Abstract
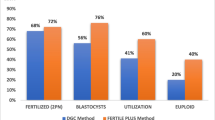
Cryopreservation of mammalian oocytes is an important way to provide a steady source of materials for research and practice of parthenogenetic activation, in vitro fertilization, and nuclear transfer. However, oocytes cryopreservation has not been common used, as there still are some problems waiting to be solved on the repeatability, safety, and validity. Then, it is necessary to investigate the damage occurred from vitrification and find a way to avoid or repair it. In this study, mouse mature oocytes were firstly pretreated in different equilibrium media, such as 5% ethylene glycol (EG) + 5% dimethyl sulfoxide (DMSO), 10% EG + 10% DMSO, and 15% EG + 15% DMSO in TCM199 supplemented with 20% fetal calf serum (FCS), for 1, 3, and 5 min, respectively, and then oocytes were transferred into vitrification solution (20% EG, 20% DMSO, 0.3 M sucrose, and 20% FCS in TCM199, M2, Dulbecco’s phosphate buffered saline, and 0.9% saline medium, respectively) and immediately loaded into glass capillaries to be plunged into liquid nitrogen. After storage from 1 h to 1 wk, they were diluted in stepwise sucrose solutions. The surviving oocytes were stained for cortical granule, meiotic spindles, and chromosomes. Oocytes without treatments were used as controls. The results showed that oocytes pretreated in 5% EG +5% DMSO group for 3–5 min or in 10% EG + 10% DMSO group for 1–3 min were better than other treatments. Oocytes vitrified in TCM199 as basic medium showed higher survival and better subsequent embryonic development than other groups. When the concentration of FCS in vitrification solution reduced below 15%, the rates of survival, fertilization, and developing to blastocyst declined dramatically. The inner diameter (0.6 mm) of glass capillaries and amount of vitrification solution (1–3 μl) achieved more rapid cooling and warming and so reduce the injury to oocytes. Cropreservation led to the exocytosis of cortical granule of oocytes (about 10%) and serious disturbance of microtubules and chromosomes. With 2 h incubation, the microtubules could repolymerize and the rate of fertilization in vitro was much higher than those of 1 and 3 h incubation groups. In conclusion, the protection of basic medium and FCS to oocytes during cryopreservation and sufficient cooling and warming rates using glass capillaries have profound effects on oocytes survival and subsequent embryonic development competence. The appropriate time for fertilization in vitro may be related to the recovery of spindles after incubation and avoiding ageing in the whole process.





Similar content being viewed by others
References
Aigner, S.; Van der, Elst J.; Siebzehnrübl, E.; Wildt, L.; Lang, N.; Van Steirteghem, A. C. The influence of slow and ultra-rapid freezing on the organization of the meiotic spindle of the mouse oocyte. Hum Reprod 76: 857–864; 1992.
Almeida, P. A.; Bolton, V. N. I maturity and chromosomal abnormalities in oocytes that fail to develop pronuclei following insemination in vitro. Hum Reprod 82: 229–232; 1993.
Aman, R. R.; Parks, J. E. Effects of cooling and rewarming on the meiotic spindle and chromosomes of in vitro-matured bovine oocytes. Biol Reprod 501: 103–110; 1994. doi:10.1095/biolreprod50.1.103.
Asada, M.; Ishibashi, S.; Ikumi, S.; Fukui, Y. Effect of polyvinyl alcohol (PVA) concentration during vitrification of in vitro matured bovine oocytes. Theriogenology 586: 1199–1208; 2002. doi:10.1016/S0093-691X(02)00948-2.
Bautista, J. A.; Dela Peña, E. C.; Katagiri, S.; Takahashi, Y.; Kanagawa, H. In vitro viability of mouse oocytes vitrified in an ethylene glycol-based solution. Jpn J Vet Res 461: 13–18; 1998.
Bernard, A.; Fuller, B. J. Cryopreservation of human oocytes: a review of current problems and perspectives. Hum Reprod Update 23: 193–207; 1996. doi:10.1093/humupd/2.3.193.
Carroll, J.; Wood, M. J.; Whittingham, D. G. Normal fertilization and development of frozen–thawed mouse oocytes: protective action of certain macromolecules. Biol Reprod 483: 606–612; 1993. doi:10.1095/biolreprod48.3.606.
Chatot, C. L.; Ziomek, C. A.; Bavister, B. D.; Lewis, J. L.; Torres, I. An improved culture medium supports development of random-breed 1-cell mouse embryos in vitro. J Reprod Fert 86: 679–688; 1989. doi:10.1530/jrf.0.0860679.
Chen, S. U.; Lien, Y. R.; Chen, H. F.; Chao, K. H.; Ho, H. N.; Yang, Y. S. Open pulled straws for vitrification of mature mouse oocytes preserve patterns of meiotic spindles and chromosomes better than conventional straws. Hum Reprod 1512: 2598–2603; 2000. doi:10.1093/humrep/15.12.2598.
Chen, S. U.; Lien, Y. R.; Cheng, Y. Y.; Chen, H. F.; Ho, H. N.; Yang, Y. S. Vitrification of mouse oocytes using closed pulled straws (CPS) achieves a high survival and preserves good patterns of meiotic spindles, compared with conventional straws, open pulled straws (OPS) and grids. Hum Reprod 1611: 2350–2356; 2001.
Dela Peña, E. C.; Takahashi, Y.; Atabay, E. C.; Katagiri, S.; Nagano, M. Vitrification of mouse oocytes in ethylene glycol-raffinose solution: effects of preexposure to ethylene glycol or raffinose on oocyte viability. Cryobiology 422: 103–111; 2001. doi:10.1006/cryo.2001.2310.
Endoh, K.; Mochida, K.; Ogonuki, N.; Ohkawa, M.; Shinmen, A.; Ito, M.; Kashiwazaki, N.; Ogura, A. The developmental ability of vitrified oocytes from different mouse strains assessed by parthenogenetic activation and intracytoplasmic sperm injection. J Reprod Dev 536: 1199–1206; 2007. doi:10.1262/jrd.19058.
Fulton, B. P.; Whittingham, D. G. Activation of mammalian oocytes by intracellular injection of calcium. Nature 273: 149–151; 1978.
Gook, D. A.; Osborn, S. M.; Johnston, W. I. Cryopreservation of mouse and human oocytes using 1,2-propanediol and the configuration of the meiotic spindle. Hum Reprod 87: 1101–1109; 1993.
Hochi, S.; Kozawa, M.; Fujimoto, T.; Hondo, E.; Yamada, J.; Oguri, N. In vitro maturation and transmission electron microscopic observation of horse oocytes after vitrification. Cryobiology 333: 300–310; 1996. doi:10.1006/cryo.1996.0030.
Huang, J. Y.; Chen, H. Y.; Tan, S. L.; Chian, R. C. Effect of choline-supplemented sodium-depleted slow freezing versus vitrification on mouse oocyte meiotic spindles and chromosome abnormalities. Fertil Steril 884 Suppl: 1093–1100; 2007. doi:10.1016/j.fertnstert.2006.12.066.
Joly, C.; Bchini, O.; Boulekbache, H.; Testart, J.; Maro, B. Effects of 1, 2-propanediol on the cytoskeletal organization of the mouse oocyte. Hum Reprod 7: 374–378; 1992.
Kim, S. H.; Ku, S. Y.; Sung, K. C.; Kang, M. J.; Kim, S. A.; Kim, H. S.; Oh, S. K.; Jee, B. C.; Suh, C. S.; Choi, Y. M.; Kim, J. G.; Moon, S. Y. Simplified EM grid vitrification is a convenient and efficient method for mouse mature oocyte cryopreservation. Yonsei Med J 473: 399–404; 2006.
Kubota, C.; Yang, X.; Dinnyes, A.; Todoroki, J.; Yamakuchi, H.; Mizoshita, K.; Inohae, S.; Tabara, N. In vitro and in vivo survival of frozen–thawed bovine oocytes after IVF, nuclear transfer, and parthenogenetic activation. Mol Reprod Dev 513: 281–286; 1998. doi:10.1002/(SICI)1098-2795(199811)51:3<281::AID-MRD7>3.0.CO;2-L.
Lane, M.; Gardner, D. K. Vitrification of mouse oocytes using a nylon loop. Mol Reprod Dev 583: 342–347; 2001. doi:10.1002/1098-2795(200103)58:3<342::AID-MRD13>3.0.CO;2-X.
Larman, M. G.; Katz-Jaffe, M. G.; Sheehan, C. B.; Gardner, D. K. 1,2-Propanediol and the type of cryopreservation procedure adversely affect mouse oocyte physiology. Hum Reprod 221: 250–259; 2007. doi:10.1093/humrep/del319.
Leibo, S. P. Cryobiology: preservation of mammalian embryos. Basic Life Sci 37: 251–272; 1986.
Martino, A.; Songsasen, N.; Leibo, S. P. Development into blastocysts of bovine oocytes cryopreserved by ultra-rapid cooling. Biol Reprod 545: 1059–1069; 1996. doi:10.1095/biolreprod54.5.1059.
Meng, Q.; Li, X.; Wu, T.; Dinnyés, A.; Zhu, S. Piezo-actuated zona-drilling improves the fertilisation of OPS vitrified mouse oocytes. Acta Vet Hung 553: 369–378; 2007. doi:10.1556/AVet.55.2007.3.11.
Nakagata, N. High survival rate of unfertilized mouse oocytes after vitrification. J Reprod Fertil 872: 479–483; 1989. doi:10.1530/jrf.0.0870479.
O'Neil, L.; Paynter, S. J.; Fuller, B. J.; Shaw, R. W.; DeVries, A. L. Vitrification of mature mouse oocytes in a 6 M Me2SO solution supplemented with antifreeze glycoproteins: the effect of temperature. Cryobiology 371: 59–66; 1998. doi:10.1006/cryo.1998.2098.
Papis, K.; Shimizu, M.; Izaike, Y. Factors affecting the survivability of bovine oocytes vitrified in droplets. Theriogenology 15;545: 651–658; 2000.
Rall, W. F.; Fahy, G. M. Ice-free cryopreservation of mouse embryos at −196°C by vitrification. Nature 313: 573–575; 1985. doi:10.1038/313573a0.
Rall, W. F.; Wood, M. J.; Kirby, C.; Whittingham, D. G. Development of mouse embryos cryopreserved by vitrification. J Reprod Fertil 802: 499–504; 1987. doi:10.1530/jrf.0.0800499.
Saunders, K. M.; Parks, J. E. Effects of cryopreservation procedures on the cytology and fertilization rate of in vitro-matured bovine oocytes. Biol Reprod 611: 178–187; 1999. doi:10.1095/biolreprod61.1.178.
Schalkoff, M. E.; Oskowitz, S. P.; Powers, R. D. Ultrastructural observations of human and mouse oocytes treated with cryopreservatives. Biol Reprod 40: 379–393; 1989. doi:10.1095/biolreprod40.2.379.
Schroeder, A. C.; Champlin, A. K.; Mobraaten, L. E.; Eppig, J. J. Developmental capacity of mouse oocytes cryopreserved before and after maturation in vitro. J Reprod Fertil 891: 43–50; 1990. doi:10.1530/jrf.0.0890043.
Shaw, P. W.; Bernard, A. G.; Fuller, B. J.; Hunter, J. H.; Shaw, R. W. Vitrification of mouse oocytes using short cryoprotectant exposure: effects of varying exposure times on survival. Mol Reprod Dev 332: 210–214; 1992. doi:10.1002/mrd.1080330214.
Shaw, P. W.; Fuller, B. J.; Bernard, A.; Shaw, R. W. Vitrification of mouse oocytes: improved rates of survival, fertilization, and development to blastocysts. Mol Reprod Dev 294: 373–378; 1991. doi:10.1002/mrd.1080290409.
Vajta, G.; Holm, P.; Greve, T.; Callesen, H. Comparison of two manipulation methods to produce in vitro fertilized, biopsied and vitrified bovine embryos. Theriogenology 15;472: 501–509; 1997.
Valojerdi, M. R.; Salehnia, M. Developmental potential and ultrastructural injuries of metaphase II (MII) mouse oocytes after slow freezing or vitrification. J Assist Reprod Genet 223: 119–127; 2005. doi:10.1007/s10815-005-4876-8.
Van der Elst, J. C.; Nerinckx, S. S.; Van Steirteghem, A. C. Slow and ultrarapid freezing of fully grown germinal vesicle-stage mouse oocytes: optimization of survival rate outweighed by defective blastocyst formation. J Assist Reprod Genet 103: 202–212; 1993. doi:10.1007/BF01239222.
Wang, M. K.; Liu, J. L.; Lian, L.; Chen, D. Y. Delivery and storage of single embryos, sperm, or cells in microglass capillaries. In Vitro Cellular & Development Biology-Animal 382: 59–61; 2002.
Wang, Y.; Xiao, Z.; Li, L.; Fan, W.; Li, S. W. Novel needle immersed vitrification: a practical and convenient method with potential advantages in mouse and human ovarian tissue cryopreservation. Hum Reprod 2310: 2256–2265; 2008. doi:10.1093/humrep/den255.
Wassarman, P. M. Regulation of mammalian fertilization by zona pellucida glycoproteins. J Reprod Fertil Suppl 42: 79–87; 1990.
Webb, M.; Howlett, S. K.; Maro, B. Parthenogenesis and cytoskeletal organization in ageing mouse eggs. J Embryol Exp Morphol 95: 131–145; 1986.
Wood, M. J.; Barros, C.; Candy, C. J.; Carroll, J.; Melendez, J.; Whittingham, D. G. High rates of survival and fertilization of mouse and hamster oocytes after vitrification in dimethylsulphoxide. Biol Reprod 493: 489–495; 1993. doi:10.1095/biolreprod49.3.489.
Acknowledgments
We thank Prof. Zhongle Chang for his critical remarks and editing this manuscript. This work was supported by grants from the project of industrialization of good breeds in Shandong province (no. 2008LZ016-06).
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor: J. Denry Sato
Rights and permissions
About this article
Cite this article
Tan, X., Song, E., Liu, X. et al. Factors affecting the survival, fertilization, and embryonic development of mouse oocytes after vitrification using glass capillaries. In Vitro Cell.Dev.Biol.-Animal 45, 420–429 (2009). https://doi.org/10.1007/s11626-009-9195-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11626-009-9195-4