Abstract
Objective
To assess the beneficial potential of ethanolic extract of avocado fruit on abnormal carbohydrate metabolic key enzymes in hepatic and renal tissues in streptozotocin (STZ) induced hyperglycemic albino rats.
Methods
Twenty-four male albino rats were randomly divided into four groups with six in each group by simple random sampling method. Group 1 as control rats; Group 2 as STZ induced diabetic rats; Group 3 as diabetic rats treated with avocado fruit extract (AFE), 300 mg/kg as aqueous suspension orally for 30 days; Group 4 as diabetic rats treated with gliclazide (5 mg/kg) in aqueous solution orally for 30 days. The rats were fasted overnight and sacrificed by cervical dislocation and the blood was collected for various biochemical analysis and excision of hepatic and kidney were done for histological analysis. Levels of fasting blood glucose, plasma insulin and glycosylated hemoglobin were estimated. The activities of key enzymes in carbohydrate metabolism such as hexokinase, pyruvate kinase, glucose-6- phosphatase, fructose-1, 6-bisphosphatase, glucose-6-phosphate dehydrogenase, glycogen synthase and glycogen phosphorylase were assayed by standard methods described in the methodology.
Results
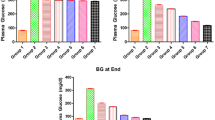
Oral administration of AFE significantly improved the altered levels of blood glucose, plasma insulin, glycosylated hemoglobin, and modulated the activities of carbohydrate metabolizing enzymes (P<0.05, respectively). The glycogen content in hepatic tissues was significantly increased in diabetic rats treated with AFE (P<0.05, respectively).
Conclusion
AFE plays a pivotal role to maintain normoglycemia in diabetes by modulating the activities of carbohydrate metabolic enzymes.
Similar content being viewed by others
References
Scheen AJ. Drug treatment of non-insulin-dependent diabetes mellitus in the 1990s. Achievements and future developments. Drugs 1997;54:355–368.
Wild S, Roglic G, Green A, Sicree R, King H. Global prevalence of diabetes: estimates for the year 2000 and projections for 2030. Diabetes Care 2004;27:1047–1053.
Villanueva M, Verti S, “Avocado: Green gold mexico, michoacan pride”. Government of the State of Michoacan. Mexico;2007.
Barry PC. Avocado: the Early Roots of Avocado History. Canku Ota 2001;33:12–29.
SK Lee, RE Young, PM Schiffman, and CW Coggins. Jr. Maturity Studies of Avocado Fruit Based on Picking Dates and Dry Weight. J Amer Soc Hort Sci 1983;108:390–394.
Stradley L. “All about avocados: history of the hass Avocado”. What’s Cooking America.net. Newberg, Yamhill County, Oregon: United States; 2004
Kadam SS, Salunkhe, DK. Avocado. In: Handbook of Fruit Science and Technology, Production, Composition, Storage and Processing. New York: Marcel Dekker Inc.;1995:363.
Rakieten N, Rakieten ML, Nadkarni MR. Studies on the diabetogenic action of streptozotocin. Cancer Chemother Reports 29;1963:91–98.
Fischer LJ, Rickert DE. Pancreatic islet-cell toxicity. Crit Rev in Toxicol 1975;3:231–263.
Trinder P. Determination of blood glucose using an oxidaseperoxidase system with a non-carcinogenic chromogen. J Clin Pathol 1969;22:158–161.
Nayak SS, Pattabiraman TN. A new colorimetric method for the estimation of glycosylated hemoglobin. Clin Chim Acta 1981;109:267–274.
Brandstrup N, Kirk JE, Bruni C. The hexokinase and phosphoglucoisomerase activities of aortic and pulmonary artery tissue in individuals of various ages. J Gerontol 1957;12:166–171.
Pogson CI, Denton RM. Effect of alloxan diabetes, starvation and refeeding on glycolytic kinase activities in rat epididymal adipose tissue. Nature 1967;216:156–157.
Koide H, Oda T. Pathological occurrence of glucose-6- phosphatase in serum in liver diseases. Clin Chim Acta 1959;4:554–561.
Gancedo JM, Gancedo C. Fructose-1, 6 diphosphatase, phosphofructokinase and glucose-6-phosphate dehydrogenase from fermenting and non-fermenting yeasts. Arch Mikrobiol 1971;76:132–138.
Ells HA, Kirkman HN. A colorimetric method for assay of erythrocytic glucose-6-phosphate dehydrogenase. Proc Soci Exp Biol Med 1961;106:607–609.
Leloir LF, Goldemberg SH. Glycogen synthetase from rat liver: (Glucose) n+(UDPG) ? (Glucose) n+1+UDP. In: Colowick SP, Kalpan NO (Eds.). Methods in Enzymology. New York: Academic Press;1962:145–147.
Cornblath M, Randle PJ, Parmeggiani A, Morgan HE. Regulation of glycogenolysis in muscle. Effects of glucagon and anoxia on lactate production, glycogen content, and phosphorylase activity in the perfused isolated rat heart. J Biolo Chem 1963;238:1592–1597.
Morales MA, Jabbagy AJ, Terenizi HR. Mutations affecting accumulation of glycogen. Neurospora News 1973;20:24–25.
Soling HD, Kleineke J. Species dependent regulation of hepatic gluconeogenesis in higher animals. In: Hanson RW, Mehlman MA eds. Gluconeogenesis: its regulation in mammalian species. New York: Wiley Interscience: 369-462.
Grover JK, Vats V. Shifting paradigm from conventional to alternative medicine. An introduction on traditional Indian medicine. Asia Pac Biotechnol News 2001;5:28–32.
Koenig RJ, Peterson CM, Jones RL, Saudek C, Lehrman, M, et al. Correlation of glucose regulation and hemoglobin AIc in diabetes mellitus. N Engl J Med 1976;295:417–420.
Asgary S, Naderi G, Sarrafzadegan N, Ghassemi N, Boshtam M, Rafie M, et al. Anti-oxidant effect of flavonoids on hemoglobin glycosylation. Pharm Acta Helv 1999;73:223–226.
Al-yassin D, Ibrahim K. A minor haemoglobin fraction and the level of fasting blood glucose. J Faculty Med 1981;23:373–380.
Nordlie RC, Foster JD, Lange AJ. Regulation of glucose production by the liver. Annu Rev Nutr 1999;19:379–406.
Shirwaikar A, Rajendran K, Barik R. Effect of aqueous bark extract of Garuga pinnata Roxb. In streptozotocin–nicotinamide induced type-? diabetes mellitus. J Ethnopharmacol 2006;107:285–290.
Gupta D, Raju J, Prakash J, Baquer NZ. Change in the lipid profile, lipogenic and related enzymes in the livers of experimental diabetic rats: effect of insulin and vanadate. Diabetes Rese Clin Pract 1999;46:1–7.
Postic C, Shiota M, Magnuson MA. Cell-specific roles of glucokinase in glucose homeostasis. Recent Prog Horm Res 2001;56:195–217.
Taylor R, Agius L. The biochemistry of diabetes. Biochem J 1988;250:625–640.
Roden M, Bernroider E. Hepatic glucose metabolism in humans-its role in health and disease. Best Prac Res: Clin Endocrinol Metab 2003;17:365–383.
Pilkis SJ, Claus TH. Hepatic gluconeogenesis/glycolysis: regulation and structure/function relationships of substrate cycle enzymes. Annu Rev Nutri 1991;11;465–515.
Xu Y, Osborne BW, Stanton RC. Diabetes causes inhibition of glucose- 6-phosphate dehydrogenase via activation of PKA, which contributes to oxidative stress in rat kidney cortex. Am J Physiol Ren Physiol 2005;289:F1040–1047.
West IC. Glucose-6-phosphate dehydrogenase: a candidate gene for diabetes. Diabet Med 2002;19:172–174.
Pederson BA, Schroeder JM, Parker GE, Smith MW, DePaoli-Roach AA, Roach PJ. Glucose metabolism in mice lacking muscle glycogen synthase. Diabet 2005;54:3466–3473.
Roesler WJ, Khandelwal RL. Quantification of glycogen synthase and phosphorylase protein in mouse liver: correlation between enzymatic protein and enzyme activity. Archives Biochem Biophys 1986;244:397–407.
Parker G, Taylor R, Jones D, McClain D. Hyperglycemia and inhibition of glycogen synthase in streptozotocintreated mice: role of O-linked N acetylglucosamine. J Biol Chem 2004;279:20636–20642.
Author information
Authors and Affiliations
Corresponding author
Additional information
Conflict of Interest None.
Rights and permissions
About this article
Cite this article
Mahadeva Rao, U.S. Salutary potential of ethanolic extract of avocado fruit on anomalous carbohydrate metabolic key enzymes in hepatic and renal tissues of hyperglycaemic albino rats. Chin. J. Integr. Med. (2017). https://doi.org/10.1007/s11655-017-2784-2
Received:
Published:
DOI: https://doi.org/10.1007/s11655-017-2784-2