Abstract
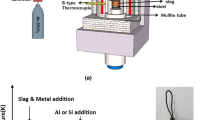
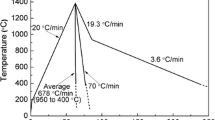
During the processing of liquid steels, nonmetallic inclusions precipitate and evolve under conditions that often involve transient changes in chemistry or temperature, which could be reflected in the final products unless sufficient time is provided for equilibration to be established. The current study is focused on documenting the changes that inclusions undergo in terms of chemistry, shape, and structure when Ti is added in smaller batches, to avoid reactions caused by locally high Ti concentrations and result in a final melt chemistry with a Ti/Al ratio of 1 in the melt corresponding to the chemistry of interstitial free (IF) steel melts in the ladle furnace. When Ti was added in two increments, the inclusion composition was altered from spherical and irregular Al2O3 to mostly irregular inclusions that included both Al and Ti after the first addition. The second addition did not cause any change, but with time, the inclusion chemistry reverted back to Al2O3 with the morphology change remaining. For the case when Ti was added in four increments, however, the inclusion chemistry was modified largely after the first Ti addition, but the inclusion morphology did not change to the irregular-dominant case until the second Ti addition was made. Part of the Ti-containing inclusions was the result of the dissolution of TiO x into Al2O3. It seems that a critical Ti/Al ratio exists in between 1/4 and 1/2, which determines the morphological change. This finding might be coincident with the required increase in Ti and the decrease of local oxygen, which causes a precipitation of a new TiO x phase as opposed to dissolution of TiO x in Al2O3. Prolonging the interval between each Ti addition would allow the inclusion change in composition, reverting from the Ti-containing dominant stage to primarily Al2O3, but not in morphology.













Similar content being viewed by others
References
Y. Gao and K. Sorimachi: ISIJ Int., 1993, vol. 33, pp. 291-97.
S. Basu, S.K. Choudhary, and N.U. Girase: ISIJ Int., 2004, vol. 44, pp. 1653-60.
C. van der Eijk, O. Grong, and J. Walmsley: Mater. Sci. Technol., 2000, vol. 16, pp. 55-64.
C. Wang, N.T. Nufher, and S. Sridhar: Metall. Mater. Trans. B, 2009, vol. 40, pp. 1005-21.
C. Wang, N.T. Nufher, and S. Sridhar: Metall. Mater. Trans. B, 2009, vol. 40, pp. 1022-34.
M.K. Sun, I.H. Jung, and H.G. Lee: Metall. Mater. Int., 2008, vol. 14, pp. 791-98.
M.A. van Ende, M.X. Guo, R. Dekkers, M. Burty, J. van Dyck, P.T. Jone, B. Blanpain, and P. Wallants: ISIJ Int., 2009, vol. 49, pp. 1133-40. -->
F. Ruby-Meyer, J. Lehmann, and H. Gaye: Scand. J. Metall., 2000, vol. 29, pp. 206-12.
I.I. Kornilov, A.E. Shmelev, V.P. Glebov, and E.S. Zhmud: Dokl. Akad. Nauk. SSSR, 1972, vol. 207, pp. 654-56.
I.I. Kornilov, F.N. Tavadze, G.N. Ronami, K.M. Kostantinov, T.A. Peradze, and Y.A. Maksimov: Soobshch. Akad. Nauk Gruz. SSR, 1972, vol. 66, pp. 133-36,
W.D. McKee, Jr. and E. Aleshin: J. Am. Ceram. Soc., 1963, vol. 46, pp. 54-58.
E.R. Winkler, J.F. Sarver, and I.B. Cutler: J. Am. Ceram. Soc., 1966, vol. 49, pp. 634-37.
S.K. Roy and R.I. Coble: J. Am. Ceram. Soc., 1968, vol. 51, pp. 1-6.
M. Ohta and K. Morita: ISIJ Int., 2002, vol. 42, pp. 474-81.
H. Amitani, K. Morita, and N. Sano: ISIJ Int., 1996, vol. 36, pp. S26.
K. Matsunaga, A. Nakamura, T. Yamamoto, and Y. Ikuhara: Phys. Rev. B, 2003, vol. 68, pp. 214102.
T. Mizoguchi, M. Sakurai, A. Nakamura, K. Matsunaga, I. Tanaka, T. Yamamoto, and Y. Ikuhara: Phys. Rev. B, 2004, vol. 70, pp. 153101.
M. Eckert, L. Bencze, D. Kath, H. Nickel, and K. Hilpert: Ber. Bunsenges. Phys. Chem., 1996, vol. 100, pp. 418-24.
N.S. Jacobson, M.P. Brady, and G.M. Mehrotra: Oxid. Met., 1999, vol. 52, pp. 537-56.
J. Nakano: Private Communication, December 17, 2009.
D.C. Park, I.H. Jung, P.C. H. Rhee, and H.G. Lee: ISIJ Int., 2004, vol. 44, pp. 1669-78.
S. Kim, J.H. Kwak, J.H. Chung, and K. Cho: Metall. Mater. Int., 2002, vol. 8, pp. 155-61.
N. Takahashi: Tetsu-to-Hagane, 1982, vol. 68, pp. S588-97.
Acknowledgments
The authors acknowledge the Center for Iron and Steelmaking Research (CISR) for financial support, and Mr. N. Kikuchi and Ms. M. Iwasa of JFE Steel for chemical analysis. Fruitful discussions with Dr. J. Lehmann (ArcelorMitall), Mr W. Tiekin (Corus), and Dr. J. Nakano (NETL) are greatly appreciated.
Author information
Authors and Affiliations
Corresponding author
Additional information
Manuscript submitted March 22, 2010.
Rights and permissions
About this article
Cite this article
Wang, C., Nuhfer, N.T. & Sridhar, S. Transient Behavior of Inclusion Chemistry, Shape, and Structure in Fe-Al-Ti-O Melts: Effect of Gradual Increase in Ti. Metall Mater Trans B 41, 1084–1094 (2010). https://doi.org/10.1007/s11663-010-9397-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11663-010-9397-x