Abstract
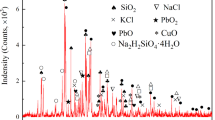
A pyrometallurgical process of selective oxidation roasting of the antimony-bearing dust using CuO is put forward, in which the antimony component is oxidized to Sb2O4 staying in the roasted residue, and arsenic is volatilized in the form of As2O3. The addition of CuO has an active effect on the arsenic volatilization, because structures of some complicated As-Sb phases in the dust are destroyed after the “Sb” component in them is oxidized to Sb2O4, and this part of arsenic might be transformed to As2O3, which continues to volatilize. However, the arsenic volatilization rate decreases with the CuO amount in a certain range, which is attributed to the greater formation of Cu3 (AsO4)2 and Cu3As. Under the conditions of roasting temperature of 673 K (400 °C), roasting time of 100 minutes, CuO amount of 34.54 mass pct, and N2 flow rate of 30 mL/min, 91.50 pct arsenic and only 8.63 pct antimony go into the smoke.












Similar content being viewed by others
References
1. M. Filella, N. Belzile, and Y.W. Chen: Earth-Sci. Rev., 2002, vol. 57, pp. 125–76.
2. X. Wang, J.P. Wang, C.H. Liu, and F.F. Zhang: China Met. Bull., 2014, vol. 23, pp. 6–10 (in Chinese with English abstract).
3. M.C. He, X.Q. Wang, F.C. Wu, and Z.Y. Fu: Sci. Total Environ., 2012, vols. 421–422, pp. 41–45.
4. H. Binz and B. Friedrich: Chem. Ingen. Techn., 2015, vol. 87, pp. 1569–79.
5. M. Lundgren, U. Leimalm, G. Hyllander, L.S. Ökvist, and B. BjÖrkman: ISIJ Int., 2010, vol. 50, pp. 1570—80.
6. R.A. Shawabkeh: Hydrometallurgy, 2010, vol. 104, pp. 61—65.
7. Y.L. Zhang and E. Kasai: ISIJ Int., 2005, vol. 45, pp. 1813—19.
8. C.S. Chen, Y.J. Shih, and Y.H. Huang: Waste Management, 2016, vol. 52, pp. 212—20.
9. Z.F. Xu, Q. Li, and H.P. Nie: Trans. Nonferrous Met. Soc. China, 2010, vol. 20, pp. s176—s181.
10. C. Vandecasteele, V. Dutré, D. Geysen, and G. Wauters: Waste Management, 2002, vol. 22, pp. 143—46.
11. V. Montenegro, H. Sano, and T. Fujisawa: Miner. Eng., 2013, vol. 49, pp. 184—89.
12. Y. Chen, T. Liao, G.B. Li, B.Z. Chen, and X.C. Shi: Miner. Eng., 2012, vol. 39, pp. 23—28.
13. H.L. Yang, M.C. He, and X.Q. Wing: Environ. Geochem. Health, 2015, vol. 37, pp. 21—33.
14. M.P. Taylor, S.A. Mould, L.J. Kristensen, and M. Rouillon: Environ. Res., 2014, vol. 135, pp. 296—303.
15. D. Dupont, S. Arnout, P.T. Jones, and K. Binnemans: J. Sustainable Metall., 2016, vol. 2, pp. 79—103.
16. J. Chen, G. Xie, and D.P. Zhao: Chin. J. Inorg. Analyt. Chem., 2014, vol. 4, pp. 11—15 (in Chinese with English abstract).
17. G.A. Brooks, W.J. Rankin, and N.B. Gray: Metall. Mater. Trans. B, 1994, vol. 25B, pp. 873–84.
18. H.B. Tang, Q.W. Qin, Y. Guo, X. Zheng, P. Xue, and G.Q. Li: Conserv. Utiliz. Miner. Res., 2014, vol. 3, pp. 35—38 (in Chinese with English abstract).
19. L. Li, R.J. Zhang, B. Liao, and X.F. Xie: Chin. J. Process Eng., 2014, vol. 14, pp. 71—77 (in Chinese with English abstract).
H.B. Yuan, Y.Y. Zhu, and J.B. Zhang: J. Central South Univ. (Sci. Technol.), 2013, vol. 44, pp. 2200—05 (in Chinese with English abstract).
21. A. Aracena, O. Jerez, and C. Antonucci: Trans. Nonferrous Met. Soc. China, 2016, vol. 26, pp. 294—300.
22. R.P. Padilla, G. Ramírez, and M.C. Ruiz: Metall. Mater. Trans. B, 2010, vol. 40B, pp. 1284-92.
23. J.E. Mauser. Metall. Mater. Trans. B, 1982, vol. 13B, pp. 511–13.
24. G.A. Brooks, W.J. Rankin, and N.B. Gray: Metall. Mater. Trans. B, 1994, vol. 25B, pp. 865–71.
25. J.S. Wang: Copper Ind. Eng., 2005, vol. 1, pp. 27—28 (in Chinese with English abstract).
26. S. Kashiwakura, H. Ohno, K. Matsubae-Yokoyama, Y. Kumagai, H. Kubo, and T. Nagasaka: J. Hazardous Mater., 2010, vol. 181, pp. 419—25.
27. X.Y. Guo, J. Shi, Y. Yia, Q.H. Tian, and D. Li: J. Environ. Chem. Eng., 2015, vol. 3, pp. 2236—42.
28. I. Mihajlovic, N. Strbac, Z. Zivkovic, R. Kovacevic, and M. Stehernik: Miner. Eng., 2007, vol. 20, pp. 26—33.
29. X.X. Jiang, G.X. He, X.G. Li, and Q. Lu: Hydrometall. China, 2010, vol. 29, pp. 199—202 (in Chinese with English abstract).
30. E. Vircikova and M. Havlik: JOM, 1999, vol. 51, pp. 20—23.
FactSage 7.0: FactPS—FACT Pure Substances Database, Thermfact/CRCT and GTT Technologies, 2015.
Acknowledgments
The authors express thanks to the National Science Fund for Distinguished Regional Scholars (Grant No. 51564034) and Scientific and Technological Leading Talent Projects in Yunnan Province (Grant No. 2015HA019) for financial support of this research.
Author information
Authors and Affiliations
Corresponding author
Additional information
Manuscript submitted July 20, 2016.
Rights and permissions
About this article
Cite this article
Zhong, DP., Li, L. & Tan, C. Separation of Arsenic from the Antimony-Bearing Dust through Selective Oxidation Using CuO. Metall Mater Trans B 48, 1308–1314 (2017). https://doi.org/10.1007/s11663-016-0896-2
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11663-016-0896-2