Abstract
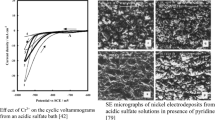
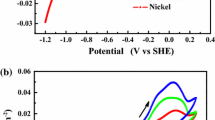
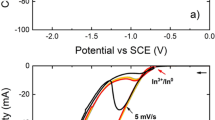
The effect of varying concentrations of Al3+ on the morphology and current efficiency of electrodeposited nickel from an acid-sulfate electrolyte was studied. Concentrations of Al3+ of 20 to 100 ppm significantly degraded the nickel-cathode morphology and current efficiency. However, it was found that higher concentrations of Al3+, in the range of 1 to 5 g/L, depending on the electrolyte parameters and operating conditions, improved the deposit morphology compared to deposition from a pure electrolyte. However, the current efficiency remained low. Physical characterization of the nickel revealed that a smoother, more uniform surface resulted from deposition in the presence of 1 g/L or greater of Al3+. Impedance analysis indicated the presence of an inhibiting layer on the cathode when low concentrations of Al3+ were present. Concentrations of Al3+ greater than 1 g/L appeared to modify this layer and the nickel-deposition mechanism, allowing improved nickel morphology and structure.
Similar content being viewed by others
References
M. Holm: Ph.D. Dissertation, University of Missouri—Rolla, Rolla, MO, 1999.
S.K. Gogia and S.C. Das: Metall. Trans. B, 1988, vol. 19B, pp. 823–30.
S.K. Gogia and S.C. Das: J. Appl. Electrochem., 1991, 21, pp. 64–72.
Z. Zhang: Ph.D. Dissertation, University of Missouri-Rolla, Rolla, MO, 1997.
K. Knuutila, O. Forsen, and A. Pehkonen: in The Electrorefining and Winning of Copper, J.E. Hoffman, ed., TMS, Warrendale, PA, 1987, pp. 129–43.
D.F. Suarez and F.A. Olson: in The Electrorefining and Winning of Copper, J.E. Hoffman, ed., TMS, Warrendale, PA, 1987, pp. 145–69.
Eric Jackson: Hydrometallurgical Extraction and Reclamation, John Wiley & Sons, New York, NY, 1986, p. 218.
X. Tang, P. Yu, T.J. O’Keefe, and G. Houlachi: in Aqueous Electrotechnologies: Progress in Theory and Practice, D. Dreisinger, ed., TMS, Warrendale, PA, 1997, pp. 115–25.
D. Gangasingh and J.B. Talbot: J. Electrochem. Soc., 1991, vol. 138, pp. 3605–11.
K. Yin and B.T. Lin: Surf. Coating Technol., 1996, 78, pp. 205–10.
C.H. Huang: Water Res., 1995, vol. 29, pp. 1821–26.
K.N. Njau and L.J.J. Janssen: J. Appl. Electrochem., 1995, 25, pp. 982–86.
J. Ji, W.C. Cooper, D.B. Dreisinger, and E. Peters: J. Appl. Electrochem., 1995, 25, pp. 642–50.
H. Deligianni and L.T. Romankiw: Proc. Symp. on Magnetic Materials, Processes and Devices, L.T. Romankiw and D.A.R. Herman, Jr., eds., The Electrochemical Society, Pennington, NJ, 1990, pp. 407–22.
J.P. Hoare: J. Electrochem. Soc., 1987, vol. 134, pp. 3102–03.
I. Epelboin and R. Wiart: J. Electrochem. Soc., 1971, vol. 118, p. 1577.
I. Epelboin, M. Joussellin, and R. Wiart: J. Electroanal. Chem., 1981, vol. 119, p. 61.
F. Chassaing, M. Joussellin, and R. Wiart: J. Electroanal. Chem., 1983, vol. 157, p. 75.
S. Watson: J. Electrochem. Soc., 1993, vol. 140, p. 2235.
S. Watson and R. Walters: J. Electrochem. Soc., 1991, vol. 138, p. 3633.
G. Lindbergh and D. Simonsson: Electrochim. Acta, 1991, vol. 36, pp. 1985–94.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
O’keefe, T.J., Holm, M. The anomalous behavior of Al3+ in nickel electrowinning from sulfate electrolytes. Metall Mater Trans B 31, 1203–1211 (2000). https://doi.org/10.1007/s11663-000-0007-1
Received:
Issue Date:
DOI: https://doi.org/10.1007/s11663-000-0007-1