Abstract
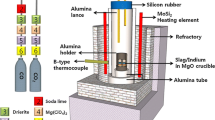
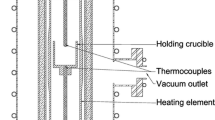
The solubility of indium in a molten CaO-SiO2-Al2O3 system was measured at 1773 K (1500 °C) to establish the dissolution mechanism of indium under a highly reducing atmosphere. The solubility of indium increases with increasing oxygen potential, whereas it decreases with increased activity of basic oxide. Therefore, a dissolution mechanism of indium can be constructed according to the following equation:
The relationship between indium capacity and sulfide capacity shows a good correlation that is consistent with theoretical expectations. The enthalpy change of the indium dissolution reaction is negative, which indicates that the dissolution is an exothermic reaction. The heat of dissolution into high-silica melts is greater than that into low-silica melts. The solubility of indium is strongly dependent on the silica content. The activity coefficient, and thus the excess free energy of In2O, decreases linearly with increasing silica content, indicating that the In2O is believed to behave as a weak basic oxide in the current CaO-SiO2-Al2O3 ternary system under reducing conditions.










Similar content being viewed by others
References
J.D. Jorgenson and M.W. George: “USGS Mineral Commodity Profile,” Open-File Report 2004-1300, USGS, Reston, VA, 2005.
www.roskill.com/report/indium. Accessed April 2011.
H.M. Liu, C.C. Wu, Y.H. Lin, and C.K. Chiang: J. Hazard Mater., 2009, vol. 172, pp. 744-48.
S. Virolainen, D. Ibana, and E. Paatero: Hydrometallurgy, 2011, vol. 107, pp. 56-61.
E. Yamasue, R. Minamino, T. Numata, K. Nakajima, S. Murakami, I. Daigo, S. Hashimoto, H. Okumura, and K. Ishihara: Mater. Trans., 2009, vol. 50, pp. 1536-40.
O. Terakado, T. Saeki, R. Irizato, and M. Hirasawa: Mater. Trans., 2010, vol. 51, pp. 1136-40.
E. Johnson, L. Oden, and J. Koch: “Laboratory Investigations on the Behavior of Accessory Elements in Lead Blast Furnace Smelting,” Report no. 8753, Albany Res. Cent., US Bur. Mines., 1988.
G.B. Hoang and D.R. Swinbourne: Min. Process. Extract. Metall. (Trans. Inst. Min. Metall. C), 2007, vol. 116, pp. 133-38.
E.T. Turkdogan: Physical Chemistry of High Temperature Technology, Academic Press, New York, NY, 1980, pp. 1-24.
J.H. Park and D.J. Min: Metall. Mater. Trans. B, 1999, vol. 30B, pp. 689-94.
H. Henao, M. Hino, and K. Itagaki: Proc. Yazawa Int. Symp., F. Kongoli, K. Itagaki, C. Yamauchi, and H.Y. Sohn, eds.,vol. 1A, TMS, Warrendale, PA, 2003, pp. 329–40.
S.H. Lee, S.M. Moon, J.H. Park, and D.J. Min: Metall. Mater. Trans. B, 2002, vol. 33B, pp. 55-59.
J.H. Park and D.J. Min: ISIJ Int., 2004, vol. 44, pp. 223-28.
J.H. Park and D.J. Min: Metall. Mater. Trans., 2006, vol. 47, pp. 2038-43.
J.H. Park, G.H. Park, and Y.B. Kang: ISIJ Int., 2010, vol. 50, pp. 1078-83.
G.H. Park, Y.B. Kang, and J.H. Park: ISIJ Int., 2011, vol. 51 (9), pp. 1375–82.
FactSage. www.factsage.com. Accessed April 2011.
C.W. Bale, E. Bélisle, P. Chartrand, S.A. Decterov, G. Eriksson, K. Hack, I.-H. Jung, Y.-B. Kang, J. Melancon, A.D. Pelton, C. Robelin, and S. Petersen: CALPHAD, 2009, vol. 33, pp. 295-311.
M.O. Suk and J.H. Park: J. Am. Ceram. Soc., 2009, vol. 92, pp. 717-23.
J.H. Park, I.H. Jung, and S.B. Lee: Met. Mater. Int., 2009, vol. 15, pp. 677-81.
J.H. Park: Met. Mater. Int., 2010, vol. 16, pp. 987-92.
J.H. Park, J.G. Park, D.J. Min, Y.E. Lee, and Y.B. Kang: J. Eur. Ceram. Soc., 2010, vol. 30, pp. 3181-86.
J.H. Park, M.O. Suk, I.H. Jung, M. Guo, and B. Blanpain: Steel Res. Int., 2010, vol. 81, pp. 860-68.
C.J.B. Finchan and F.D. Richardson: J. Iron Steel Inst., 1954, vol. 178, pp. 4-15.
C.J.B. Finchan and F.D. Richardson: Proc. Roy. Soc. London A, 1954, vol. 223A, pp. 40-62.
M.R. Kalyanram, T.G. MacFarlane, and H.B. Bell: J. Iron Steel Inst., 1960, vol. 195, pp. 58-64.
C.H.P. Lupis: Chemical Thermodynamics of Materials, Prentice Hall, New York, NY, 1993, pp. 114-20.
J. Valderraman and K.T. Jacob: Thermochim. Acta, 1997, vol. 21, pp. 215-24.
Acknowledgments
The authors would like to thank Professor Dong Joon Min, POSCO Professor, Materials Science and Engineering, Yonsei University, Seoul, Korea, for providing X-ray fluorescence analysis.
Author information
Authors and Affiliations
Corresponding author
Additional information
Manuscript submitted May 5, 2011.
An erratum to this article is available at http://dx.doi.org/10.1007/s11663-014-0247-0.
Rights and permissions
About this article
Cite this article
Ko, K.Y., Park, J.H. Dissolution Behavior of Indium in CaO-SiO2-Al2O3 Slag. Metall Mater Trans B 42, 1224–1230 (2011). https://doi.org/10.1007/s11663-011-9566-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11663-011-9566-6