Abstract
Objective
To investigate the effects of Weikang Capsule (胃康胶囊, WKC) on aspirin-related gastric and small intestinal mucosal injury by magnetically controlled capsule endoscopy (MCCE).
Methods
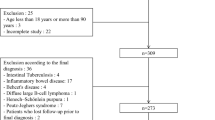
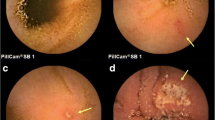
Patients taking enteric-coated aspirin aged 40–75 years were enrolled in Beijing Anzhen Hospital, Capital Medical University from January 2019 to December 2019. The patients continued taking aspirin Tablet (100 mg per day) and underwent MCCE before and after 1-month combined treatment with WKC (0.9 g per time orally, 3 times per day). The gastrointestinal symptom score, gastric Lanza score, the duodenal, jejunal and ileal mucosal injury scores were used to evaluate the gastrointestinal injury before and after treatment. Adverse events including nausea, vomiting, abdominal pain, abdominal distension, abdominal discomfort, dizziness, or headache during MCCE and combined treatment were observed and recorded.
Results
Twenty-two patients (male/female, 13/9) taking enteric-coated aspirin aged 59.5 ± 11.3 years with a duration of aspirin use of 28.0 (1.0, 48.0) months were recruited. Compared with pre-treatment, the gastrointestinal symptom rating scale scores, gastric Lanza scores, and duodenal mucosal injury scores were significantly reduced after 1-month WKC treatment (P<0.05), and jejunal and ileal mucosal injury scores showed no obvious change. No adverse events occurred during the trial.
Conclusions
WKC can alleviate gastrointestinal symptoms, as well as gastric and duodenal mucosal injuries, in patients taking enteric-coated aspirin; it does not aggravate jejunal or ileal mucosal injury, which may be an effective alternative for these patients (Clinical trial registry No. ChiCTR1900025451).
Similar content being viewed by others
References
Iwamoto J, Saito Y, Honda A, Matsuzaki Y. Clinical features of gastroduodenal injury associated with long-term low-dose aspirin therapy. World J Gastroenterol 2013;19:1673–1682.
Hiraishi H, Shimada T, Terano A. Involvement of oxidative stress in the pathogenesis of NSAID-induced gastric mucosal damage. J Gastroenterol 2000;35:567–569.
Endo H, Sakai E, Taniguchi L, Kessoku T, Komiya Y, Ezuka A, et al. Risk factors for small-bowel mucosal breaks in chronic low-dose aspirin users: data from a prospective multicenter capsule endoscopy registry. Gastrointest Endosc 2014;80:826–834.
Chen WC, Lin KH, Huang YT, Tsai TJ, Sun WC, Chuah SK, et al. The risk of lower gastrointestinal bleeding in low-dose aspirin user. Aliment Pharmacol Ther 2017;45:1542–1550.
Wallace JL. Mechanisms, prevention and clinical implications of nonsteroidal anti-inflammatory drug-enteropathy. World J Gastroenterol 2013;19:1861–1876.
Lué A, Lanas A. Protons pump inhibitor treatment and lower gastrointestinal bleeding: balancing risks and benefits. World J Gastroenterol 2016;22:10477–10481.
Chinese Expert Recommendation on Prevention of Gastrointestinal Injury Related to Antithrombotic Therapy group. The Chinese expert recommendation on prevention of gastrointestinal injury related to antithrombotic therapy. Chin J Intern Med (Chin) 2016;55:564–567.
Yasuda S, Nakao K, Nishimura K, Miyamoto Y, Sumita Y, Shishido T, et al. The current status of cardiovascular medicine in Japan—analysis of a large number of health records from a nationwide claim-based database, JROAD-DPC. Circ J 2016;80:2327–2335.
Endo H, Sakai E, Kato T, Umezawa S, Higurashi T, Ohkubo H, et al. Small bowel injury in low-dose aspirin users. J Gastroenterol 2015;50:378–386.
Geriatrics Branch of Chinese Medical Association, Editorial Board of Chinese Journal of Internal Medicine, Editorial Board of Chinese Journal of Geriatrics. Aspirin use in patients with atherosclerotic cardiovascular disease: the 2016 Chinese expert consensus statement. Chin J Intern Med (Chin) 2017;56:68–80.
Chen X, Gao F, Zhang J. Screening for gastric and small intestinal mucosal injury with magnetically controlled capsule endoscopy in asymptomatic patients taking enteric-coated aspirin. Gastroenterol Res Pract 2018;2018:2524689.
Lanza FK, Collaku A, Liu DJ. Endoscopic comparison of gastroduodenal injury with over-the-counter doses of new fast-dissolving ibuprofen and paracetamol formulations: a randomized, placebo-controlled, 4-way crossover clinical trial. Clin Exp Gastroenterol 2018;11:169–177.
Gao F, Chen X, Zhang J. Prevalence of gastric and small intestinal mucosal injury in elderly patients taking enteric-coated aspirin by magnetically controlled capsule endoscopy. Gastroenterol Res Pract 2019;2019:1582590.
Suzuki T, Masui A, Nakamura J, Shiozawa H, Aoki J, Nakae H, et al. Yogurt containing Lactobacillus gasseri mitigates aspirin-induced small bowel injuries: a prospective, randomized, double-blind, placebo-controlled trial. Digestion 2017;95:49–54.
Ding RF, Guo YH, Han WP, Wang AY, Xie LP, Zhao PC. Preventive effects of Jinghua Weikang Capsule on NSAID-induced injury to the mucosa of the small intestine: an experimental research. Chin J Integr Tradit West Med (Chin) 2013;33:522–525.
Fan TY, Feng QQ, Jia CR, Fan Q, Li CA, Bai XL. Protective effect of Weikang Decoction and partial ingredients on model rat with gastric mucosa ulcer. World J Gastroenterol 2005;11:1204–1209.
Takeuchi K, Satoh H. NSAID-induced small intestinal damage—roles of various pathogenic factors. Digestion 2015;91:218–232.
Otani K, Tanigawa T, Watanabe T, Shimada S, Nadatani Y, Nagami Y, et al. Microbiota plays a key role in non-steroidal anti-inflammatory drug-induced small intestinal damage. Digestion 2017;95:22–28.
Watanabe T, Tanigawa T, Nadatani Y, Otani K, Machida H, Okazaki H, et al. Mitochondrial disorders in NSAIDs-induced small bowel injury. J Clin Biochem Nutr 2011;48:117–121.
Tai FWD, McAlindon ME. NSAIDs and the small bowel. Curr Opin Gastroenterol 2018;34:175–182.
Nadatani Y, Watanabe T, Suda W, Nakata A, Matsumoto Y, Kosaka S, et al. Gastric acid inhibitor aggravates indomethacin-induced small intestinal injury via reducing Lactobacillus johnsonii. Sci Rep 2019;9:17490.
Acknowledgments
We thank all of the subjects for their participation in this study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by Beijing Chinese Medicine Science and Technology Development Fund (No. JJ2018–02)
Conflict of Interest
The authors declare that they have no competing interests.
Author Contributions
Du L was responsible for drafting the manuscript and interpreting the data. Gao F was responsible for data collection and analyses. Zhang J was responsible for the study design and revision of the manuscript.
Rights and permissions
About this article
Cite this article
Du, L., Gao, F. & Zhang, J. Effect of Weikang Capsule (胃康胶囊) on Aspirin-Related Gastric and Small Intestinal Mucosal Injury. Chin. J. Integr. Med. 27, 621–625 (2021). https://doi.org/10.1007/s11655-021-3300-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11655-021-3300-2