Abstract
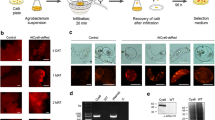
The FDA-approved anti-cancer compound paclitaxel is currently produced commercially by Taxus plant cell suspension cultures. One major limitation to the use of plant cell culture as a production platform is the low and variable product yields. Therefore, methods to increase and stabilize paclitaxel production are necessary to ensure product security, especially as the demand for paclitaxel continues to rise. Although a stable transformation method for Taxus suspension cultures has been developed, stable transformant yields are low (around 1% of experiments) and the method does not translate to the Taxus cuspidata Siebold and Zucc. and Taxus canadensis Marshall cell lines utilized in this study. Therefore, a new method for Agrobacterium-mediated transformation of Taxus callus and suspension cultures was developed through identification of the optimal Agrobacterium strain, inclusion of an anti-necrotic cocktail (silver nitrate, cysteine, and ascorbic acid) and increased recovery time for cells after cocultivation, the time following infection with Agrobacterium tumefaciens. Application of the increased recovery time to transformation of T. cuspidata line PO93XC resulted in 200 calluses staining positive for GUS. Additionally, two transgenic lines have been maintained with stable transgene expression for over 5 yr. This method represents an improvement over existing transformation methods for Taxus cultures and can be applied for future metabolic engineering efforts.







Similar content being viewed by others
References
Akcay UC, Mahmoudian M, Kamci H, Yucel M, Oktem HA (2009) Agrobacterium tumefaciens-mediated genetic transformation of a recalcitrant grain legume, lentil (Lens culinaris Medik). Plant Cell Rep 28(3):407–417. https://doi.org/10.1007/s00299-008-0652-4
Altunkaya A, Gökmen V (2008) Effect of various inhibitors on enzymatic browning, antioxidant activity and total phenol content of fresh lettuce (Lactuca sativa). Food Chem 107(3):1173–1179. https://doi.org/10.1016/j.foodchem.2007.09.046
Cragg GM, Newman DJ (2009) Nature: a vital source of leads for anticancer drug development. Phytochem Rev 8(2):313–331. https://doi.org/10.1007/s11101-009-9123-y
Cusido RM, Onrubia M, Sabater-Jara AB, Moyano E, Bonfill M, Goossens A, Pedreño MA, Palazon J (2014) A rational approach to improving the biotechnological production of taxanes in plant cell cultures of Taxus spp. Biotechnol Adv 32(6):1157–1167. https://doi.org/10.1016/j.biotechadv.2014.03.002
Dalton S, Bettany A, Timms E, Morris P (1995) The effect of selection pressure on transformation frequency and copy number in transgenic plants of tall fescue (Festuca arundinacea Schreb.) Plant Sci 108(1):63–70. https://doi.org/10.1016/0168-9452(95)04127-G
Dong Y, Duan W, He H, Su P, Zhang M, Song G, Fu C, Yu L (2015) Enhancing taxane biosynthesis in cell suspension culture of Taxus chinensis by overexpressing the neutral/alkaline invertase gene. Process Biochem 50(4):651–660. https://doi.org/10.1016/j.procbio.2015.01.018
Enríquez-Obregón GA, Prieto-Samsónov DL, de la Riva GA, Pérez M, Selman-Housein G, Vázquez-Padrón RI (1999) Agrobacterium-mediated Japonica rice transformation: a procedure assisted by an antinecrotic treatment. Plant Cell Tissue Organ Cult 59(3):159–168. https://doi.org/10.1023/A:1006307527449
Fettneto AG, Zhang WY, Dicosmo F (1994) Kinetics of taxol production, growth, and nutrient uptake in cell suspenesions of Taxus cuspidata. Biotechnol Bioeng 44(2):205–210. https://doi.org/10.1002/bit.260440209
Jones AMP, Saxena PK (2013) Inhibition of phenylpropanoid biosynthesis in Artemisia annua L.: a novel approach to reduce oxidative browning in plant tissue culture. PLoS One 8(10):e76802. https://doi.org/10.1371/journal.pone.0076802
Ketchum REB, Wherland L, Croteau RB (2007) Stable transformation and long-term maintenance of transgenic Taxus cell suspension cultures. Plant Cell Rep 26(7):1025–1033. https://doi.org/10.1007/s00299-007-0323-x
Kolewe ME, Henson MA, Roberts SC (2010) Characterization of aggregate size in Taxus suspension cell culture. Plant Cell Rep 29(5):485–494. https://doi.org/10.1007/s00299-010-0837-5
Kolewe ME, Henson MA, Roberts SC (2011) Analysis of aggregate size as a process variable affecting paclitaxel accumulation in Taxus suspension cultures. Biotechnol Prog 27(5):1365–1372. https://doi.org/10.1002/btpr.655
Krügel T, Lim M, Gase K, Halitschke R, Baldwin IT (2002) Agrobacterium-mediated transformation of Nicotiana attenuata, a model ecological expression system. Chemoecology 12(4):177–183. https://doi.org/10.1007/PL00012666
Li FL, Ma XJ, XL H, Hoffman A, Dai JG, Qiu DY (2011) Antisense-induced suppression of taxoid 14 beta-hydroxylase gene expression in transgenic Taxus x media cells. Afr J Biotechnol 10:8720–8728
Li S, Zhang P, Zhang M, Fu C, Yu L (2013) Functional analysis of a WRKY transcription factor involved in transcriptional activation of the DBAT gene in Taxus chinensis. Plant Biol 15(1):19–26. https://doi.org/10.1111/j.1438-8677.2012.00611.x
Li ST, CH F, Zhang M, Zhang Y, Xie S, LJ Y (2012) Enhancing taxol biosynthesis by overexpressing a 9-cis-epoxycarotenoid dioxygenase gene in transgenic cell lines of Taxus chinensis. Plant Mol Biol Rep 30(5):1125–1130. https://doi.org/10.1007/s11105-012-0436-4
Opabode JT (2006) Agrobacterium-mediated transformation of plants: emerging factors that influence efficiency. Biotechnol Mol Biol Rev 1:12–20
Patil RA, Kolewe ME, Normanly J, Walker EL, Roberts SC (2012) Contribution of taxane biosynthetic pathway gene expression to observed variability in paclitaxel accumulation in Taxus suspension cultures. Biotechnol J 7(3):418–427. https://doi.org/10.1002/biot.201100183
Patil RA, Kolewe ME, Roberts SC (2013) Cellular aggregation is a key parameter associated with long term variability in paclitaxel accumulation in Taxus suspension cultures. Plant Cell Tissue Organ Cult 112(3):303–310. https://doi.org/10.1007/s11240-012-0237-3
Shekhawat UKS, Ganapathi TR, Srinivas L, Bapat VA, Rathore TS (2008) Agrobacterium-mediated genetic transformation of embryogenic cell suspension cultures of Santalum album L. Plant Cell Tissue Organ Cult 92(3):261–271. https://doi.org/10.1007/s11240-007-9330-4
Song GQ, Sink KC (2004) Agrobacterium tumefaciens-mediated transformation of blueberry (Vaccinium corymbosum L.) Plant Cell Rep 23(7):475–484. https://doi.org/10.1007/s00299-004-0842-7
Vaucheret H, Beclin C, Elmayan T, Feuerbach F, Godon C, Morel JB, Mourrain P, Palauqui JC, Vernhettes S (1998) Transgene-induced gene silencing in plants. Plant J 16(6):651–659. https://doi.org/10.1046/j.1365-313x.1998.00337.x
Vongpaseuth K, Roberts SC (2007) Advancements in the understanding of paclitaxel metabolism in tissue culture. Curr Pharm Biotechnol 8(4):219–236. https://doi.org/10.2174/138920107781387393
Wilson SA, Cummings EM, Roberts SC (2014) Multi-scale engineering of plant cell cultures for promotion of specialized metabolism. Curr Opin Biotechnol 29:163–170. https://doi.org/10.1016/j.copbio.2014.07.001
Wilson SA, Roberts SC (2012) Recent advances towards development and commercialization of plant cell culture processes for the synthesis of biomolecules. Plant Biotechnol J 10(3):249–268. https://doi.org/10.1111/j.1467-7652.2011.00664.x
Zhang D, Yang R, Wang S, Dong Z (2014) Paclitaxel: new uses for an old drug. Drug Des Devel Ther 8:279–284. https://doi.org/10.2147/DDDT.S56801
Zhang P, Li ST, Liu TT, CH F, Zhou PP, Zhao CF, LJ Y (2011) Overexpression of a 10-deacetylbaccatin III-10 beta-O-acetyltransferase gene leads to increased taxol yield in cells of Taxus chinensis. Plant Cell Tissue Organ Cult 106(1):63–70. https://doi.org/10.1007/s11240-010-9894-2
Zhang X, Henriques R, Lin S-S, Niu Q-W, Chua N-H (2006) Agrobacterium-mediated transformation of Arabidopsis thaliana using the floral dip method. Nat Protoc 1(2):641–646. https://doi.org/10.1038/nprot.2006.97
Acknowledgements
Wilson acknowledges the support of the National Science Foundation-sponsored Institute for Cellular Engineering IGERT program (DGE-0654128). Roberts also acknowledges support from the National Institutes of Health (RO1 GM070852).
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor: Todd Jones
Rights and permissions
About this article
Cite this article
Wilson, S.A., Keen, P., McKee, M.C. et al. Development of an Agrobacterium-mediated transformation method for Taxus suspension cultures. In Vitro Cell.Dev.Biol.-Plant 54, 36–44 (2018). https://doi.org/10.1007/s11627-017-9876-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11627-017-9876-8