Abstract
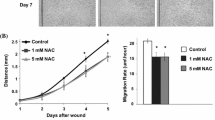
Dermal fibroblasts (DF) constitute one of key cells involved in wound healing. However, the functions they perform in wound conditions remain poorly understood. This study involved exposing DF to low nutrition and to low nutrition + LPS for 5 d as conditions representing the wound. Although DF exhibited increasing metabolic activity in time under all conditions including control, the proliferation did not change in both low nutrition and low nutrition + LPS. Only the low nutrition + LPS was found to potentiate the migration and pro-inflammatory phenotype (IL6 release) of DF. The potential of DF to contract collagen hydrogel declined only under low nutrition as a consequence of low cell number. The expression of α-SMA was reduced under both conditions independently of the cell number. The remodeling capability of DF was affected under both conditions as documented by the enhanced MMP2 activity. Finally, the production of collagen type I was not affected by either condition. The study shows that low nutrition as the single factor is able to delay the healing process. Moreover, the addition of the mild pro-inflammatory stimulus represented by LPS may amplify the cell response in case of decreased α-SMA expression or excite DF to produce IL6 impairing the healing process.








Similar content being viewed by others
References
Abbott A (2003) Cell culture: biology’s new dimension. Nature. https://doi.org/10.1038/424870a
Akkoc A, Kahraman MM, Vatansever A et al (2016) Lipopolysaccharide (LPS) induces matrix metalloproteinase-2 and -9 (MMP-2 and MMP-9) in bovine dermal fibroblasts. Pak Vet J 36:189–193
Basso FG, Soares DG, Pansani TN et al (2015) Effect of LPS treatment on the viability and chemokine synthesis by epithelial cells and gingival fibroblasts. Arch Oral Biol. https://doi.org/10.1016/j.archoralbio.2015.04.010
Bolívar S, Santana R, Ayala P et al (2017) Lipopolysaccharide activates toll-like receptor 4 and prevents cardiac fibroblast-to-myofibroblast differentiation. Cardiovasc Toxicol. https://doi.org/10.1007/s12012-017-9404-4
Burke JP, Cunningham MF, Watson RWG et al (2010) Bacterial lipopolysaccharide promotes profibrotic activation of intestinal fibroblasts. Br J Surg. https://doi.org/10.1002/bjs.7045
Cai XJ, Chen L, Li L et al (2010) Adiponectin inhibits lipopolysaccharide-induced adventitial fibroblast migration and transition to myofibroblasts via AdipoR1-AMPK-iNOS pathway. Mol Endocrinol. https://doi.org/10.1210/me.2009-0128
Caley MP, Martins VLC, O’Toole EA (2015) Metalloproteinases and wound healing. Adv Wound Care. https://doi.org/10.1089/wound.2014.0581
Cho JS, Kang JH, Um JY et al (2014) Lipopolysaccharide induces pro-inflammatory cytokines and mmp production via TLR4 in nasal polyp-derived fibroblast and organ culture. PLoS One. https://doi.org/10.1371/journal.pone.0090683
Crompton R, Williams H, Ansell D et al (2016) Oestrogen promotes healing in a bacterial LPS model of delayed cutaneous wound repair. Lab Investig. https://doi.org/10.1038/labinvest.2015.160
Darby I, Skalli O, Gabbiani G (1990) Alpha-smooth muscle actin is transiently expressed by myofibroblasts during experimental wound-healing. Lab Investig 63:21–29
Desmouliere A, Geinoz A, Gabbiani F, Gabbiani G (1993) Transforming growth-factor-beta-1 induces alpha-smooth muscle actin expression in granulation-tissue myofibroblasts and in quiescent and growing cultured fibroblasts. J Cell Biol 122:103–111. https://doi.org/10.1083/jcb.122.1.103
Ejiri H, Nomura T, Hasegawa M et al (2015) Use of synthetic serum-free medium for culture of human dermal fibroblasts to establish an experimental system similar to living dermis. Cytotechnology. https://doi.org/10.1007/s10616-014-9709-0
Eleftheriadis T, Liakopoulos V, Lawson B, et al (2011) Lipopolysaccharide and hypoxia significantly alters interleukin-8 and macrophage chemoattractant protein-1 production by human fibroblasts but not fibrosis related factors. Hippokratia 15:238–243
Greaves NS, Ashcroft KJ, Baguneid M, Bayat A (2013) Current understanding of molecular and cellular mechanisms in fibroplasia and angiogenesis during acute wound healing. J Dermatol Sci
Gvirtz R, Ogen-shtern N, Cohen G (2020) Kinetic cytokine secretion profile of LPS-induced inflammation in the human skin organ culture. Pharmaceutics. https://doi.org/10.3390/pharmaceutics12040299
He Z, Gao Y, Deng Y et al (2012) Lipopolysaccharide induces lung fibroblast proliferation through toll-like receptor 4 signaling and the phosphoinositide3-kinase-Akt pathway. PLoS One. https://doi.org/10.1371/journal.pone.0035926
Hehenberger K, Kratz G, Hansson A, Brismar K (1998) Fibroblasts derived from human chronic diabetic wounds have a decreased proliferation rate, which is recovered by the addition of heparin. J Dermatol Sci. https://doi.org/10.1016/S0923-1811(97)00042-X
Hinz B, Phan SH, Thannickal VJ et al (2007) The myofibroblast: one function, multiple origins. Am J Pathol. https://doi.org/10.2353/ajpath.2007.070112
Howard EW, Crider BJ, Updike DL et al (2012) MMP-2 expression by fibroblasts is suppressed by the myofibroblast phenotype. Exp Cell Res. https://doi.org/10.1016/j.yexcr.2012.03.007
Iocono JA, Colleran KR, Remick DG et al (2000) Interleukin-8 levels and activity in delayed-healing human thermal wounds. Wound Repair Regen. https://doi.org/10.1046/j.1524-475X.2000.00216.x
Jin J, Sundararaj KP, Samuvel DJ et al (2012) Different signaling mechanisms regulating IL-6 expression by LPS between gingival fibroblasts and mononuclear cells: seeking the common target. Clin Immunol. https://doi.org/10.1016/j.clim.2012.01.019
Kim MS, Song HJ, Lee SH, Lee CK (2014) Comparative study of various growth factors and cytokines on type I collagen and hyaluronan production in human dermal fibroblasts. J Cosmet Dermatol. https://doi.org/10.1111/jocd.12073
Kostarnoy AV, Gancheva PG, Logunov DY et al (2013) Topical bacterial lipopolysaccharide application affects inflammatory response and promotes wound healing. J Interf Cytokine Res. https://doi.org/10.1089/jir.2012.0108
Lemons JMS, Coller HA, Feng XJ et al (2010) Quiescent fibroblasts exhibit high metabolic activity. PLoS Biol. https://doi.org/10.1371/journal.pbio.1000514
Li WW, Carter MJ, Mashiach E, Guthrie SD (2017) Vascular assessment of wound healing: a clinical review. Int Wound J. https://doi.org/10.1111/iwj.12622
Li X-J, Huang F-Z, Wan Y et al (2018) Lipopolysaccharide stimulated the migration of NIH3T3 cells through a positive feedback between β-catenin and COX-2. Front Pharmacol. https://doi.org/10.3389/fphar.2018.01487
Liang CC, Park AY, Guan JL (2007) In vitro scratch assay: a convenient and inexpensive method for analysis of cell migration in vitro. Nat Protoc. https://doi.org/10.1038/nprot.2007.30
Lindner D, Zietsch C, Becher PM et al (2012) Differential expression of matrix metalloproteases in human fibroblasts with different origins. Biochem Res Int. https://doi.org/10.1155/2012/875742
Martins VL, Caley M, O’Toole EA (2013) Matrix metalloproteinases and epidermal wound repair. Cell Tissue Res 351:255–268. https://doi.org/10.1007/s00441-012-1410-z
Mihara M, Moriya Y, Ohsugi Y (1996) IL-6-soluble IL-6 receptor complex inhibits the proliferation of dermal fibroblasts. Int J Immunopharmacol. https://doi.org/10.1016/0192-0561(95)00106-9
Rampersad SN (2012) Multiple applications of alamar blue as an indicator of metabolic function and cellular health in cell viability bioassays. Sensors (switzerland). https://doi.org/10.3390/s120912347
Rolfe MW, Kunkel SL, Standiford TJ et al (1991) Pulmonary fibroblast expression of interleukin-8: a model for alveolar macrophage-derived cytokine networking. Am J Respir Cell Mol Biol. https://doi.org/10.1165/ajrcmb/5.5.493
Schwarz F, Jennewein M, Bubel M et al (2013) Soft tissue fibroblasts from well healing and chronic human wounds show different rates of myofibroblasts in vitro. Mol Biol Rep. https://doi.org/10.1007/s11033-012-2223-6
Smith RS, Smith TJ, Blieden TM, Phipps RP (1997) Fibroblasts as sentinel cells. Synthesis of chemokines and regulation of inflammation. Am J Pathol 151:317–322
Stunova A, Vistejnova L (2018) Dermal fibroblasts-a heterogeneous population with regulatory function in wound healing. Cytokine Growth Factor Rev 39:137–150. https://doi.org/10.1016/j.cytogfr.2018.01.003
Tomasek JJ, Gabbiani G, Hinz B, et al (2002) Myofibroblasts and mechano-regulation of connective tissue remodelling. Nat Rev Mol Cell Biol 3:349–363. https://doi.org/10.1038/nrm809
Tracy LE, Minasian RA, Caterson EJ (2016) Extracellular matrix and dermal fibroblast function in the healing wound. Adv Wound Care 5:119–136. https://doi.org/10.1089/wound.2014.0561
Wall IB, Moseley R, Baird DM et al (2008) Fibroblast dysfunction is a key factor in the non-healing of chronic venous leg ulcers. J Invest Dermatol. https://doi.org/10.1038/jid.2008.114
Xi ZD, Xie CY, Xi Y Bin (2016) Macrophage migration inhibitory factor enhances lipopolysaccharide-induced fibroblast proliferation by inducing toll-like receptor 4. BMC Musculoskelet Disord. https://doi.org/10.1186/s12891-016-0895-0
Xia Y, Pauza ME, Feng L, Lo D (1997) RelB regulation of chemokine expression modulates local inflammation. Am J Pathol 151:375–387
Xing Z, Jordana M, Braciak T et al (1993) Lipopolysaccharide induces expression of granulocyte/macrophage colony-stimulating factor, interleukin-8, and interleukin-6 in human nasal, but not lung, fibroblasts: evidence for heterogeneity within the respiratory tract. Am J Respir Cell Mol Biol. https://doi.org/10.1165/ajrcmb/9.3.255
Zgheib C, Xu J, Liechty KW (2014) Targeting inflammatory cytokines and extracellular matrix composition to promote wound regeneration. Adv Wound Care. https://doi.org/10.1089/wound.2013.0456
Zhang J, Wu L, Qu JM (2011) Inhibited proliferation of human lung fibroblasts by LPS is through IL-6 and IL-8 release. Cytokine. https://doi.org/10.1016/j.cyto.2011.02.018
Acknowledgements
The authors acknowledge the invaluable assistance provided by Iveta Paurova via her support in terms of the provision of laboratory services.
Funding
The study was supported by project no. CZ.02.1.01/0.0/0.0/16_019/0000787 “Fighting Infectious Diseases,” awarded by the Ministry of Education, Youth and Sports of the Czech Republic and financed from the European Regional Development Fund; by project no. NU20-02–00368 awarded by Czech Health Research Council and by the Ministry of Health of the Czech Republic, by project GA UK No. 128417 awarded by the Charles University; and financed from the European Regional Development Fund and by the Cooperation Program, research area MED/DIAG.
Author information
Authors and Affiliations
Contributions
Anna Zavadakova and Lucie Vistejnova designed the experiments and prepared the figures. Anna Zavadakova and Pavla Tonarova performed the experiments and collected the data. Data analysis was performed by all authors. The first draft of the manuscript was written by Anna Zavadakova and all authors commented on previous versions of the manuscript. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests. The funders had no role in the design of the study; in the collection, analysis or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Additional information
Editor: John W. Harbell
Supplementary Information
Below is the link to the electronic supplementary material.
11626_2022_713_MOESM1_ESM.jpg
Supplementary file1 Fig. S1 Representative images of collagen hydrogels seeded by dermal fibroblasts and monitored for contraction under control, low nutrition and low nutrition + LPS conditions for 13 days.Suppl. (JPG 544 KB)
11626_2022_713_MOESM2_ESM.jpg
Supplementary file2 Fig. S2 Images of whole wells of 96-well test plates with dermal fibroblasts cultured under control, low nutrition and low nutrition + LPS conditions for 5 days. With respect to the quantification of the α-SMA, nine sequential images covering the whole well area were captured by means of an Olympus UPlanFL N 4x/0.13 objective followed by automatic whole well area picture decomposition using VisiView software. The number of nuclei and the area of the α-SMA were determined from the same well by means of ImageJ Fiji (National Institute of Health, Bethseda, USA). The results were expressed as the mean of the α-SMA area at day 5 normalized to the cell count at day 5 ± standard deviation from 8 independent NHDF donors. (JPG 2652 KB)
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zavadakova, A., Vistejnova, L. & Tonarova, P. Functional responses of dermal fibroblasts to low nutrition and pro-inflammatory stimuli mimicking a wound environment in vitro. In Vitro Cell.Dev.Biol.-Animal 58, 643–657 (2022). https://doi.org/10.1007/s11626-022-00713-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11626-022-00713-7