Discuss. 2016, vol. 190, pp. 241-258.
25. Xiao Y. Yan and Derek J. Fray, J. Appl. Electrochem. 2009, vol. 39, pp. 1349-1360.
26. X. Y. Yan and D. J. Fray, Metall. Mater. Trans. B 2002, vol. 33, pp. 685-693.
27. E Gordo, G Z Chen and D J Fray, Electrochim. Acta 2004, vol. 49, pp. 2195-2208.
28. Shuqiang Jiao and Hongmin Zhu, J. Alloys Compd 2007, vol. 438, pp. 243-246.
29. Shulan Wang, Shichao Li, Longfei Wan and Chuanhua Wang, Int. J. Min. Met. Mater 2012, vol. 19, pp. 212-216.
30. M. Yang, X. Lv, R. Wei, J. Xu and C. Bai, Metall. Mater. Trans. B 2018, vol. 49, pp. 1331-1345.
31. Donald A. Wenz, Irving Johnson and Raymond D. Wolson, J. Chem. Eng. Data 1969, vol. 14, pp. 250-252.
32. Carsten Schwandt and Derek J. Fray, Ztschrift Für Naturforschung A 2007, vol. 62, pp. 655-670.
33. George S Perry and Lindsay G Macdonald, J. Nucl. Mater. 1985, vol. 130, pp. 234-241.
M. A. Cooksey, M. P. Taylor and J. J. J. Chen, JOM 2008, 60, 51-57.
35. M. Ma, D. Wang, W. Wang, X. Hu, X. Jin and G. Z. Chen, J. Alloys Compd. 2006, vol. 420, pp. 37-45.
36. Zhongren Zhou, Yingjie Zhang, Yixin Hua, Peng Dong, Cunying Xu, Yan Li and Ding Wang, Electrochim. Acta 2018, vol. 271, pp. 490-497.
37. J. Sure, A. R. Shankar, S. Ramya, C. Mallika and U. K. Mudali, Carbon 2014, vol. 67, pp. 643-655.
D. Vishnu, J. Sure and K. S. Mohandas, Carbon 2015, 93, 782-792.
Acknowledgments
This work was supported financially support by the National Natural Science Foundation of China (Grant No. 51674054), and supported by the Chongqing Key Laboratory of Vanadium-Titanium Metallurgy and New Materials, Chongqing University, Chongqing 400044, PR China
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Manuscript submitted August 18, 2020, accepted January 3, 2021.
Rights and permissions
About this article
Cite this article
Ma, T., Luo, X., Yang, Y. et al. Reducing Carbon Contamination by Controlling CO32− Formation During Electrochemical Reduction of TiO2. Metall Mater Trans B 52, 1061–1070 (2021). https://doi.org/10.1007/s11663-021-02078-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11663-021-02078-w