Abstract
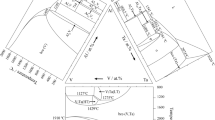
Equilibrium phase relations in the PbO-Al2O3-SiO2 system have been investigated experimentally by means of high-temperature equilibration, quenching, and electron probe X-ray microanalysis (EPMA). The system has 21 primary phase fields including three monoxides (PbO, Al2O3, and SiO2), seven binary compounds (Al6Si2O13, PbAl2O4, PbAl12O19, Pb2Al2O5, PbSiO3, Pb2SiO4, and Pb4SiO6), and eleven ternary compounds (PbAl2Si2O8, Pb3Al10SiO20, Pb4Al2Si2O11, Pb4Al4SiO12, Pb4Al4Si3O16, Pb4Al4Si5O20, Pb5Al2Si10O28, Pb6Al2Si6O21, Pb8Al2Si4O19, Pb12Al2Si17O49, and Pb12Al2Si20O55). Three new ternary compounds, Pb4Al4SiO12, Pb4Al4Si5O20, and Pb12Al2Si17O49, were observed and characterized by EPMA. No extensive solid solution in any of the compounds was found in the present study. The liquidus isotherms were experimentally determined in most of the primary phase fields in the temperature range from 923 to 1873 K, and the ternary phase diagram of the PbO-Al2O3-SiO2 system has been constructed.
Similar content being viewed by others
References
E. Jak, S. Degterov, B. Zhao, A.D. Pelton, and P.C. Hayes: Proc. Zinc and Lead Processing Symp., Calgary, CIM, Montreal, 1998, pp. 313–33.
E. Jak, S. Degterov, B. Zhao, A.D. Pelton, and P.C. Hayes: Metall. Mater. Trans. B, 2000, vol. 31B, pp. 621–30.
B. Zhao: Ph.D. Thesis, The University of Queensland, Queensland, 1999.
N.L. Bowen and J.W. Greig: J. Am. Ceram. Soc., 1924, vol. 7, pp. 238–54.
E.C. Shears and W.A. Archibald: Iron and Steel, London, 1954, vol. 27, pp. 26–30 and 61–66.
F.J. Klug, S. Prochazka, and R.H. Doremus: J. Am. Ceram. Soc., 1987, vol. 70, pp. 750–59.
J.W. Welch: Nature 1960, vol. 186, pp. 546–47.
S. Aramaki and R. Roy: J. Am. Ceram. Soc., 1962, vol. 45, pp. 229–42.
N.A. Toropov and F.Ya. Galakhov: Dokl. Akad. Nauk SSSR, 1951, vol. 78, pp. 299–302.
S.P. Chaudhuri: Ceram. Int., 1987, vol. 13, pp. 167–75.
G. Eriksson and A.D. Pelton: Metall. Trans. B, 1993, vol. 24B, pp. 807–16.
R.M. Smart and F.P. Glasser: J. Am. Ceram. Soc., 1974, vol. 57 (9), pp. 378–82.
R.F. Geller, A.S. Creamer, and E.N. Bunting: Research Paper No. RP 705, National Bureau of Standards, Gaithersburg, MD, 1934, vol. 13, pp. 237–44.
U. Kuxmann and P. Fischer: Erzmetallurgy, 1974, vol. 27, pp. 533–37.
P.D. Calvert and R.R. Shaw: J. Am. Ceram. Soc., 1970, vol. 53, pp. 350–52.
E. Jak, S. Degterov, P. Wu, P.C. Hayes, and A.D. Pelton: Metall. Mater. Trans. B, 1997, vol. 28B, pp. 1011–18.
R.F. Geller and E.N. Bunting: J. Res. Nat. Bur. Stand., 1943, vol. 31, pp. 255–70.
B.R. Powell, Jr.: Report No. LBL-8219, Lawrence Berkely Lab., University of California, Berkeley, CA, 1978, p. 54.
C.W. Bale, A.D. Pelton, and W.T. Thompson: Facility for the Analysis of Chemical Thermodynamics (F*A*C*T), Ecole Polytechnique, Montreal, 2000 (http://www.crct.polymtl.ca).
Y. Dessureault: Ph.D. Thesis, Ecole Polytechnique, Montreal, 1993.
V.S. Haensel, A. Willgallis, and H. Heyer: Glastechn. Ber., 1976, vol. 9, pp. 207–10.
P. Benna, M. Tribaudino, and E. Bruno: Am. Mineral., 1999, vol. 84, pp. 120–29.
A.O. Mylyanych, M.A. Sheredko, and S.K. Melnyk: J. Anal. At. Spectrom, 1999, vol. 14, pp. 513–21.
E. Sugimoto and Z. Kosuka: Trans. Jpn. Inst. Met., 1978, vol. 19, pp. 275–80.
E. Jak, N. Liu, and P.C. Hayes: Metall. Mater. Trans. B, 1998, vol. 29B, pp. 541–53.
E. Jak, S. Degterov, P.C. Hayes, and A.D. Pelton: Can. Metall. Q., 1998, vol. 37, pp. 41–47.
E. Jak, H.G. Lee, and P.C. Hayes: Kor. IMMJ, 1995, vol. 1, pp. 1–8.
J. Shyu and Y. Yang: J. Mater. Sci., 1996, vol. 31, pp. 4859–63.
H.J. Scheel: Z. Kristallogr., 1971, vol. 133, pp. 264–72.
J.F. Argyle and F.A. Hummel: J. Am. Ceram. Soc., 1963, vol. 46, pp. 10–14.
J.J. Comer, W.J. Croft, M. Kestigian, and J.R. Carter: Mater. Res. Bull., 1967, vol. 2, pp. 293–302.
K. Torkar, H. Krischner, and H. Moser: Ber. Dtsch. Keram. Ges., 1966, vol. 43, pp. 259–63.
JCPDS-ICCD No. 27-1183, Powder Diffraction File, International Centre for Diffraction Data, Pennsylvania, 1999.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Chen, S., Zhao, B., Hayes, P.C. et al. Experimental study of phase equilibria in the PbO-Al2O3-SiO2 system. Metall Mater Trans B 32, 997–1005 (2001). https://doi.org/10.1007/s11663-001-0088-5
Received:
Issue Date:
DOI: https://doi.org/10.1007/s11663-001-0088-5