Abstract
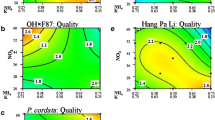
Mineral nutrition in the media used for growth of in vitro plants is often difficult to optimize due to complex chemical interactions of required nutrients. The response of plant tissue to standard growth media varies widely due to the genetic diversity of the plant species studied. This study was designed as the initial step in determining the optimal mineral nutrient requirements for micropropagation of shoot tips from a collection of genetically diverse pear germplasm. Five mineral nutrient factors were defined from Murashige and Skoog salts: NH4NO3, KNO3, mesos (CaCl2·2H20–KH2PO4–MgSO4), micronutrients (B, Cu, Co, I, Mn, Mo, and Zn), and Fe-EDTA. Each factor was varied over a range of concentrations. Treatment combinations were selected using response surface methods. Five pears in three species (Pyrus communis ‘Horner 51,’ ‘Old Home × Farmingdale 87,’ ‘Winter Nelis,’ Pyrus dimorphophylla, and Pyrus ussuriensis ‘Hang Pa Li’) were grown on each treatment combination, responses were measured, and each response was analyzed by analysis of variance. The analyses resulted in the identification of the following factors with the single largest effects on plant response: shoot quality (mesos), leaf spotting/necrosis (mesos), leaf size (mesos), leaf color (mesos, NH4NO3, and KNO3), shoot number (NH4NO3 and Fe), nodes (NH4NO3 and KNO3), and shoot length (mesos and Fe). Factors with the largest effects (mesos and Fe) were similar among the genotypes. This approach was very successful for defining the appropriate types and concentrations of mineral nutrients for micropropagation of diverse pear genotypes.




Similar content being viewed by others
References
Adelberg JW, Delgado MP, Tomkins JT (2010) Spent medium analysis for liquid culture micropropagation of Hemerocallis on Murashige and Skoog medium. In Vitro Cell Dev Biol- Plant 46:95–107
Aranda-Peres AN, Martinelli AP (2009) Adjustment of mineral elements in the culture medium for the micropropagation of three Vriesea Bromeliads from the Brazilian Atlantic Forest: The importance of calcium. HortScience 44(1):106–112
Bell RL, Reed BM (2002) In vitro tissue culture of pear: advances in techniques for micropropagation and germplasm preservation. Acta Hort 596:412–418
Bell RL, Srinivasan C, Lomberk D (2009) Effect of nutrient media on axillary shoot proliferation and preconditioning for adventitious shoot regeneration of pears. In Vitro Cell Dev Biol-Plant 45:708–714
Design-Expert Design-Expert 8 (2010) Minneapolis, MN: Stat-Ease, Inc
Driver JA, Kuniyuki AH (1984) In vitro propagation of Paradox walnut rootstock. HortScience 19:507–509
Evens TJ, Niedz RP ARS-Media: Ion Solution Calculator (2008) Version 1. Ft. Pierce, FL: U.S. Horticultural Research Laboratory
Kintzios S, Stavropoulou E, Skamneli S (2004) Accumulation of selected macronutrients and carbohydrates in melon tissue cultures: association with pathways of in vitro dedifferentiation and differentiation (organogenesis, somatic embryogenesis). Plant Sci 167:655–664
Lloyd G, McCown B (1980) Commercially feasible micropropagation of mountain laurel, Kalmia latifolia, by use of shoot-tip culture. Comb Proceed Int Plant Prop Soc 30:421–427
Murashige T (1974) Plant propagation through tissue cultures. Ann Rev Plant Physiol 25:135–166
Murashige T, Nakano R (1965) Morphogenetic behaviour of tobacco tissue cultures and implication of plant senescence. Am J Bot 52(8):819–827
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497
Nas MN, Read PE (2004) A hypothesis for the development of a defined tissue culture medium of higher plants and micropropagation of hazelnuts. Scientia Hortic 101:189–200
Niedz RP, Evens TJ (2006) A solution to the problem of ion confounding in experimental biology. Nature Methods 3:417
Niedz RP, Evens TJ (2007) Regulating plant tissue growth by mineral nutrition. In Vitro Cell Dev Biol-Plant 43:370–381
Niedz RP, Hyndman SE, Evens TJ (2007) Using a Gestalt to measure the quality of in vitro responses. Scientia Hortic 112:349–359
Preece J (1995) Can nutrient salts partially substitute for plant growth regulators? Plant Tiss Cult Biotech 1:26–37
Quoirin M, Lepoivre P (1977) Improved media for in vitro culture of Prunus. Acta Hort 78:437–442
Ramage CM, Williams RR (2002) Mineral nutrition and plant morphogenesis. In Vitro Cell Dev Biol-Plant 38:116–124
Reed BM (1995) Screening Pyrus germplasm for in vitro rooting response. HortScience 30:1292–1294
Singha S (1986) Pear (Pyrus communis). In: Bajaj YPS (ed) Biotechnology in Agriculture and Forestry. Springer-Verlag, Heidelberg, Berlin, pp 198–206
Singha S, Townsend EC, Oberly GH (1985) Mineral nutrient status of crabapple and pear shoots cultured in vitro on varying concentrations of three commercial agars. J Am Soc Hort Sci 110:407–411
Sotiropoulos TE, Fotopoulos S, Dimassi KN, Tsirakoglou V (2006) Response of the pear rootstock to boron and salinity in vitro. Biol Plant 50:779–781
Thakur A, Kanwar JS (2008) Micropropagation of ‘wild pear’ Pyrus pyrifolia (Burm.F.) Nakai. I. Explant establishment and shoot multiplication. Not Bot Hort Agrobot Cluj 36:103–108
Williams RR (1993) Mineral nutrition in vitro—a mechanistic approach. Aust J Bot 41:237–251
Zhao H, Gu N (1990) Pear. In: Chen Z, Evans DA, Sharp WR, Ammirato PV, Sondahl MR (eds) Handbook of plant cell culture. McGraw-Hill, New York, pp 264–277
Acknowledgments
We thank NCGR lab personnel for assistance with collection of the data for this study. This project was funded by a grant from the Oregon Association of Nurseries and the Oregon Department of Agriculture and by United States Department of Agriculture-Agricultural Research Service CRIS project 5358-21000-0-38-00D.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor: Lynn Dahleen
Rights and permissions
About this article
Cite this article
Reed, B.M., Wada, S., DeNoma, J. et al. Improving in vitro mineral nutrition for diverse pear germplasm. In Vitro Cell.Dev.Biol.-Plant 49, 343–355 (2013). https://doi.org/10.1007/s11627-013-9504-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11627-013-9504-1