Abstract
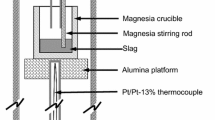
The carbon solubility in the CaO-SiO2-3MgO-CaF2 slag system at 1773 K (1500 °C) was investigated under CO/Ar and CO/N2 gases. Higher extended basicity [(CaO + MgO)/SiO2) increased the carbon solubility in the slag as the activity of free oxygen ions (\( \varvec{a}_{{{\mathbf{O}}^{2 - } }} \)] promoted the reaction of the free carbide mechanism. Higher CaF2 also resulted in higher carbon dissolution into the slag as fluorine ions interact with the bridged oxygen (O0) in the melt structure to increase the activity of the free oxygen ions in the melt. Structural information obtained from the Fourier transformed infra-red (FT-IR) and Raman revealed a depolymerization of the network structure as the simpler structural units of NBO/Si = 4 increased and the Si-O-Si bending vibrations decreased with higher basicity and CaF2 content. This correlated well with higher free oxygen ions (O2−) in the slag system and subsequently higher carbon dissolution. A correlation of the theoretical optical basicity (Λth) with the logarithm of the carbon content in slag showed a relatively similar trend and an increase of carbon was observed with higher optical basicity.





Similar content being viewed by others
References
K. Schwerdtfeger and H.G Schubert: Metall. Trans. B., 1977, vol. 8B, pp. 535-540.
R.A. Berryman and I.D. Sommerville: Metall. Trans. B., 1992, vol. 23B, pp. 223-227.
I. Sohn, D.J. Min, and J.H. Park: Steel Res., 1999, vol. 70, pp. 215-220.
J.-Y. Park and I. Sohn: Metall. Mater. Trans. B., 2013, vol. 44B, pp. 123-132.
M. Kuwata and H. Suito: Metall. Mater. Trans. B., 1996, vol. 28B, pp. 57-64.
J.H. Park, D.J. Min, and H.S. Song: ISIJ Int., 2002, vol. 42, pp. 127-131.
G. J. W. Kor and F. D. Richardson: Trans. of the Metall. Society of AIME, 1969, vol. 245, pp. 319-327.
A. Mitchell: Trans. Faraday Society, 1967, vol. 63, pp. 1408-1417.
D. Kumar, R. G. Ward and D. J. Williams: Disc. Faraday Society, 1961, vol. 32, pp. 147-155.
G.-H. Kim, C.-S., Kim, and I. Sohn: ISIJ Int., 2013, vol. 53, pp. 170–76.
J.-Y. Park, S.-J. Park, W.-S. Chang, and I. Sohn: J. Am. Cer. Soc., 2012, vol. 95, pp. 1756-1763.
M. Kuwata and H. Suito: MMTB, 1996, vol. 27B, pp. 57-64.
B.O. Mysen, D. Virgo, and C. M. Scarfe: Am. Mineral., 1980, vol. 65, pp. 690-710.
P. McMillan: Am. Mineral., 1984, vol. 69, pp. 645-659.
H.W. Nesbitt, G.M. Bancroft, G.S. Henderson, R.Ho, K.N. Dalby, Y. Huang, and Z. Yan: J. Non-Cryst. Solids, 2011, vol. 357, pp. 170-180.
J. A. Duffy and M.D. Ingram: Phys. Chem. Glasses, 1975, vol. 16, pp. 119-123.
C. J. B. Fincham and F. D. Richardson: Proc. Roy. Soc., 1954, vol. A223, pp. 40-62.
T. Nakamura, T. Yokoyama, and J.M. Toguri: ISIJ Int., 1993, vol. 33, pp. 204-209.
J.H. Park and D.J. Min: Metall. Mater. Trans. B., 1999, vol. 30B, pp. 1045-1052.
J.H. Park, G.H. Park and Y.E. Lee: ISIJ Int., 2010, vol. 50, pp. 1078-1083.
This study was partially supported by the BK21PLUS Project at the Division of the Eco-Humantronics Information Materials, the National Science Foundation of Korea Project No. 2012-8-0486 and the Priority Research Centers Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (2009-0093823).
Author information
Authors and Affiliations
Corresponding author
Additional information
Manuscript submitted August 27, 2013.
Rights and permissions
About this article
Cite this article
Park, JY., Jung, SM. & Sohn, I. Carbon Solubility in the CaO-SiO2-3MgO-CaF2 Slag System. Metall Mater Trans B 45, 329–333 (2014). https://doi.org/10.1007/s11663-014-0028-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11663-014-0028-9