Abstract
Summary
The OSARIDELPHI study evaluated the level of agreement between specialists in osteoporosis regarding the management of patients with high-risk fractures in Spain. The results provide expert-based recommendations for prevention, diagnosis, and treatment related to fracture risk. Therefore, the study facilitates clinical decision-making for managing this patient’s profile.
Purpose
To evaluate the level of agreement between specialists in osteoporosis regarding the management of patients with high-risk fractures in Spain.
Methods
A two-round Delphi study was performed using an online survey. In round 1, panel members rated their level of agreement with assessments on a 9-point Likert scale. Item selection was based on acceptance by ≥ 66.6% of panel experts and the agreement of the scientific committee. In round 2, the same panelists evaluated non-consensus items in round 1.
Results
A total of 80 panelists participated in round 1; of these, 78 completed the round 2 survey. In round 1, 122 items from 4 dimensions (definition of fracture risk: 11 items, prevention and diagnosis: 38 items, choice of treatment: 24 items, and treatment-associated quality of life: 49 items) were evaluated. The consensus was reached for 90 items (73.8%). Panelists agreed that categorizing high risk, very high risk, or imminent risk determines secondary prevention actions (97.5%). Experts agreed that treatment with bone-forming drugs should be considered in case of a very high risk of fracture, and a sequential change to antiresorptive drugs should be made after 1–2 years (97.5%). Panelists also recommended corrective action plans for non-adherent patients to improve adherence (97.5%). A total of 131 items were finally accepted after round 2.
Conclusion
This Delphi study provides expert-based recommendations on clinical decision-making for managing patients with osteoporosis at high risk of fracture.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Osteoporosis is a skeletal disease characterized by decreased bone mass and deterioration of bone microarchitecture, leading to increased fragility and consequent fracture risk [1]. It is the most common metabolic bone disease in Western countries, affecting 25–32% of Spanish women over 50 years and almost 50% over 75 years [2]. In the absence of more recent updates on disease prevalence data, in 2010, it was estimated that in Spain, there were around 1.9 million cases in women and 400,000 cases in men, with an estimated cost of 2.8% of total health expenses [3, 4]. The incidence has increased in the following years, as well as the direct costs of incident fractures. Thus, in 2019, it was calculated that in Spain, there were around 2.9 million cases, of which almost 80% were women, with an estimated cost of 3.8% of total healthcare expending [5, 6].
Fragility fractures are a significant cause of disability, morbidity, and mortality in the population [7]. Hence the importance of prevention and early diagnosis, especially in patients at high risk and very high risk of fracture. A previous fracture due to this pathology, especially recent in time, is considered one of the risk factors identified for fragility fractures [8,9,10,11,12]. However, patients perceive them to be due to the environment or accidental falls. They often do not consider that they should be screened, observe preventive strategies, or even receive therapy for osteoporosis [13]. Recently, different studies analyzed the risks of imminent fractures in various cohorts in Spain [14, 15]. These studies showed that higher risks occur in women aged ≥ 80 years, and the 10-year risk levels were estimated between 1.8 and 21.5% in women and between 0.7 and 10.8% in men using the QFRACTURE tool [14]. More recently, an estimate of the incidence ratio of subsequent fractures in the 3 years following an initial fracture observed that 3.2% of women patients ≥ 50 years with a previous fracture experienced a new fracture every following year [15]. Despite all these data, underdiagnosis and under-treatment of osteoporosis are quite common, as is the case of vertebral fractures, which are the most frequent [16,17,18]. Vertebral fractures are often asymptomatic or mildly painful, and radiographic detection routines are scarce [17].
Optimal management of this patients’ profile has also impacted the quality of life of people with osteoporosis. Due to the presence of comorbidities, they are often polymedicated, usually associated with lower adherence to treatment, in addition to the dosage of the drugs and the side effects of the different therapies [19]. Indeed, patient follow-up program strategies are critical for those at increased risk of fracture to improve long-term treatment persistence and prevent secondary fractures [20,21,22].
Consequently, it is relevant to establish consensus among specialists in managing patients with osteoporosis to have effective strategies for prevention and diagnosis. It is also important to adapt the best therapy to each patient profile and to optimize the different lines of treatment over time, from the early stages of the disease. It is essential to consider the current availability of therapies, particularly biological treatments, in patients at high risk of fracture. In this sense, updated guidelines and recommendations from a number of organizations, including European Society for Clinical and Economic Aspects of Osteoporosis (ESCEO) and the International Osteoporosis Foundation (IOF) are available [23,24,25]. However, these recommendations need to be adapted locally, taking into account the characteristics of the population and the specific health resources at regional level.
Based on this background, we conducted the OSARIDELPHI study (from the Spanish title: Manejo clínico del paciente con OSteoporosis de Alto RIesgo de fractura en España: prevención y diagnóstico de la patología, opciones de tratamiento más adecuadas y su impacto en la calidad de vida del paciente). This Delphi method study addressed multiple key questions and controversies related to managing the patient with osteoporosis of high-risk fracture. A two-Delphi survey was used to seek expert-based opinions to develop a set of consensus guidelines that may support physicians in clinical decision-making and improve real-world practice in this patient’s profile.
Methods
Study design
The OSARIDELPHI project was a nationwide Spanish multicenter 2-round Delphi study to seek expert opinion on the clinical management of the patient with osteoporosis of high risk of fractures in Spain.
The approval of the Institutional Review Board (IRB) or by the equivalent ethics committee(s) was not required as this Delphi study does not involve human subject research. No patient data were collected for this study, which was based entirely on the feedback and opinions provided by experts.
The Delphi process is a widely accepted scientific method of systematic information collecting from a group of experts (termed the Delphi expert panel) on controversial or complex topics [26]. Each panel expert provides opinions individually and anonymously without the biasing effect of dominant individuals or group pressure [27,28,29]. The Delphi process ends when an agreement has been reached on the discussed topics.
The Delphi project was carried out in six steps: (1) literature review (by the scientific committee); (2) discussion and questionnaire domain/item generation by the scientific committee in a teleconference meeting; (3) selection of the Delphi expert panel and the invitation to candidates to participate in Delphi process; (4) domain/item set evaluation by the panel experts through 2 rounds using an online platform through a web platform (two-round Delphi approach); (5) final discussion of items that did not reach consensus in preceding rounds among the scientific committee experts; (6) final consensus analysis.
Delphi process
Selection of Delphi participants
The expert scientific committee comprised five specialists experienced in managing patients with osteoporosis of high-risk fracture and recognized experts in the field.
A total of 80 specialists from hospitals distributed across Spain were invited to participate in the project as members of the Delphi expert panel in both round 1 and round 2 of the Delphi process. The panel experts were selected based on their extensive experience and knowledge of the management of patients with osteoporosis.
The expert panel members were provided with an informative leaflet outlining the aims and the study procedure and including an electronic link to the online survey. Experts received personalized access to the online survey. Panel members participated in the project through 2 rounds of the Delphi process using the online questionnaire. The purpose of the expert panel was to reach a consensus based on the current clinical evidence and their daily practice in and knowledge of the management of patients with osteoporosis.
Selection of Delphi questionnaire dimensions and items
The scientific committee carried out a systematic literature review regarding managing patients with osteoporosis of high-risk fractures, focusing on current controversial and unmet topics. After a careful and critical review of the selected literature and based on their knowledge of the clinical management of the pathology, the scientific committee developed the first set of domains and items for the Delphi questionnaire in a teleconference meeting.
Round 1
The members of the Delphi expert panel were asked between July and September 2021 to rate their level of agreement with each questionnaire item on a 9-point Likert scale from 1 (completely disagree) to 9 (completely agree). Each item was categorized according to the scores as rejected (scores 1–3), undetermined (scores 4–6), or accepted (scores 7–9). Panelists were also encouraged to provide comments after scoring each item using open-text comment fields included in the online survey.
After analyzing the data obtained from the first Delphi round, the scientific committee experts participated in a teleconference meeting, where the Delphi survey results were presented and discussed. Item selection was based on the acceptance of questionnaire items by ≥ 66.6% of the expert panel and the agreement of the scientific committee. Statements not achieving 66.6% agreement were removed or modified according to the feedback provided by the expert panel. All statements were assessed, given the experts’ suggestions. After round 1 was completed and the expert comments had been summarized, amendments were made to some questionnaire items. Where necessary, new items were generated and included. The updated questionnaire was redistributed to the panelists for round 2.
Round 2
In round 2, the same panel members were asked between February and March 2022 to evaluate the list of items that did not meet consensus from round 1, using the same voting method described for the initial round. For this evaluation, the panel members were provided with a summary of the opinions issued anonymously by the participants in the first round, in addition to any other information that the scientific committee deemed appropriate to make available to the panelists to achieve consensus. Thus, the panelists could reflect upon the group’s responses after the first round and re-evaluate the non-consensus items in view of the other experts’ feedback.
After analysis of the responses as described for round 1, the statements not meeting expert agreement were retained for discussion.
Concluding round
The concluding round comprised a teleconference meeting among the scientific committee experts to assess those items that did not reach a consensus in round 2. The scientific committee members discussed the non-consensus items until an agreement was reached to retain or eliminate the item from the final consensus guidelines.
Statistical analysis
A descriptive statistical analysis of the data obtained from the expert panel’s assessment of the Delphi questionnaire items in rounds 1 and 2 was conducted. The distribution of frequencies of panel responses on the 9-point scale was calculated to establish the level of consensus for each questionnaire item.
A descriptive statistical analysis of the characteristics of the Delphi expert panel was also performed, including calculation of central tendency and dispersion (mean ± standard deviation, median and interquartile range) for quantitative variables and frequencies and valid percentages for qualitative variables.
The statistical analysis was performed using the Statistical Package for the Social Sciences (SPSS) version 18.0 (SPSS Inc., Chicago, IL, USA).
Results
Panel experts
A total of 80 specialists from 69 centers distributed throughout Spain agreed to participate as Delphi panel experts. All 80 experts participated in round 1, whereas 78 completed the round 2 survey. The characteristics of the Delphi experts are summarized in Table 1. Briefly, most participants were associate physicians (n = 73, 91.3%) in public hospitals (n = 74; 92.5%) with a median (range) of 16.5 (9.0–24.5) years of professional experience. Most participants were rheumatologists (n = 46; 57.5%), followed by traumatologists (n = 18; 22.5%). Remarkably, 34 (42.5%) and 20 (25.0%) participants indicated that they had in their hospitals a multidisciplinary team and fracture-liaison service, respectively, as resources related to the management of osteoporosis patients at risk of fracture.
Results from round 1 and round 2
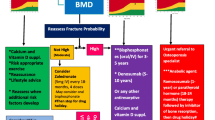
Figure 1 illustrates the results of the Delphi study. In round 1, panel members evaluated 122 items from the following dimensions:
-
1.
Definition of the profile of fracture risk: 11 items
-
2.
Prevention and diagnosis of osteoporosis with high and very high fracture risk: 38 items, subdivided into two sections (underdiagnosis and diagnosis/prevention)
-
3.
Choice of the most appropriate treatment for the specific patient profile: 24 items, subdivided into four sections (availability of current therapeutic alternatives, treatment lines/optimization, biosimilar availability, and adverse events)
-
4.
Treatment-associated quality of life in patients with osteoporosis at high risk of fracture: 49 items, subdivided into two sections (adherence to treatment and patient care programs)
The consensus was reached for 90 items (73.8%), which were accepted without modifications. After the scientific committee meeting, 4 items were removed, and 32 (14 non-consensus items and 18 items that had reached consensus) were reformulated and expanded to 92 items to be evaluated in round 2.
The items that did not meet consensus or were reformulated after round 1 were put forward for inclusion in round 2, along with accompanying comments. Forty-one items reached an agreement in round 2. Fifty-one items did not achieve consensus in round 2, and after discussion, the scientific committee experts decided to maintain non-consensus. At the end of the Delphi process, a total of 131 items were finally retained.
Supplementary Tables 1-4 summarize the results from the Delphi process and the level of agreement after the 2 rounds for the statements related to the definition of the fracture risk (Supplementary Table 1), the prevention and diagnosis of osteoporosis patients with high and very high–risk fracture risk (Supplementary Table 2), the choice of the appropriate therapy for the specific patient profile (Supplementary Table 3), and the treatment-associated quality of life in patients with a high risk of fracture (Supplementary Table 4).
To simplify the interpretation of the results, a set of recommendations was developed with the most relevant items that reached expert consensus in the Delphi process (Table 2).
Discussion
The results of this Delphi study demonstrate a high degree of consensus between experts involved in the management of osteoporosis patients with a high and very high–risk of fracture. This expert panel study provides insights on important topics such as the factors associated with more probability of high and very high–risk of fracture, the diagnosis, and the treatment choice for the specific patient profile.
Overall, according to the Delphi survey, experts agreed on the categorization of the fracture risk due to osteoporosis is determined by the history of previous osteoporotic fractures, a recent major fragility fracture, the family history of hip fracture, the treatment with high-dose glucocorticoids, the 10-year fracture risk probability, and the data of the bone densitometry. The IOF and ESCEO recommended that the risk of fracture should be expressed as absolute risk, i.e., the probability of fracture over a 10-year interval [30]. The assessment strategy to categorize risk was recently improved, including low and high risk and very high risk [23, 24]. Very high risk was defined as a fracture probability above the upper assessment threshold after a FRAX® assessment, including bone mineral density (BMD) if available [23, 24]. The panel of experts of the present Delphi survey also agreed that the implementation of secondary prevention actions is a consequence of this categorization. This result is in line with previous recommendations suggesting that preventive treatment given as soon as possible after fracturing in patients with very high fracture risk would avoid a higher number of new fractures and reduce the attendant morbidity [23, 24].
The panelists recommended follow-up systematically and proactively to the patients at high and very high risk of fracture (agreed in round 1 by 100% of the experts). In addition, experts, besides age and female gender, recommended including in the assessment the following risk factors: personal and parental history of fragility fractures (97.5%); history of treatment with glucocorticoids at doses ≥ 5 mg/day for ≥ 3 months (98.8%); causes of secondary osteoporosis (98.8%); history of falls in the past 3 months (92.5%), radiological imaging tests (92.5%); and BMD (97.5%). Another aspect related to risk assessment and categorization was recently highlighted as a new pivotal point in osteoporosis: FRAX® arithmetically integrating with novel risk factors [25, 31]. Accordingly, it is widely recognized that BMD alone for fracture risk assessment is less sensitive than algorithms, such as FRAX®, which incorporate risk indicators in addition to BMD [31].
The experts consistently agreed that treatment with bone-forming drugs should be considered in the very high fracture risk category. A sequential change to antiresorptive medications should be assessed after 1 or 2 years (97.5%). In addition, panelists recommended that the monitoring criteria for the different available alternatives of treatment should be defined by the improvement in bone densitometry (87.5%) and bone remodeling markers (78.8%), the absence of new fractures (93.8%), and the decrease in the risk of further fractures (95%). Regarding long-term treatment, panelists agreed that the duration of treatment with oral bisphosphonates, intravenous bisphosphonates, teriparatide, and denosumab is currently well defined at 5 (71.8%), 3 (69.2%), 2 (94.9%), and 10 years (71.8%).
Although osteoporosis is a well-recognized problem with a choice of widely available treatments, a large treatment gap exists [32]. Some treatments for osteoporosis (i.e., oral bisphosphonates, menopausal hormonal therapy, and selective estrogen receptor modulators) have suboptimal efficacy due to the difficulties in meeting treatment goals with such therapies in the highest fracture-risk patients [33]. The treatment stratification according to baseline fracture risk permits targeting the most effective treatments for this patient profile [34]. Specifically, guidance thresholds leading to distinguish high and very high fracture risk have optimized the use of anabolic agents [32, 34]. In patients at the highest fracture risk, treatment initiation with an anabolic (bone-forming) agent, such as teriparatide, followed by an antiresorptive to maintain the gains in bone mineral density, appears now a highly appropriate strategy to achieve a rapid and sustained reduction in fracture risk [8, 30]. In this sense, the availability of romosozumab and abaloparatide would be after the completion of the present Delphi study (conducted between July 2021 and March 2022). This recommendation has strong evidential support from recently published studies comparing anabolic vs. antiresorptive therapies [35,36,37,38]. These studies demonstrated a more rapid and significant fracture risk reduction with the former, compared with oral antiresorptive treatments alone [35,36,37,38]. However, these benefits must be maintained by following the anabolic with an antiresorptive drug [36, 39, 40]. Indeed, the evidence suggests that the treatment sequence is important, such that an anabolic agent given before an antiresorptive agent is more effective than the opposite sequence [41, 42]. This model suggests the need for physicians to be able to identify the patients who would most benefit from anabolic therapy [32].
The decision on when to stop antiresorptive treatment in a patient who has received a prior anabolic agent is complex. Recent data showed that discontinuation of up to a year might be acceptable in the case of previously alendronate- and zoledronate-treated patients [43]. In contrast, in the other agents (risedronate, ibandronate, raloxifene, teriparatide, denosumab, and romosozumab), the bone loss at the femoral neck and total hip is higher, indicating the importance of continuation of these antiresorptive agents [43]. Considering all these data, the consensus is that prolonged antiresorptive therapy will be necessary after anabolic treatment if the patient remains at high or very high fracture risk. However, if a patient is no longer at high or very high fracture risk, it may be possible to stop treatment for a maximum of 2 years, except for denosumab, due to the risk of rebound vertebral fractures [43].
This Delphi study also highlighted that experts agreed on the importance of the existence of fracture-liaison service (FLS) units in managing patients with high and very high fracture risk. According to experts, these FLS units should be composed of professionals who cover the different aspects crucial in managing this patient profile and with varying levels of specialization. However, there are differences in consensus regarding the current use of FLS (71.8% of the panelists stated that FLS units currently manage this patient profile) and what should happen (97.4% of the experts agreed that an FLS unit should monitor these patients). Notably, implementing FLS has increasing evidence since these units can improve access to better management and treatment to reduce future fractures [44,45,46]. A recent systematic review and meta-analysis evaluated the clinical impact of FLS implementation based on the results of several studies encompassing 48,045 patients [46]. The results suggested that FLS significantly improves the rates of dual-energy X-ray absorptiometry (DXA) scanning and antiresorptive therapy prescription and reduces new fracture rates [46]. Regarding possible controversies, a lack of consensus was observed on the current clinical application of bone remodeling markers in the early diagnosis of the disease (20.5%) and the prediction of fracture risk and bone mass loss (42.3%). The lack of consensus was also reflected in the simultaneous combination of a bone-forming drug with an antiresorptive treatment (50%). These results at the time of the Delphi survey reflect the absence of solid recommendations regarding using bone remodeling markers as a current diagnostic tool and of scientific evidence concerning the simultaneous combination of available therapies.
One of the limitations of the present study was a greater representation of specialists in rheumatology or traumatology among the participating experts compared to the rest of the specialties responsible for the management of patients with osteoporosis. Probably, the fact that there was no participation of primary care physicians explains the high percentage of experts in whose centers there were multidisciplinary units and FLS. In addition, it should be taken into account that the Delphi methodology itself means that the fact that experts express a high degree of agreement does not directly imply that a recommendation is necessarily effective. The results of the study represent the starting point for the development of recommendation documents and management guidelines. In summary, the results of this Delphi study suggested that, although the general lines of recommendations and suggestions are in line with the management recommendations established by, among others, the IOF or the ESCEO, an adaptation to the specific characteristics of the Spanish population and available health resources is needed. Specifically, regional differences can be observed in the implementation of FLS units, which may determine an adaptation of the recommendations to the Spanish reality.
Conclusion
The expert consensus recommendations derived from this Delphi panel study may provide support and guidance on clinical decision-making regarding osteoporosis management in specific clinical patients’ profiles, specifically in real-world patients with high or very high fracture risk. Additionally, this consensus analysis may encourage discussion on controversial issues addressed in the consensus statements.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Kanis JA, McCloskey EV, Johansson H, Oden A, Melton LJ 3rd, Khaltaev N (2008) A reference standard for the description of osteoporosis. Bone 42:467–475
Sanchez-Riera L, Wilson N, Kamalaraj N et al (2010) Osteoporosis and fragility fractures. Best Pract Res Clin Rheumatol 24:793–810
González Y, Sicras A, Larraínzar R, Sorio F, Canals L, Lizán L, Calvo E (2014) Estimación de los costes sanitarios relacionados con las fracturas osteoporóticas en pacientes posmenopáusicas en España. PharmacoEconomics Spanish Res Articles 12:1–9
Hernlund E, Svedbom A, Ivergard M, Compston J, Cooper C, Stenmark J, McCloskey EV, Jonsson B, Kanis JA (2013) Osteoporosis in the European Union: medical management, epidemiology and economic burden. A report prepared in collaboration with the International Osteoporosis Foundation (IOF) and the European Federation of Pharmaceutical Industry Associations (EFPIA). Arch Osteoporos 8:136
Kanis JA, Norton N, Harvey NC, Jacobson T, Johansson H, Lorentzon M, McCloskey EV, Willers C, Borgström F (2021) SCOPE 2021: a new scorecard for osteoporosis in Europe. Arch Osteoporos 16:82
Willers C, Norton N, Harvey NC, Jacobson T, Johansson H, Lorentzon M, McCloskey EV, Borgström F, Kanis JA (2022) Osteoporosis in Europe: a compendium of country-specific reports. Arch Osteoporos 17:23
Nazrun AS, Tzar MN, Mokhtar SA, Mohamed IN (2014) A systematic review of the outcomes of osteoporotic fracture patients after hospital discharge: morbidity, subsequent fractures, and mortality. Ther Clin Risk Manag 10:937–948
Kanis JA, Cooper C, Rizzoli R et al (2017) Identification and management of patients at increased risk of osteoporotic fracture: outcomes of an ESCEO expert consensus meeting. Osteoporos Int 28:2023–2034
Klotzbuecher CM, Ross PD, Landsman PB, Abbott TA 3rd, Berger M (2000) Patients with prior fractures have an increased risk of future fractures: a summary of the literature and statistical synthesis. J Bone Miner Res 15:721–739
Papaioannou A, Kennedy C (2015) Diagnostic criteria for osteoporosis should be expanded. Lancet Diabetes Endocrinol 3:234–236
Roux C, Briot K (2017) Imminent fracture risk. Osteoporos Int 28:1765–1769
van Geel TA, Huntjens KM, van den Bergh JP, Dinant GJ, Geusens PP (2010) Timing of subsequent fractures after an initial fracture. Curr Osteoporos Rep 8:118–122
Alami S, Hervouet L, Poiraudeau S, Briot K, Roux C (2016) Barriers to effective postmenopausal osteoporosis treatment: a qualitative study of patients’ and practitioners’ views. PLoS One 11(6):e0158365. https://doi.org/10.1371/journal.pone.0158365
González López-Valcárcel B, Sosa Henríquez M (2013) Estimate of the 10-year risk of osteoporotic fractures in the Spanish population. Med Clin 140:104–109
Montoya-García MJ, Giner M, Marcos R, García-Romero D, Olmo-Montes FJ, Miranda MJ, Hernández-Cruz B, Colmenero MA, Vázquez-Gámez MA (2021) Fragility fractures and imminent fracture risk in the spanish population: a retrospective observational cohort study. J Clin Med. 10(5):1082. https://doi.org/10.3390/jcm10051082
Herrera A, Carpintero P, Fernandez-Perez C, Hernandez-Vaquero D, Martinez AA, Suso S (2011) Secondary prevention in osteoporotic fractures. The GIOS project. Medicina clinica 136:50–55
Delmas PD, van de Langerijt L, Watts NB, Eastell R, Genant H, Grauer A, Cahall DL (2005) Underdiagnosis of vertebral fractures is a worldwide problem: the IMPACT study. J Bone Miner Res 20:557–563
Sanfélix-Genovés J, Reig-Molla B, Sanfélix-Gimeno G, Peiró S, Graells-Ferrer M, Vega-Martínez M, Giner V (2010) The population-based prevalence of osteoporotic vertebral fracture and densitometric osteoporosis in postmenopausal women over 50 in Valencia, Spain (the FRAVO study). Bone 47:610–616
Yeam CT, Chia S, Tan HCC, Kwan YH, Fong W, Seng JJB (2018) A systematic review of factors affecting medication adherence among patients with osteoporosis. Osteoporos Int 29:2623–2637
Naranjo A, Molina A, Quevedo A, Rubino FJ, Sanchez-Alonso F, Rodriguez-Lozano C, Ojeda S (2021) Fracture liaison service model: treatment persistence 5 years later. Arch Osteoporos 16:60
Naranjo A, Ojeda S, Giner M et al (2020) Best practice framework of fracture liaison services in Spain and their coordination with primary care. Arch Osteoporos 15:020–0693
Noordin S, Allana S, Masri BA (2018) Establishing a hospital based fracture liaison service to prevent secondary insufficiency fractures. Int J Surg 54:328–332
Kanis JA, Harvey NC, McCloskey E et al (2020) Algorithm for the management of patients at low, high and very high risk of osteoporotic fractures. Osteoporos Int 31:1–12
Kanis JA, Johansson H, Harvey NC, Lorentzon M, Liu E, Vandenput L, McCloskey EV (2021) An assessment of intervention thresholds for very high fracture risk applied to the NOGG guidelines : a report for the National Osteoporosis Guideline Group (NOGG). Osteoporos Int 32:1951–1960
Kanis JA, McCloskey EV, Harvey NC, Cooper C, Rizzoli R, Dawson-Hughes B, Maggi S, Reginster JY (2022) Intervention thresholds and diagnostic thresholds in the management of osteoporosis. Aging Clin Exp Res 34(12):3155–3157. https://doi.org/10.1007/s40520-022-02216-7
Yeh JS, Van Hoof TJ, Fischer MA (2016) Key features of academic detailing: development of an expert consensus using the delphi method. Am Health Drug Benefits 9:42–50
Hasson F, Keeney S, McKenna H (2000) Research guidelines for the Delphi survey technique. J Adv Nurs 32:1008–1015
James D, Warren-Forward H (2015) Research methods for formal consensus development. Nurse Res 22:35–40
Keeney S, Hasson F, McKenna H (2006) Consulting the oracle: ten lessons from using the Delphi technique in nursing research. J Adv Nurs 53:205–212
Kanis JA, Cooper C, Rizzoli R, Reginster JY (2019) European guidance for the diagnosis and management of osteoporosis in postmenopausal women. Osteoporos Int 30:3–44
Kanis JA, Harvey NC, Johansson H, Liu E, Vandenput L, Lorentzon M, Leslie WD, McCloskey EV (2020) A decade of FRAX: how has it changed the management of osteoporosis? Aging Clin Exp Res 32:187–196
Curtis EM, Reginster JY, Al-Daghri N et al (2022) Management of patients at very high risk of osteoporotic fractures through sequential treatments. Aging Clin Exp Res 34:695–714
Cummings SR, Cosman F, Lewiecki EM et al (2017) Goal-directed treatment for osteoporosis: a progress report from the ASBMR-NOF Working Group on Goal-Directed Treatment for Osteoporosis. J Bone Miner Res 32:3–10
McClung MR (2021) Role of bone-forming agents in the management of osteoporosis. Aging Clin Exp Res 33:775–791
Kendler DL, Marin F, Zerbini CAF et al (2018) Effects of teriparatide and risedronate on new fractures in post-menopausal women with severe osteoporosis (VERO): a multicentre, double-blind, double-dummy, randomised controlled trial. Lancet 391:230–240
Saag KG, Petersen J, Brandi ML, Karaplis AC, Lorentzon M, Thomas T, Maddox J, Fan M, Meisner PD, Grauer A (2017) Romosozumab or alendronate for fracture prevention in women with osteoporosis. N Engl J Med 377:1417–1427
Barrionuevo P, Kapoor E, Asi N et al (2019) Efficacy of pharmacological therapies for the prevention of fractures in postmenopausal women: a network meta-analysis. J Clin Endocrinol Metab 104:1623–1630
Díez-Pérez A, Marin F, Eriksen EF, Kendler DL, Krege JH, Delgado-Rodríguez M (2019) Effects of teriparatide on hip and upper limb fractures in patients with osteoporosis: a systematic review and meta-analysis. Bone 120:1–8
Bone HG, Cosman F, Miller PD et al (2018) ACTIVExtend: 24 months of alendronate after 18 months of abaloparatide or placebo for postmenopausal osteoporosis. J Clin Endocrinol Metab 103:2949–2957
Leder BZ, Tsai JN, Neer RM, Uihlein AV, Wallace PM, Burnett-Bowie SA (2016) Response to therapy with teriparatide, denosumab, or both in postmenopausal women in the DATA (Denosumab and Teriparatide Administration) study randomized controlled trial. J Clin Densitom 19:346–351
Cosman F, Nieves JW, Dempster DW (2017) Treatment sequence matters: anabolic and antiresorptive therapy for osteoporosis. J Bone Miner Res 32:198–202
Kanis JA, Cooper C, Rizzoli R, Reginster JY (2018) Review of the guideline of the American College of Physicians on the treatment of osteoporosis. Osteoporos Int 29:1505–1510
Elbers LPB, Raterman HG, Lems WF (2021) Bone mineral density loss and fracture risk after discontinuation of anti-osteoporotic drug treatment: a narrative review. Drugs 81:1645–1655
Javaid MK, Kyer C, Mitchell PJ et al (2015) Effective secondary fracture prevention: implementation of a global benchmarking of clinical quality using the IOF Capture the Fracture® Best Practice Framework tool. Osteoporos Int 26:2573–2578
Javaid MK (2021) Efficacy and efficiency of fracture liaison services to reduce the risk of recurrent osteoporotic fractures. Aging Clin Exp Res 33:2061–2067
Barton DW, Piple AS, Smith CT, Moskal SA, Carmouche JJ (2021) The clinical impact of fracture liaison services: a systematic review. Geriatr Orthop Surg Rehabil 12:2151459320979978
Acknowledgements
The authors acknowledge Lola Andreu-Pérez and Alicia Subtil-Rodríguez (both from Evidenze Clinical Research) for project coordination and editorial assistance based on the authors’ input.
Funding
This study was sponsored by STADA.
Author information
Authors and Affiliations
Contributions
All authors contributed to study conception, design, analysis, interpretation of the data, and drafting and revising the article.
Corresponding author
Ethics declarations
Conflicts of interest
L.A. and M.M.-R. declare no conflict of interest. J.M.C.-T. has received consulting, speaking, and lecture fees from Amgen, UCB, Lilly, and Gedeon-Richter and consulting fees from STADA. C.C. is an employee of STADA. E.D.-M. has received consulting, speaking and lecture, and travel reimbursement fees from Amgen, Lilly, STADA, and UCB. J.M.O. has received consulting and lecture fees from Gedeon-Richter and UCB, consulting fees from FAES and STADA, and lecture fees from Amgen, Lilly, and Grünenthal.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Arboleya, L., Cancio-Trujillo, J.M., Chaves, C. et al. A Delphi consensus on the management of Spanish patients with osteoporosis at high risk of fracture: OSARIDELPHI study. Arch Osteoporos 18, 110 (2023). https://doi.org/10.1007/s11657-023-01318-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11657-023-01318-7