Abstract
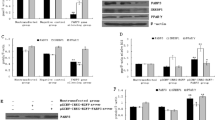
14-3-3 proteins are an acidic protein family that is highly conserved and widely distributed in eukaryotic cells. Recent studies have found that 14-3-3 proteins play critical roles in cell signal transductions, cell growth and differentiation, and protein synthesis. 14-3-3γ is an important member of 14-3-3 protein family. In our previous study, we found that 14-3-3γ was upregulated by estrogen in dairy cow mammary epithelial cell (DCMEC), but the function and mechanism of 14-3-3γ is not known. In this experiment, we first cultured and purified the primary DCMEC and found 14-3-3γ located both in the cytoplasm and nucleus by using immunofluorescence assay. Methionine, lysine, estrogen, and prolactin could upregulate the expression of 14-3-3γ, stimulate the secretion of β-casein and triglyceride, and raise the cell viability of DCMEC. We constructed a stable 14-3-3γ overexpression cell line of DCMEC and found that the expressions of mTOR and p-mTOR, the secretion of triglyceride and β-casein (CSN2), and the cell viability of DCMEC were all upregulated. We also observed the effects of 14-3-3γ gene silencing and gained consistent results with 14-3-3γ overexpression. These findings reveal that 14-3-3γ affects the mTOR pathway and regulates lactogenesis in DCMECs.




Similar content being viewed by others
References
Aitken A (2006) 14-3-3 proteins: a historic overview. Semin Cancer Biol 16:162–172
Ajjappala BS, Kim YS, Kim MS, Lee MY, Lee KY, Ki HY, Cha DH, Baek KH (2009) 14-3-3 gamma is stimulated by IL-3 and promotes cell proliferation. J Immunol 182:1050–1060
Autieri M, Haines D, Romanic A, Ohlstein E (1996) Expression of 14-3-3 gamma in injured arteries and growth factor- and cytokine-stimulated human vascular smooth muscle cells. Cell Growth Differ 7:1453–1460
Bakan I, Laplante M (2012) Connecting mTORC1 signaling to SREBP-1 activation. Curr Opin Lipidol 23:226–234
Bertram PG, Zeng C, Thorson J, Shaw AS, Zheng X (1998) The 14-3-3 proteins positively regulate rapamycin-sensitive signaling. Curr Biol 8:1259–1267
Burgos SA, Cant JP (2010) IGF-1 stimulates protein synthesis by enhanced signaling through mTORC1 in bovine mammary epithelial cells. Domest Anim Endocrinol 38:211–221
Burgos SA, Dai M, Cant JP (2010) Nutrient availability and lactogenic hormones regulate mammary protein synthesis through the mammalian target of rapamycin signaling pathway. J Dairy Sci 93:153–161
Bustad HJ, Skjaerven L, Ying M, Halskau Ø, Baumann A, Rodriguez-Larrea D, Costas M, Underhaug J, Sanchez-Ruiz JM, Martinez A (2012) The peripheral binding of 14-3-3c to membranes involves isoform-specific histidine residues. PLoS ONE 7:e49671
Fu H, Subramanian RR, Masters SC (2000) 14-3-3 proteins: structure, function, and regulation. Annu Rev Pharmacol Toxicol 40:617–647
Hu H, Wang JQ, Bu DP, Wei HY, Zhou LY et al (2009) In vitro culture and characterization of a mammary epithelial cell line from Chinese Holstein dairy cow. PLoS ONE 4:e7636
Huang YL, Zhao F, Luo CC, Zhang X, Si Y, Sun Z, Zhang L, Li QZ, Gao XJ (2013) SOCS3-mediated blockade reveals major contribution of JAK2/STAT5 signaling pathway to lactation and proliferation of dairy cow mammary epithelial cells in vitro. Molecules 18:12987–13002
Kasahara K, Goto H, Izawa I, Kiyono T, Watanabe N, Elowe S, Nigg EA, Inagaki M (2013) PI 3-kinase-dependent phosphorylation of Plk1-Ser99 promotes association with 14-3-3γ and is required for metaphase-anaphase transition. Nat Commun 4:1882
Ke Z, Liu HY, Zhou MM, Liu JX (2010) Establishment and characterization of a lactating bovine mammary epithelial cell model for the study of milk synthesis. Cell Biol Int 34:717–721
Lam T, Lisa M, Clayton A, Li G, Egest J, Xu ZH, Casali P (2013) Scaffold functions of 14-3-3 adaptors in B cell immunoglobulin class switch DNA recombination. PLoS ONE 8:80141
Li HM, Wang CM, Li QZ, Gao XJ (2012) MiR-15a decreases bovine mammary epithelial cell viability and lactation and regulates growth hormone receptor expression. Molecules 17:12037–12048
Liang MY, Hou XM, Qu B, Zhang N, Li N, Cui YJ, Li QZ, Gao XJ (2014) Functional analysis of FABP3 in the milk fat synthesis signaling pathway of bovine mammary epithelial cells. In Vitro Cell Dev Biol Anim 50:865–873
Liu XF, Li M, Li QZ, Lu LM, Tong HL, Gao XJ (2012) Stat5a increases lactation of dairy cow mammary gland epithelial cells cultured in vitro. In Vitro Cell Dev Biol Anim 48:554–561
Lu S, Becker KA, Hagen MJ, Yan H, Roberts AL, Mathews LA, Schneider SS, Siegelmann HT, MacBeth KJ, Tirrell SM, Blanchard JL, Jerry DJ (2008) Transcriptional responses to estrogen and progesterone in mammary gland identify networks regulating p53 activity. Endocrinology 149:4809–4820
Lu LM, Li QZ, Huang JG, Gao XJ (2013) Proteomic and functional analyses reveal MAPK1 regulates milk protein synthesis. Molecules 18:263–275
Mori H, Inoue M, Yano M, Wakabayashi H, Kido H (2000) 14-3-3τ associates with a translational control factor FKBP12-rapamycin-associated protein in T-cells after stimulation by per vanadate. FEBS Lett 467:61–64
Olayioye MA, Guthridge MA, Stomski FC, Lopez AF, Visvader JE, Lindeman GJ (2003) Threonine 391 phosphorylation of the human prolactin receptor mediates a novel interaction with 14-3-3 proteins. J Biol Chem 278:32929–32935
Pantschenko AG, Woodcock-Mitchell J, Bushmich SL, Yang TJ (2000) Establishment and characterization of a caprine mammary epithelial cell line (CMEC). In Vitro Cell Dev Biol Anim 36:26–37
Peterfy M, Harris TE, Fujita N, Reue K (2010) Insulin-stimulated interaction with 14-3-3 promotes cytoplasmic localization of lipin-1 in adipocytes. J Biol Chem 285:3857–3864
Peterson DG, Matitashvili EA, Bauman DE (2004) The inhibitory effect of trans-10, cis-12 CLA on lipid synthesis in bovine mammary epithelial cells involves reduced proteolytic activation of the transcription factor SREBP-1. American Society for Nutritional Sciences. 2523-2527
Porstmann T, Santos CR, Griffiths B, Cully M, Wu M, Leevers S, Griffiths JR, Chung YL, Schulze A (2008) SREBP activity is regulated by mTORC1 and contributes to Akt-dependent cell growth. Cell Metab 8:224–236
Sancak Y, Bar-Peled L, Zoncu R, Markhard AL, Nada S, Sabatini DM (2010) Agulator-Rag complex targets mTORC1 to the lysosomal surface and is necessary for its activation by amino acids. Cell 141:290–303
Tong HL, Li QZ, Gao XJ, Yin DY (2012) Establishment and characterization of a lactating dairy goat mammary gland epithelial cell line. In Vitro Cell Dev Biol Anim 48:149–155
Wang L, Lin Y, Bian Y, Liu L, Shao L, Lin L, Qu B, Zhao F, Gao X, Li Q (2014) Leucyl-tRNA synthetase regulates lactation and cell proliferation via mTOR signaling in dairy cow mammary epithelial cells. Int J Mol Sci 15:5952–5969
Yu C, Luo CC, Qu B, Khudhair N, Gu X, Zang Y, Wang CH, Zhang N, Li Q, Gao X (2014) Molecular network including eIF1AX, RPS7, and 14-3-3γ regulates protein translation and cell proliferation in bovine mammary epithelial cells. Arch Biochem Biophys 564:142–155
Zhang X, Zhao F, Si Y, Huang YL, Yu CP, Luo CC, Zhang N, Li QZ, Gao XJ (2014) Negative feedback-loop regulation of GSK3β phosphorylation and mTOR/S6K1 signaling pathway independent of AKT to regulate milk synthesis and cell proliferation of dairy cow mammary epithelial cells. Molecules 19:9435–9452
Zhao J, Du Y, Horton JR, Upadhyay AK, Lou B, Bai Y, Zhang X, Du L, Li M, Wang B, Zhang L, Barbieri JT, Khuri FR, Cheng X, Fu H (2012) Discovery and structural characterization of a small molecule 14-3-3 protein-protein interaction inhibitor. Proc Natl Acad Sci U S A 108:16212–16216
Zhao J, Meyerkord CL, Du Y, Khuri FR, Fu H (2011) 14-3-3 proteins as potential therapeutic targets. Semin Cell Dev Biol 22:705–712
Acknowledgments
This study was financially supported by 863 Project of Ministry of Science and Technology of China (No: 2013AA102504-03) and the Major State Basic Research Development Program of China (973 Program, 2011CB100804).
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor: T. Okamoto
Rights and permissions
About this article
Cite this article
Khudhair, N., Luo, C., Khalid, A. et al. 14-3-3γ affects mTOR pathway and regulates lactogenesis in dairy cow mammary epithelial cells. In Vitro Cell.Dev.Biol.-Animal 51, 697–704 (2015). https://doi.org/10.1007/s11626-015-9879-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11626-015-9879-x