ABSTRACT
Background
The Atrial fibrillation Better Care (ABC) pathway was proposed for a more holistic or integrated care approach to atrial fibrillation (AF) management. We investigated whether adherence with the ABC pathway reduced the risk of adverse clinical outcomes in real-world AF patients starting vitamin K antagonist (VKAs) therapy.
Methods
Prospective cohort study including AF outpatients starting VKA therapy from July 2016 to June 2018. Patients were considered as adherent if all ABC pathway criteria (A: Avoid stroke; B: Better symptom control; and C: Cardiovascular risk factors/comorbidities management) were fulfilled. The primary endpoints were all-cause mortality, net clinical outcomes (NCOs), major adverse cardiovascular events (MACE), and composite thrombotic/thromboembolic events at 2 years.
Results
We enrolled 1045 patients (51.6% female; median age 77 [70–83] years). Of these, 63.0% (658) were adherent to the ABC pathway and 37% (387) were considered non-adherent. Compared to non-adherent patients, those who were ABC adherent had lower event rates for all-cause mortality (13.76 vs. 6.56; p<0.001), NCOs (19.65 vs. 11.94; p<0.001), and MACE (11.88 vs. 7.75; p=0.006) during the follow-up. Adjusted Cox regression analyses demonstrated that the ABC pathway adherent care reduced the risks of all-cause mortality (aHR 0.57, 95% CI 0.42–0.78), NCOs (aHR 0.72, 95% CI 0.56–0.92), and cardiovascular mortality (aHR 0.54, 95% CI 0.32–0.90). Event-free survivals for all-cause mortality, NCOs (both log-rank p-values <0.001), and MACE (log-rank p-value = 0.004) were also higher in ABC pathway adherent patients.
Conclusions
In this real-world prospective cohort of AF patients starting VKA therapy, adherence to the ABC pathway management at baseline significantly reduced the risk of NCOs, all-cause mortality, and cardiovascular death at 2 years.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
INTRODUCTION
The management of atrial fibrillation (AF) has evolved during the last decade. The search for a more comprehensive, integrated, and holistic management of AF patients had led to the proposal of the Atrial fibrillation Better Care (ABC) pathway1. This simple strategy, which aims to streamline primary and secondary care of patients with AF, is based on three pillars: “A” avoid stroke; “B” better symptom control; and “C” cardiovascular risk factor and other comorbidities management1.
Recognizing this need for a more integrated care approach to AF management, the 2020 European Society of Cardiology (ESC) guidelines on the diagnosis and management of AF have promoted use of the ABC pathway as a simplified and concise approach that integrates the care of AF patients across various levels of healthcare professionals and between specialties, as well as facilitating patient engagement2. The key message is that the principals of AF management could be as “Easy as ABC….” (3,4,5).
The ABC pathway has been tested in retrospective post hoc analyses of clinical trial cohorts6,7, nationwide claims data8, observational cohorts9,10, and one prospective cluster randomized trial, the mAFA-II trial11. In all studies, there was general consistency that ABC pathway adherent care was associated with a reduction in clinical outcomes, and in the long-term extension cohort from the mAFA-II trial there was good adherence and persistence12. Despite the ABC pathway provides a simple decision-making framework to enable consistent equitable care, the proportion of adherent patients is overall suboptimal, and this is associated with a higher risk of major adverse outcomes13,14.
Given the relatively limited prospective evidence of the relationship of the ABC pathway and adverse events in AF patients, particularly in those taking oral anticoagulation (OAC) therapy with vitamin K antagonists (VKAs), we aimed to investigate if adherence to this approach at baseline reduced the risk of adverse clinical outcomes in real-world AF patients starting VKA therapy.
METHODS
A detailed description of the prospective Murcia AF Project Phase II cohort has previously been published15. In brief, this is a prospective observational cohort study including outpatients newly diagnosed with AF and naïve for OAC in an anticoagulation clinic of a tertiary hospital (Murcia, Spain), from July 1, 2016, to June 30, 2018. Eligible patients were those who started OAC with VKAs for the first time. Only those patients older than 18 years with documented evidence of AF on ECG and not previously anticoagulated for another reason were included. Patients with prosthetic heart valves, rheumatic mitral valves, or other type of severe valvular AF were excluded. In order to perform a prospective cohort study that reliably reflects the “real-world” clinical practice, no other exclusion criteria were established. At baseline, a complete medical history was obtained by collecting socio-demographic and anthropometric data, comorbidities, concomitant therapies, and results of the most recent lab tests. In addition, stroke (CHA2DS2-VASc) and bleeding (HAS-BLED) risk scores were calculated.
The study protocol was approved by the Ethics Committee from the University Hospital Morales Meseguer (reference: EST: 20/16) and was carried out in accordance with the ethical standards established in the 1964 Declaration of Helsinki and its subsequent amendments. Informed consent was required for participation in this study.
Atrial Fibrillation Better Care Pathway Assessment
The ABC pathway was evaluated at baseline according to its original definition, as follows:
-
“A” Criterion: A patient would qualify as adherent for this criterion if properly prescribed and treated with an OAC. As all patients were included in the context of starting VKA therapy (which is common practice in Spain, where a trial of VKA is mandated before consideration of direct-acting OACs [DOACs]) and no previous data about the time in therapeutic range (TTR) were available, the “A” criterion was considered fulfilled if VKA was correctly prescribed according to thromboembolic risk (i.e., CHA2DS2-VASc >1 in males or CHA2DS2-VASc ≥2 in females).
-
“B” Criterion: Defined as the presence of symptoms related to AF, classified by the recommended European Heart Rhythm Association (EHRA) symptom scale. Any patient with an EHRA score of I (no symptoms) or II (mild symptoms not affecting daily life) was considered adherent to this criterion whereas patients with EHRA score of III (severe symptoms) or IV (disabling symptoms) were considered non-adherent. Data on symptoms were collected at baseline.
-
“C” Criterion: Defined as the optimal management/medical treatment of the main cardiovascular comorbidities: hypertension, coronary artery disease, peripheral artery disease, heart failure, stroke/transient ischemic attack (TIA), and diabetes mellitus. Optimal medical treatment was defined as follows: (i) for hypertension, we considered controlled blood pressure if <160/90 mmHg was recorded at baseline and treated with appropriate drugs; (ii) for coronary artery disease, treatment with angiotensin-converting enzyme (ACE) inhibitors, beta-blockers, and statins; (iii) for peripheral artery disease, treatment with statins; (iv) for previous stroke/TIA, treatment with statins; (v) for heart failure, we considered treatment with ACE inhibitors/angiotensin receptor blockers and beta-blockers; (vi) for diabetes mellitus, treatment with insulin or oral antidiabetics. To be included as adherent to “C” criterion, all considered risk factors should have been well controlled and/or treated with appropriate cardiovascular preventive drugs.
A patient was considered as fully ABC pathway adherent (“ABC adherent care”) if all the three criteria were fulfilled.
Follow-up and Clinical Outcomes
Follow-up was performed according to the standard of care at each routine visit to the outpatient anticoagulation clinic or visits for the anticoagulation control. If the patient never attends to these visits, medical records and telephone calls were used to obtain the information needed and vital status, with no specific interventions and no specific visits for study purposes. Follow-up was extended for 2 years in ABC pathway adherent and non-adherent patients, with no difference between both groups. During this period, all adverse events were recorded. Of note, patients lost to follow-up were <1% of the overall sample and thus unlikely to skew results with a sensitivity analysis evaluating them as intention-to-treat.
For the present study, the primary endpoints were all-cause mortality, net clinical outcomes (as the composite of major bleeding, ischemic stroke/TIA, and all-cause mortality), major adverse cardiovascular events (MACE, as the composite of fatal/nonfatal myocardial infarction, cardiovascular death, and ischemic stroke/TIA), and composite thrombotic/thromboembolic events (any of the following: myocardial infarction, ischemic stroke/TIA, venous thromboembolism [VTE, including both deep vein thrombosis and pulmonary embolism]). Secondary outcomes were the individual outcomes of ischemic stroke, TIA, myocardial infarction, VTE, major bleeding (defined based on 2005 International Society on Thrombosis and Haemostasis (ISTH) criteria (16)), intracranial hemorrhage (ICH), clinically relevant non-major bleeding (CRNMB, according to the 2015 ISTH criteria (17)), and cardiovascular death. The investigators identified, confirmed, and recorded all clinical outcomes.
Statistical Analyses
Continuous variables were expressed as mean ± standard deviation (SD) or median and interquartile range (IQR) as appropriate, while categorical variables were expressed as absolute frequencies and percentages. The Pearson Chi-squared test was used to compare proportions and differences between continuous and categorical variables were assessed using the Mann-Whitney U test or the Student t test, as appropriate.
Cox proportional hazard regression models were performed to determine the association between the ABC pathway and the primary endpoints. A univariate significance level of 0.05 was required to allow a variable into the multivariate model (SLENTRY = 0.05) and a multivariate significance level of 0.05 was required for a variable to stay in the model (SLSTAY = 0.05). Results were reported as hazard ratio (HR) with 95% confidence interval (CI).
Annual event rates with their Poisson 95% CI were calculated for ABC pathway adherent and non-adherent as the number of adverse clinical outcomes divided by the exposure period in patients-years (PYs), and expressed as number of events per 100 PYs. The difference between two annual event rates and the associated p-value was calculated. Annual event rates (i.e., incidence rates) were also compared and reported as incidence rate ratio (IRR). To calculate IRR, the event rates for every endpoint in the non-adherent group (R1) were divided by the event rates for every endpoint in the adherent group (R2). An IRR <1 indicated that the incident rate was lower in the non-adherent group compared to that in the adherent group. An IRR >1 indicated that the incident rate was higher in the non-adherent group compared to that in the adherent group. IRRs = 1 indicated no differences in the incidence rates of both groups. Finally, survival analyses by Kaplan-Meier estimates were performed to assess differences in event-free survival distributions, which were compared using the log-rank test.
A p-value <0.05 was accepted as statistically significant. Statistical analyses were performed using SPSS v. 25.0 (SPSS, Inc., Chicago, IL, USA), and MedCalc v. 16.4.3 (MedCalc Software bvba, Ostend, Belgium) for Windows.
RESULTS
Overall, 1254 AF patients were initially evaluated. Of these, 1064 patients fulfilled inclusion/exclusion criteria and were included, but 14 patients were lost to follow-up giving a final study cohort of 1050 patients. Of these, 1045 (51.6% female; median age 77, IQR 70–83 years) with a median CHA2DS2-VASc of 4 (IQR 3–5) and HAS-BLED of 2 (IQR 2–3), had complete data for the present analysis. Table 1 gives a summary of baseline clinical characteristics.
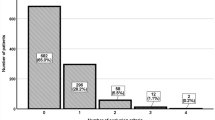
Regarding the ABC pathway, 97.3% (n = 1017) of patients fulfilled “A” criterion at baseline; 85.2% (n = 890) fulfilled “B” criterion; and 77.4% (n = 809) fulfilled “C” criterion. Overall, 3.0% (32) of patients were adherent to only one criterion, 34.0% (355) were adherent to two criteria and 63.0% (658) were adherent to the three criteria. Thus, 63.0% (658) were categorized as adherent to the ABC pathway at baseline, whereas 37% (387) were considered not adherent.
Although there was no significant difference in age, patients non-adherent to the ABC pathway presented with more prevalent diabetes, heart failure, renal impairment, and vascular disease (both coronary and peripheral artery diseases), as well as with higher CHA2DS2-VASc and HAS-BLED scores, compared to adherent patients (Table 1).
In terms of OAC therapy, the mean TTR was lower in those non-adherent to the ABC pathway compared to ABC-adherent patients (59.4%±22.3% vs. 63.9%±21.1%; p=0.002), and among those non-adherent to the ABC pathway at baseline, a higher proportion presented with a TTR <65% than TTR ≥65% (56.4% vs. 43.6%, p=0.023). In addition, 144 (21.9%) patients adherent to the ABC pathway switched to a DOAC during the follow-up, and 67 (17.3%) non-adherent patients did so (p=0.195).
Clinical Outcomes During the Follow-up
During a median follow-up of 2 years, there were 172 (16.5%) all-cause deaths, 261 (25.0%) net clinical outcomes, 164 (15.7%) MACEs, and 110 (10.5%) composite thrombotic/thromboembolic events. Compared to patients non-adherent to the ABC pathway, compliant patients showed significantly lower event rates for the primary outcomes of net clinical outcomes (19.65 [95% CI 16.32–23.46] vs. 11.94 [95% CI 10.04–14.10] per 100 PYs; p<0.001) and MACE (11.88 [95% CI 9.32–14.91] vs. 7.75 [95% CI 6.23–9.52] per 100 PYs; p=0.006). The primary outcome of all-cause mortality was higher in patients non-adherent to the ABC pathway than that in adherent patients (13.76 [95% CI 11.08–16.90] vs. 6.56 [95% CI 5.21–8.15] per 100 PYs; p<0.001) with IRR of 2.10 (95% CI 1.54–2.87) and this was also observed for cardiovascular death (IRR 2.41, 95% CI 1.51–3.89, p<0.001).
A detailed comparison of annual event rates and incidence rate ratios according to the ABC pathway groups is shown in Table 2.
Univariate and Multivariate Analyses
Regarding the risk assessment, univariate analyses showed that all risks of outcomes were lower in patients adherent to the ABC pathway, being significant for all-cause mortality, cardiovascular mortality, net clinical outcomes, and MACE (Fig. 1, panel A). After adjusting for several comorbidities, the ABC adherent care favored the reduction of the risk of all-cause mortality (aHR 0.57, 95% CI 0.42–0.78), cardiovascular mortality (aHR 0.54, 95% CI 0.32–0.90), and net clinical outcomes (aHR 0.72, 95% CI 0.56–0.92) (Fig. 1, panel B). Adjusted HRs for the primary outcomes according to the number of ABC pathway criteria fulfilled are shown in Supplementary Figure 1.
Forest plot of non-adjusted and adjusted hazard ratios for the primary and secondary outcomes according to the ABC pathway adherent care. TIA, transient ischemic attack; CRNMB, clinically relevant non-major bleeding; MACE, major adverse cardiovascular events. *Adjusted hazard ratios by the following variables: age, sex, hypertension, diabetes, ischemic stroke/TIA/SE, vascular disease, heart failure, chronic kidney disease, history of bleeding, alcohol abuse, hepatic disease, and cancer.
Kaplan-Meier analyses showed that event-free survival was generally reduced in non-adherent patients to the ABC pathway as compared to adherent patients. At 2 years, non-adherent patients presented significantly lower survival in terms of net clinical outcomes and all-cause mortality (both log-rank p-values <0.001), as well as MACE (log-rank p-value = 0.004), but not in terms of composite thrombotic/thromboembolic events (log-rank p-value = 0.411) (Fig. 2).
DISCUSSION
In this prospective cohort study including real-world AF patients, we found that patients adherent to the ABC pathway at baseline had a significantly lower risk of net clinical outcomes, all-cause mortality, and cardiovascular death.
Despite differences in some comorbidities between ABC pathway adherent and non-adherent patients, many patients in the non-adherent group were included in such group not exclusively for having a particular comorbidity but for having the comorbidity not correctly managed. Therefore, the differential point between adherent and non-adherent patients is not the proportion of comorbidities itself but if they were appropriately addressed. Moreover, the risks of net clinical outcomes, all-cause mortality, and cardiovascular death are presented after adjustment for specific comorbidities, including those that were significantly different between groups. This novel approach, not so focused on the disease itself but more on the patient, could be useful in our population, and represents a paradigm shift in relation to the management of patients with AF, which has tended to focus on individual strategies (e.g., stroke prevention, or rate vs. rhythm control).
Although this is the first prospective study investigating the usefulness of the ABC pathway in our national context, overall, our results reinforce previous retrospective observations, despite that the proportion of adherent patients to the ABC pathway was higher in our cohort compared to prior studies.
Since its first description in 20171, the ABC pathway has been tested in different regions of the world showing that better adherence improves patient outcomes. For example, Proietti et al. first performed a post hoc analysis in 3169 AF patients from the AFFIRM trial. The authors found that patients managed according to the ABC pathway had lower risk of all-cause death (aHR 0.35; 95% CI 0.17–0.75), composite outcome of stroke/major bleeding/cardiovascular death (aHR 0.35; 95% CI 0.18–0.68), and first hospitalization (aHR 0.65; 95% CI 0.53–0.80)6. Subsequently, Pastori et al. investigated the impact of implementing the ABC pathway on cardiovascular events in consecutive real-world AF patients, showing significantly lower risks of the composite outcome of fatal/nonfatal ischemic stroke and myocardial infarction (MI), TIA, cardiac revascularization, and cardiovascular death (aHR, 0.44, 95% CI 0.24–0.80)9. The ABC pathway is particularly beneficial in men and in AF patients with a 2MACE score ≥3, since these presented a higher risk of MACE10.
Another study using a nationwide population cohort demonstrated that patients who complied with the ABC pathway presented a significantly lower risk of all-cause death (aHR 0.82; 95% CI 0.78–0.86) and the composite outcome of all-cause mortality, ischemic stroke, major bleeding, and myocardial infarction (aHR 0.86; 95% CI 0.83–0.89). Importantly, the risk of all-cause death and composite outcome were progressively lowered with the increasing numbers of ABC pathway criteria fulfilled8. In a retrospective analysis from the Gulf SAFE registry, an observational study including AF patients from the Middle East, patients adherent to the ABC shower significantly lower risk of the composite outcome of ischemic stroke/systemic embolism, all-cause mortality, and cardiovascular hospitalization (aHR 0.53; 95% CI 0.36–0.8) and all-cause mortality alone (aHR 0.46; 95% CI 0.25–0.86)19. Similarly, ABC pathway compliance showed an independent association with reduction of all-cause death and the composite of all-cause death, ischemic stroke, and intracranial hemorrhage in a large cohort of Chinese AF patients20.
The ABC pathway has also been introduced using mobile health (mHealth) technology for AF care. For example, in a cluster randomized trial that randomized AF patients to usual care or to an integrated care approach based on a mobile AF Application (mAFA) incorporating the ABC pathway, the mAFA intervention decreased the risk of the composite outcome of “ischemic stroke/systemic thromboembolism, death, and rehospitalization” (HR 0.39; 95% CI 0.22–0.67) and rehospitalization (HR 0.32; 95% CI 0.17–0.60)11. Finally, the impact on outcomes of an ABC adherent management has been evaluated in the ESC-EHRA EURObservational Research Programme in AF General Long-Term Registry. After adjusting for several confounding factors, again the ABC adherent care showed an association with a lower risk of the composite of any thromboembolism/acute coronary syndrome/cardiovascular death (HR 0.59, 95% CI 0.44–0.79), cardiovascular death (HR 0.52, 95% CI 0.35–0.78), and all-cause death (HR 0.57, 95% CI 0.43–0.78)21. Indeed, the clinical usefulness of the ABC pathway is evident even in high-risk populations such as frailty, diabetes, or those with multiple comorbidities, polypharmacy, or prior hospitalizations7,22,23,24; and application of the ABC pathway may even reduce healthcare costs related to cardiovascular events25.
However, most of the studies regarding the ABC pathway were performed in DOAC-treated AF populations whereas VKAs are still widely used in several countries globally. In such patients, quality of anticoagulation and appropriate TTR is central. Recently, we have reported that the ABC pathway adherent patients had better TTR, and more ABC criteria fulfilled increased the probability of achieving good TTR26. Thus, ABC pathway management is not only associated with a lower number of clinical outcomes but also results in better anticoagulation control, which in turn is associated with a better prognosis.
The above evidence reinforces the hypothesis that AF management requires a holistic approach. In fact, not only do the 2020 ESC guidelines for the management of AF2, 2021 Asia Pacific Heart Rhythm Society guidelines27, the 2018 CHEST guidelines28, and the Korean Heart Rhythm Society guidelines29 suggest focusing on such approach, but the 2020 Canadian Cardiovascular Society/Canadian Heart Rhythm Society guidelines also suggest a structured, integrated, multidisciplinary, patient-focused approach for patients with AF30.
Beyond stroke prevention, symptom management and control of cardiovascular risk factors are equally needed in AF. Indeed, multi-morbidity is common among AF patients and contributes to worse clinical outcomes and quality of life31. Therefore, the importance and necessity of modifying cardiovascular risk factors is crucial, and this has demonstrated to decrease disease burden and progression32. Integrated care, by putting the patient into the center and including other healthcare professionals, aids this objective18,33. Previous specialized AF clinics and nurse-led AF clinics have already included this approach with promising results34,35,36.
Limitations
The main limitation of the study lies in its observational nature, with a Caucasian-based population and single-center design. Another potential limitation is patient selection since we included only patients starting OAC therapy with VKA for the first time. Previous studies have shown that the initial period of OAC is characterized by an increased risk of adverse events, particularly bleeding ones, and during the first 3 months of VKA therapy, which may have some influence on the results37,38,39. However, our dataset was collected prospectively, under a careful follow-up. Thus, all events (even very early ones) were recorded. Importantly, patients lost to follow-up were excluded for the present analysis.
We must recognize a potential drug bias because inter-physician changes of drugs prescription might have an impact on the ABC adherent care and therefore on clinical outcomes. However, we specifically tested the impact of baseline adherence to the ABC pathway on mid-term outcomes. Although several physicians could be responsible for pharmacological therapies prescribed to patients, the same hematologist from an anticoagulation clinic was in charge of all patients of the study, thus avoiding the bias of different criteria from different hematologists regarding anticoagulation therapy. Notwithstanding, the management may differ in other settings, and the generalization to other centers with less intense follow-up or including mainly patients under DOACs requires further investigation. Finally, we did not revaluate adherence to the ABC pathway during the 2 years follow-up, which we recognize could have some impact on clinical outcomes.
CONCLUSIONS
In a large prospective cohort of real-world AF patients starting VKA therapy, adherence to ABC pathway management was demonstrated to significantly reduce the risk of net clinical outcomes, all-cause mortality, and cardiovascular death at 2 years. A structured, holistic, and integrated care approach, based on the ABC pathway, is advantageous for AF management, improving patient outcomes.
References
Lip GYH. The ABC pathway: an integrated approach to improve AF management. Nat Rev Cardiol. 2017;14(11):627-8.
Hindricks G, Potpara T, Dagres N, Arbelo E, Bax JJ, Blomström-Lundqvist C, et al. 2020 ESC Guidelines for the diagnosis and management of atrial fibrillation developed in collaboration with the European Association of Cardio-Thoracic Surgery (EACTS). Eur Heart J. 2021;42(5):373-498.
Mahmood M, Lip GY. Management of atrial fibrillation: easy as ABC. Minerva Med. 2019;110(1):27-34.
Ding WY, Lip GYH, Potpara TS. Atrial fibrillation: Can it be as easy as CC to ABC? Eur J Clin Invest. 2020;50(11):e13413.
Boriani G, Vitolo M, Lane DA, Potpara TS, Lip GY. Beyond the 2020 guidelines on atrial fibrillation of the European society of cardiology. Eur J Intern Med. 2021.
Proietti M, Romiti GF, Olshansky B, Lane DA, Lip GYH. Improved Outcomes by Integrated Care of Anticoagulated Patients with Atrial Fibrillation Using the Simple ABC (Atrial Fibrillation Better Care) Pathway. Am J Med. 2018;131(11):1359-66.e6.
Proietti M, Romiti GF, Olshansky B, Lane DA, Lip GYH. Comprehensive Management With the ABC (Atrial Fibrillation Better Care) Pathway in Clinically Complex Patients With Atrial Fibrillation: A Post Hoc Ancillary Analysis From the AFFIRM Trial. J Am Heart Assoc. 2020;9(10):e014932.
Yoon M, Yang PS, Jang E, Yu HT, Kim TH, Uhm JS, et al. Improved Population-Based Clinical Outcomes of Patients with Atrial Fibrillation by Compliance with the Simple ABC (Atrial Fibrillation Better Care) Pathway for Integrated Care Management: A Nationwide Cohort Study. Thromb Haemost. 2019;119(10):1695-703.
Pastori D, Pignatelli P, Menichelli D, Violi F, Lip GYH. Integrated Care Management of Patients With Atrial Fibrillation and Risk of Cardiovascular Events: The ABC (Atrial fibrillation Better Care) Pathway in the ATHERO-AF Study Cohort. Mayo Clin Proc. 2019;94(7):1261-7.
Pastori D, Menichelli D, Violi F, Pignatelli P, Lip GYH. The Atrial fibrillation Better Care (ABC) pathway and cardiac complications in atrial fibrillation: a potential sex-based difference. The ATHERO-AF study. European Journal of Internal Medicine. 2020;85:80-5.
Guo Y, Lane DA, Wang L, Zhang H, Wang H, Zhang W, et al. Mobile Health Technology to Improve Care for Patients With Atrial Fibrillation. J Am Coll Cardiol. 2020;75(13):1523-34.
Guo Y, Guo J, Shi X, Yao Y, Sun Y, Xia Y, et al. Mobile health technology-supported atrial fibrillation screening and integrated care: A report from the mAFA-II trial Long-term Extension Cohort. Eur J Intern Med. 2020;82:105-11.
Stevens D, Harrison SL, Kolamunnage-Dona R, Lip GYH, Lane DA. The Atrial Fibrillation Better Care pathway for managing atrial fibrillation: a review. Europace. 2021;23(10):1511-27.
Romiti GF, Pastori D, Rivera-Caravaca JM, Ding WY, Gue YX, Menichelli D, et al. Adherence to the 'Atrial Fibrillation Better Care' Pathway in Patients with Atrial Fibrillation: Impact on Clinical Outcomes-A Systematic Review and Meta-Analysis of 285,000 Patients. Thromb Haemost. 2021;122(3):406-14.
Rivera-Caravaca JM, Marín F, Esteve-Pastor MA, Gálvez J, Lip GYH, Vicente V, et al. Murcia atrial fibrillation project II: protocol for a prospective observational study in patients with atrial fibrillation. BMJ Open. 2019;9(12):e033712.
Schulman S, Kearon C. Definition of major bleeding in clinical investigations of antihemostatic medicinal products in non-surgical patients. J Thromb Haemost. 2005;3(4):692-4.
Kaatz S, Ahmad D, Spyropoulos AC, Schulman S. Definition of clinically relevant non-major bleeding in studies of anticoagulants in atrial fibrillation and venous thromboembolic disease in non-surgical patients: communication from the SSC of the ISTH. J Thromb Haemost. 2015;13(11):2119-26.
Gallagher C, Hendriks JM, Nyfort-Hansen K, Sanders P, Lau DH. Integrated care for atrial fibrillation: the heart of the matter. Eur J Prev Cardiol. 2020.
Gumprecht J, Domek M, Proietti M, Li YG, Asaad N, Rashed W, et al. Compliance of Atrial Fibrillation Treatment with the Atrial Fibrillation Better Care (ABC) Pathway Improves the Clinical Outcomes in the Middle East Population: A Report from the Gulf Survey of Atrial Fibrillation Events (SAFE) Registry. J Clin Med. 2020;9(5).
Wang YF, Jiang C, He L, Du X, Sang CH, Long DY, et al. Integrated Care of Atrial Fibrillation Using the ABC (Atrial fibrillation Better Care) Pathway Improves Clinical Outcomes in Chinese Population: An Analysis From the Chinese Atrial Fibrillation Registry. Front Cardiovasc Med. 2021;8:762245.
Proietti M, Lip GYH, Laroche C, Fauchier L, Marin F, Nabauer M, et al. Relation of outcomes to ABC (Atrial Fibrillation Better Care) pathway adherent care in European patients with atrial fibrillation: an analysis from the ESC-EHRA EORP Atrial Fibrillation General Long-Term (AFGen LT) Registry. Europace. 2020.
Yang PS, Sung JH, Jang E, Yu HT, Kim TH, Lip GYH, et al. Application of the simple atrial fibrillation better care pathway for integrated care management in frail patients with atrial fibrillation: A nationwide cohort study. J Arrhythm. 2020;36(4):668-77.
Domek M, Gumprecht J, Li YG, Proietti M, Rashed W, Al Qudaimi A, et al. Compliance of atrial fibrillation treatment with the ABC pathway in patients with concomitant diabetes mellitus in the Middle East based on the Gulf SAFE registry. Eur J Clin Invest. 2021;51(3):e13385.
Proietti M, Vitolo M, Lip GYH. Integrated care and outcomes in patients with atrial fibrillation and comorbidities. Eur J Clin Invest. 2021:e13498.
Pastori D, Farcomeni A, Pignatelli P, Violi F, Lip GY. ABC (Atrial fibrillation Better Care) Pathway and Healthcare Costs in Atrial Fibrillation: The ATHERO-AF Study. Am J Med. 2019;132(7):856-61.
Roldán V, Martínez-Montesinos L, López-Gálvez R, García-Tomás L, Lip GYH, Rivera-Caravaca JM, et al. Relation of the 'Atrial Fibrillation Better Care (ABC) Pathway' to the Quality of Anticoagulation in Atrial Fibrillation Patients Taking Vitamin K Antagonists. J Pers Med. 2022;12(3).
Chao TF, Joung B, Takahashi Y, Lim TW, Choi EK, Chan YH, et al. 2021 Focused Update Consensus Guidelines of the Asia Pacific Heart Rhythm Society on Stroke Prevention in Atrial Fibrillation: Executive Summary. Thromb Haemost. 2022;122(1):20-47.
Lip GYH, Banerjee A, Boriani G, Chiang CE, Fargo R, Freedman B, et al. Antithrombotic Therapy for Atrial Fibrillation: CHEST Guideline and Expert Panel Report. Chest. 2018;154(5):1121-201.
Joung B, Lee JM, Lee KH, Kim TH, Choi EK, Lim WH, et al. 2018 Korean Guideline of Atrial Fibrillation Management. Korean Circ J. 2018;48(12):1033-80.
Andrade JG, Aguilar M, Atzema C, Bell A, Cairns JA, Cheung CC, et al. The 2020 Canadian Cardiovascular Society/Canadian Heart Rhythm Society Comprehensive Guidelines for the Management of Atrial Fibrillation. Can J Cardiol. 2020;36(12):1847-948.
Lip GYH, Tran G, Genaidy A, Marroquin P, Estes C, Harrelll T. Prevalence/incidence of atrial fibrillation based on integrated medical/pharmacy claims, and association with co-morbidity profiles/multi-morbidity in a large US adult cohort. Int J Clin Pract. 2021:e14042.
Gallagher C, Middeldorp ME, Hendriks JM, Lau DH, Sanders P. Lifestyle as a Risk Factor for Atrial Fibrillation. Card Electrophysiol Clin. 2021;13(1):263-72.
Bhat A, Khanna S, Chen HHL, Gupta A, Gan GCH, Denniss AR, et al. Integrated Care in Atrial Fibrillation: A Road Map to the Future. Circ Cardiovasc Qual Outcomes. 2021:Circoutcomes120007411.
Pluymaekers N, Hermans ANL, van der Velden RMJ, Gawałko M, den Uijl DW, Buskes S, et al. Implementation of an on-demand app-based heart rate and rhythm monitoring infrastructure for the management of atrial fibrillation through teleconsultation: TeleCheck-AF. Europace. 2020.
Rivera-Caravaca JM, Gil-Perez P, Lopez-García C, Veliz-Martínez A, Quintana-Giner M, Romero-Aniorte AI, et al. A nurse-led atrial fibrillation clinic: Impact on anticoagulation therapy and clinical outcomes. Int J Clin Pract. 2020;74(12):e13634.
van den Dries CJ, van Doorn S, Rutten FH, Oudega R, van de Leur S, Elvan A, et al. Integrated management of atrial fibrillation in primary care: results of the ALL-IN cluster randomized trial. Eur Heart J. 2020;41(30):2836-44.
Hylek EM, Evans-Molina C, Shea C, Henault LE, Regan S. Major hemorrhage and tolerability of warfarin in the first year of therapy among elderly patients with atrial fibrillation. Circulation. 2007;115(21):2689-96.
Garcia DA, Lopes RD, Hylek EM. New-onset atrial fibrillation and warfarin initiation: high risk periods and implications for new antithrombotic drugs. Thromb Haemost. 2010;104(6):1099-105.
Granger CB, Lopes RD, Hanna M, Ansell J, Hylek EM, Alexander JH, et al. Clinical events after transitioning from apixaban versus warfarin to warfarin at the end of the Apixaban for Reduction in Stroke and Other Thromboembolic Events in Atrial Fibrillation (ARISTOTLE) trial. Am Heart J. 2015;169(1):25-30.
Funding
Open Access funding provided thanks to the CRUE-CSIC agreement with Springer Nature. This work was supported by the Spanish Ministry of Economy, Industry, and Competitiveness, through the Instituto de Salud Carlos III after independent peer review (research grant: PI17/01375 co-financed by the European Regional Development Fund) and group CB16/11/00385 from CIBERCV.
Author information
Authors and Affiliations
Author notes
Gregory Y.H. Lip and Francisco Marín are Joint senior authors
Contributions
José Miguel Rivera-Caravaca, Vanessa Roldán, and Lorena Martínez-Montesinos contributed to data collection, performed statistical analyses, and drafted the manuscript. Vicente Vicente critically revised the manuscript. Gregory Y.H. Lip and Francisco Marín conceived the study and critically revised the manuscript. All the authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
GYHL: Consultant and speaker for BMS/Pfizer, Boehringer Ingelheim and Daiichi-Sankyo. No fees are received personally.
There is nothing to disclose for other authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 177 kb)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Rivera-Caravaca, J.M., Roldán, V., Martínez-Montesinos, L. et al. The Atrial Fibrillation Better Care (ABC) Pathway and Clinical Outcomes in Patients with Atrial Fibrillation: the Prospective Murcia AF Project Phase II Cohort. J GEN INTERN MED 38, 315–323 (2023). https://doi.org/10.1007/s11606-022-07567-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11606-022-07567-5