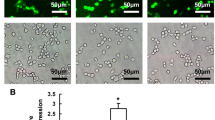
Abstract
Glutamate neurotoxicity is involved in neurodegenerative diseases, including Alzheimer’s and Parkinson’s diseases. Excess glutamate causes caspase-independent programmed cell death via oxidative stress and calcium influx. Our previous study showed that calpain-1 localizes to both the cytoplasm and mitochondria, where apoptosis-inducing factor (AIF) is cleaved by calpain-1 and translocates to the nucleus to induce DNA fragmentation. The autoinhibitory region of calpain-1 conjugated with the cell-penetrating peptide HIV1-Tat (namely Tat-μCL) specifically prevents the activity of mitochondrial calpain-1 and attenuates neuronal cell death in animal models of retinitis pigmentosa, as well as glutamate-induced cell death in mouse hippocampal HT22 cells. In the present study, we constructed a lentiviral vector expressing the Tat-μCL peptide and evaluated its protective effect against glutamate-induced cell death in HT22 cells. Lentiviral transduction with Tat-μCL significantly suppressed glutamate-induced nuclear translocation of AIF and DNA fragmentation. The findings of the present study suggest that the stable expression of Tat-μCL may be a potential gene therapy modality for neurodegenerative diseases.



Similar content being viewed by others
References
Ambrosi G, Cerri S, Blandini F (2014) A further update on the role of excitotoxicity in the pathogenesis of Parkinson’s disease. J Neural Transm 121:849–859. https://doi.org/10.1007/s00702-013-1149-z
Candé C, Cohen I, Daugas E, Ravagnan L, Larochette N, Zamzami N, Kroemer G (2002) Apoptosis-inducing factor (AIF): a novel caspase-independent death effector released from mitochondria. Biochimie 84:215–222. https://doi.org/10.1016/s0300-9084(02)01374-3
Candé C, Vahsen N, Garrido C, Kroemer G (2004) Apoptosis-inducing factor (AIF): caspase-independent after all. Cell Death Differ 11:591–595. https://doi.org/10.1038/sj.cdd.4401400
Chen A, Wang H, Zhang Y, Wang X, Yu L, Xu W, Xu W, Lin Y (2017) Paeoniflorin exerts neuroprotective effects against glutamate-induced PC12 cellular cytotoxicity by inhibiting apoptosis. Int J Mol Med 40:825–833. https://doi.org/10.3892/ijmm.2017.3076
Choi DW, Rothman SM (1990) The role of glutamate neurotoxicity in hypoxic-ischemic neuronal death. Annu Rev Neurosci 13:171–182. https://doi.org/10.1146/annurev.ne.13.030190.001131
Chukai Y, Iwamoto T, Itoh K, Tomita H, Ozaki T (2021) Characterization of mitochondrial calpain-5. Biochim Biophys Acta Mol Cell Res 1868:118989. https://doi.org/10.1016/j.bbamcr.2021.118989
D’Souza SW, Slater P (1995) Excitatory amino acids in neonatal brain: contributions to pathology and therapeutic strategies. Arch Dis Child Fetal Neonatal Ed 72:F147–F150. https://doi.org/10.1136/fn.72.3.f147
Fukui M, Song J-H, Choi J, Choi HJ, Zhu BT (2009) Mechanism of glutamate-induced neurotoxicity in HT22 mouse hippocampal cells. Eur J Pharmacol 617:1–11. https://doi.org/10.1016/j.ejphar.2009.06.059
Goll DE, Thompson VF, Li H, Wei W, Cong J (2003) The calpain system. Physiol Rev 83:731–801. https://doi.org/10.1152/physrev.00029.2002
González-Reyes RE, Nava-Mesa MO, Vargas-Sánchez K, Ariza-Salamanca D, Mora-Muñoz L (2017) Involvement of astrocytes in Alzheimer’s disease from a neuroinflammatory and oxidative stress perspective. Front Mol Neurosci 10:427. https://doi.org/10.3389/fnmol.2017.00427
Gouko R, Onuma M, Eitsuka T, Katayama M, Takahashi K, Nakagawa K, Inoue-Murayama M, Kiyono T, Fukuda T (2018) Efficient immortalization of cells derived from critically endangered Tsushima leopard cat (Prionailurus bengalensis euptilurus) with expression of mutant CDK4, Cyclin D1, and telomerase reverse transcriptase. Cytotechnology 70:1619–1630. https://doi.org/10.1007/s10616-018-0254-0
Han X, Liu C, Zhang K, Guo M, Shen Z, Liu Y, Zhuo Z, Cao M, Li Y (2018) Calpain and JNK pathways participate in isoflurane-induced nucleus translocation of apoptosis-inducing factor in the brain of neonatal rats. Toxicol Lett 285:60–73. https://doi.org/10.1016/j.toxlet.2017.12.022
Hanna RA, Campbell RL, Davies PL (2008) Calcium-bound structure of calpain and its mechanism of inhibition by calpastatin. Nature 456:409–412. https://doi.org/10.1038/nature07451
Hlavatý J, Tonar Z, Renner M, Panitz S, Petznek H, Schweizer M, Schüle S, Kloke B-P, Moldzio R, Witter K (2017) Tropism, intracerebral distribution, and transduction efficiency of HIV- and SIV-based lentiviral vectors after injection into the mouse brain: a qualitative and quantitative in vivo study. Histochem Cell Biol 148:313–329. https://doi.org/10.1007/s00418-017-1569-1
Keaney J, Campbell M (2015) The dynamic blood-brain barrier. FEBS J 282:4067–4079. https://doi.org/10.1111/febs.13412
Lewerenz J, Maher P (2015) Chronic glutamate toxicity in neurodegenerative diseases-what is the evidence ? Front Neurosci 9:469. https://doi.org/10.3389/fnins.2015.00469
Liang Z, Currais A, Soriano-Castell D, Schubert D, Maher P (2021) Natural products targeting mitochondria: emerging therapeutics for age-associated neurological disorders. Pharmacol Ther 221:107749. https://doi.org/10.1016/j.pharmthera.2020.107749
Matsushita M, Kitoh H, Ohkawara B, Mishima K, Kaneko H, Ito M, Masuda A, Ishiguro N, Ohno K (2013) Meclozine facilitates proliferation and differentiation of chondrocytes by attenuating abnormally activated FGFR3 signaling in achondroplasia. PLoS ONE 8:e81569. https://doi.org/10.1371/journal.pone.0081569
Ohtsuki S, Terasaki T (2007) Contribution of carrier-mediated transport systems to the blood–brain barrier as a supporting and protecting interface for the brain; importance for CNS drug discovery and development. Pharm Res 24:1745–1758. https://doi.org/10.1007/s11095-007-9374-5
Ozaki T, Tomita H, Tamai M, Ishiguro S-I (2007) Characteristics of mitochondrial calpains. J Biochem 142:365–376. https://doi.org/10.1093/jb/mvm143
Ozaki T, Yamashita T, Ishiguro S (2008) ERp57-associated mitochondrial μ -calpain truncates apoptosis-inducing factor. Biochim Biophys Acta 1783:1955–1963. https://doi.org/10.1016/j.bbamcr.2008.05.011
Ozaki T, Yamashita T, Ishiguro S-I (2009) Mitochondrial m-calpain plays a role in the release of truncated apoptosis-inducing factor from the mitochondria. Biochim Biophys Acta - Mol Cell Res 1793:1848–1859. https://doi.org/10.1016/j.bbamcr.2009.10.002
Ozaki T, Nakazawa M, Yamashita T, Sorimachi H, Hata S, Tomita H, Isago H, Baba A, Ishiguro S (2012) Intravitreal injection or topical eye-drop application of a μ-calpain C2L domain peptide protects against photoreceptor cell death in Royal College of Surgeons’ rats, a model of retinitis pigmentosa. Biochim Biophys Acta - Mol Basis Dis 1822:1783–1795. https://doi.org/10.1016/j.bbadis.2012.07.018
Sugawara M, Abe T, Kasai S, Itoh K, Ozaki T (2021) Calpain-1 C2L domain peptide protects mouse hippocampus-derived neuronal HT22 cells against glutamate-induced oxytosis. Biochem Biophys Rep 27:101101. https://doi.org/10.1016/j.bbrep.2021.101101
Tajes M, Ramos-Fernández E, Weng-Jiang X, Bosch-Morató M, Guivernau B, Eraso-Pichot A, Salvador B, Fernàndez-Busquets X, Roquer J, Muñoz FJ (2014) The blood-brain barrier: structure, function and therapeutic approaches to cross it. Mol Membr Biol 31:152–167. https://doi.org/10.3109/09687688.2014.937468
Wang J, Wang F, Mai D, Qu S (2020) Molecular mechanisms of glutamate toxicity in Parkinson’s disease. Front Neurosci 14:585584. https://doi.org/10.3389/fnins.2020.585584
Zhang YM, Bhavnani BR (2006) Glutamate-induced apoptosis in neuronal cells is mediated via caspase-dependent and independent mechanisms involving calpain and caspase-3 proteases as well as apoptosis inducing factor (AIF) and this process is inhibited by equine estrogens. BMC Neurosci 7:49. https://doi.org/10.1186/1471-2202-7-49
Acknowledgements
We would like to thank Dr. Hiroyuki Miyoshi (RIKEN, BioResource Center) for providing the lentiviral packaging and CSII backbone plasmids. We also thank Dr. Shuya Kasai (Hirosaki University Graduate School of Medicine, Center for Advanced Medical Research) for carefully proofreading the manuscript.
Funding
The present study was supported, in part, by the Uehara Memorial Foundation, Japan, for Taku Ozaki.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare are no competing interests.
Rights and permissions
About this article
Cite this article
Oikawa, T., Fukuda, T., Yamashita, T. et al. Lentiviral expression of calpain-1 C2-like domain peptide prevents glutamate-induced cell death in mouse hippocampal neuronal HT22 cells. In Vitro Cell.Dev.Biol.-Animal 58, 289–294 (2022). https://doi.org/10.1007/s11626-022-00683-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11626-022-00683-w