Abstract
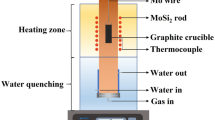
CaO-SiO2-Nd2O3 slags were equilibrated at 1773 K and 1873 K (1500 °C and 1600 °C) for 24 hours in Ar, and quenched in water to determine the operative phase relations. The composition and crystallinity of the phases in equilibrium were determined by EPMA-WDS and EBSD, respectively. Based on these analyses, the liquid stability region was accurately determined, and a large part of the isothermal section of the phase diagram was constructed. Data resulting from this work can be used to generate a thermodynamic database for rare-earth oxide-containing systems and to support further investigation on separation of rare earths from metallurgical slags or other residues through high-temperature processing.









Similar content being viewed by others
References
K. Binnemans, P. T. Jones, B. Blanpain, T. Van Gerven, Y. Yang, A. Walton, and M. Buchert: J. Clean. Prod., 2013, vol 51, pp.1-22.
K. Binnemans, P. T. Jones, B. Blanpain, T. Van Gerven, and Y. Pontikes: J. Clean. Prod., 2015, vol 99, pp. 17-38.
S. Chu: Critical Materials Strategy, U.S. Department of Energy, December 2011. http://energy.gov/sites/prod/files/DOE_CMS2011_FINAL_Full.pdf
R.J. Weber and D.J. Reisman: Rare Earth Element: A Review of Production, Processing, Recycling, and Associated Environmental Issues, U.S. Environmental Protection Agency, August 2012. http://www.epa.gov/superfund/remedytech/tsp/download/2012_spring_meeting/fff_wed/4_weber-rare_earth_minerals.pdf.
L.T. Peiro, G.V. Mendez, and R.U. Ayres: Rare and Critical Metals as By-products and the Implications for Future Supply, INSEAD Social Innovation Centre, 2011. http://www.insead.edu/facultyresearch/research/doc.cfm?did=48916
T.G. Goonan: Rare earth elements—End use and recyclability, U.S. Geological Survey Scientific Investigations Report 5094, 2011. http://pubs.usgs.gov/sir/2011/5094/
T. Müller, and B. Friedrich: J. Power Sources, 2006, vol 158 [2], pp.1498-1509.
K. Tang, A. Ciftja, C. van der Eijk, S. Wilson, and G. Tranell: J. Min. Metall. Sect. B Metall., 2013, vol 49, 233-36.
T. Elwert, D. Goldmann, T. Schirmer, and K. Strauss: Chem. Ing. Tech., 2014, vol 86, pp. 840-47.
M. Hillert, B. Sundman, and Z. Wang: Met. Trans. B, 1990, vol 21B, pp. 303-12.
J. E. Saal, D. Shin, A. J. Stevenson, G. L. Messing, and A. Liu: J. Am. Ceram. Soc. 2010, vol 93 [12], pp. 4158-67.
E. M. Levin, C. R. Robbins, H. F. McMurdie, and M. K. Reser, Phase Diagram For Ceramists, American ceramic society, Columbus, OH, 1969.
W. Wong-Ng, W. Laws, K. R. Talley, Q. Huang, Y. Yan, J. Martin, and J. A. Kaduk: J. Solid State Chem., 2014, vol 215, pp. 128-34.
N.A. Toropov and N.F. Fedorov, Izv. Akad. Nauk SSSR: Neorg. Mater., 1965 vol. 1 [1], pp. 126–130; Inorg. Mater. (Engl. Transl.), 1965, vol 1 [1], pp. 110–13.
C. M. Jantzen, and F.P. Glasser: Am. Ceram. Soc. Bull., 1979, vol 58 [4], pp. 459-66.
M. Zhao, L. Song, X. Fan, The Boundary Theory of Phase Diagrams and Its Application, Springer Berlin Heidelberg, Berlin, Germany, 2011.
J. A. Fahey, W. J. Weber, and F. J. Rotella: J. Solid State Chem.,1985, vol 60, pp. 145-58.
T.H. Le, N.R. Brooks, K. Binnemans, B. Blanpain, M. Guo, and L. Van Meervelt: Acta crystallogr. E, 2015, accepted.
Acknowledgments
The authors acknowledge the support from the Hercules Foundation (Project No. ZW09-09) in the use of the FEG-EPMA and FEI-Nova Nanosem system and from the GOA/13/008 project at the KU Leuven.
Author information
Authors and Affiliations
Corresponding author
Additional information
Manuscript submitted December 1, 2015.
Rights and permissions
About this article
Cite this article
Le, T.H., Malfliet, A., Blanpain, B. et al. Phase Relations of the CaO-SiO2-Nd2O3 System and the Implication for Rare Earths Recycling. Metall Mater Trans B 47, 1736–1744 (2016). https://doi.org/10.1007/s11663-016-0634-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11663-016-0634-9