Abstract
Background
Experience with neoadjuvant chemoradiation (CXRT) has raised questions regarding the additional benefit of surgery after locally advanced esophageal adenocarcinoma patients achieve a clinical response to CXRT. We sought to quantify the value of surgery by comparing the overall (OS) and disease-free survival (DFS) of trimodality-eligible patients treated with definitive CXRT vs. CXRT followed by esophagectomy.
Methods
We identified 143 clinical stage III esophageal adenocarcinoma patients that were eligible for trimodality therapy. All patients successfully completed neoadjuvant CXRT and were considered appropriate candidates for resection. Patients that were medically inoperable were excluded. Cox regression models were used to identify significant predictors of survival.
Results
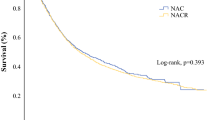
Among the 143 patients eligible for surgery after completing CXRT, 114 underwent resection and 29 did not. Poorly differentiated tumors (HR = 2.041, 95% CI = 1.235–3.373) and surgical resection (HR = 0.504, 95% CI = 0.283–0.899) were the only independent predictors of OS. Patients treated with surgery had a 50 and 54 % risk reduction in overall and cancer-specific mortality, respectively. Median OS (41.2 vs. 20.3 months, p = 0.012) and DFS (21.5 vs. 11.4 months, p = 0.007) were significantly improved with the addition of surgery compared to definitive CXRT.
Conclusions
Surgery provides a significant survival benefit to trimodality-eligible esophageal adenocarcinoma patients with locally advanced disease.





Similar content being viewed by others
References
Cen P, Banki F, Cheng L, Khali K, Du XL, Fallon M, Amaot RJ, Kaiser LR. Changes in age, stage distribution, and survival of patients with esophageal adenocarcinoma over three decades in the United States. Ann Surg Oncol 2012; 19(5):1685–1691.
Simard EP, Ward EM, Siegel R, Jemal A. Cancers with increasing incidence trends in the United States: 1999 through 2008. CA Cancer J Clin 2012; 62(2):118–128.
Gebski V, Burmeister B, Smithers BM, Foo K, Zalcberg J, Simes J. Survival benefits from neoadjuvant chemoradiotherapy or chemotherapy in oesophageal carcinoma: a meta-analysis. Lancet Oncol 2007; 8:226–234.
Fiorica F, Di Bona D, Schepis F, Licata A, Shahied L, Venturi A, Falchi AM, Craxi A, Camma C. Preoperative chemoradiotherapy for oesophageal cancer: a systematic review and meta-analysis. Gut 2004; 53:925–930.
Hofstetter W, Swisher SG, Correa AM, Hess K, Putnam JB, Ajani JA, Dolormente M, Francisco R, Komaki RR, Lara A, Martin F, Rice DC, Sarabia AJ, Smythe WR, Vaporciyan AA, Walsh GL, Roth JA. Treatment outcomes of resected esophageal cancer. Ann Surg 2002; 236(3):376–84.
Kim JY, Hofstetter WL. Esophagectomy after chemoradiation: who and when to operate. Semin Thoracic Cardiovasc Surg 2012; 24:288–293.
Swisher SG, Winter KA, Komaki RU, Ajani JA, Wu TT, Hofstetter WL, Konski AA, Willett CG. A Phase II study of a paclitaxel-based chemoradiation regimen with selective surgical salvage for resectable locoregionally advanced esophageal cancer: initial reporting of RTOG 0246. Int J Radiat Oncol Biol Phys 2012; 82(5):1967–1972.
Marks JL, Hofstetter W, Correa AM, Mehran RJ, Rice D, Roth J, Walsh G, Vaporciyan A, Erasmus J, Chang J, Maru D, Lee JH, Lee J, Ajani JA, Swisher SG. Salvage esophagectomy after failed definitive chemoradiation for esophageal adenocarcinoma. Ann Thorac Surg 2012; 94(4):1126–32.
Murphy CC, Hofstetter WL, Correa AM, Ajani JA, Komaki RU, Swisher SG. Utilization of surgery in trimodality eligible patients with locally advanced adenocarcinoma in a non-protocol setting. Dis Esophagus 2013. doi:10.1111/dote.12019.
American Joint Committee on Cancer. AJCC Cancer Staging Manual. 6th ed. Philadelphia: Lippincott-Raven; 2002, pp. 91–98.
Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chron Dis. 1987; 40(5):373–385.
D’Agostino RB. Propensity score methods for bias reduction in the comparison of a treatment to a non-randomized control group. Stat Med. 1998; 17:2265–2281.
Lordick F, Ott K, Krause BJ, Weber WA, Becker K, Stein HJ, Lorenzen S, Schuster T, Wieder H, Hermann K, Bredenkamp R, Hofler H, Fink U, Peschel C, Schwaiger M, Siewert JR. PET to assess early metabolic response and to guide treatment of adenocarcinoma of the oesophagogastric junction: the MUNICON phase II trial. Lancet Oncol 2007; 8:797–805.
Tougeron D, Scotte M, Hamidou H, Di Fiore F, Paillot B, Michot F, Michel P. Definitive chemoradiotherapy in patients with esophageal adenocarcinoma: an alternative to surgery? J Surg Oncol 2012; 105:761–766.
McKenzie S, Mailey B, Artinyan A, Metchikian M, Shibata S, Kernstine K, Kim J. Improved outcomes in the management of esophageal cancer with the addition of surgical resection to chemoradiation therapy. Ann Surg Oncol 2011; 18:551–558.
Merkow RP, Bilimoria KY, McCarter MD, Chow WB, Gordon HS, Stewart AK, Ko CY, Bentrem DJ. Effect of histologic subtype on treatment and outcomes for esophageal cancer in the United States. Cancer 2012; 118(13):3268–3276.
Weber WA, Ott K, Becker K, Dittler HJ, Helmberger H, Avril NE, Meisetschlager G, Busch R, Siewert JR, Schwaiger M, Fink U. Prediction of response to preoperative chemotherapy in adenocarcinomas of the esophagogastric junction by metabolic imaging. J Clin Oncol 2001; 19(12):3058–3065.
Downey RJ, Akhurst T, Ilson D, Ginsberg R, Bains MS, Gonen M, Koong H, Gollub M, Minsky BD, Zakowski M, Turnbull A, Larson SM, Rusch V. Whole body 18FDG-PET and the response of esophageal cancer to induction therapy: results of a prospective trial. J Clin Oncol 2003; 21(3):428–432.
Willis J, Cooper GS, Isenberg G, Sivak MV, Levitan N, Clayman J, Chak A. Correlation of EUS measurement with pathologic assessment of neoadjuvant therapy response in esophageal carcinoma. Gastrointest Endosc 2002; 55(6):655–661.
van Hagen P, Hulshof MC, van Lanschot JJ, Steyerberg EW, van Berge Henegouwen MI, Wijnhoven BP, Richel DJ, Nieuwenhuijzen GA, Hospers GA, Bonenkamp JJ, Cuesta MA, Blaisse RJ, Busch OR, ten Kate FJ, Creemers GJ, Punt CJ, Plukker JT, Verheul HM, Spillenaar Bilgen EJ, van Dekken H, van der Sangen MJ, Rozema T, Biermann K, Beukema JC, Piet AH, van Rij CM, Reinders JG, Tilanus HW, van der Gaast A; CROSS Group. Preoperative chemoradiotherapy for esophageal or junctional cancer. N Engl J Med 2012; 366(22):2074–2084.
Al-Sarraf M, Martz K, Herskovic A, Leichman L, Brindle JS, Vaitkevicius VK, Cooper J, Byhardt R, Davis L, Emami B. Progress report of combined chemoradiotherapy versus radiotherapy alone in patients with esophageal cancer: an intergroup study. J Clin Oncol 1997; 15(1):277–284.
Cooper JS, Guo MD, Herskovic A, Macdonald JS, Martenson JA Jr, Al-Sarraf M, Byhardt R, Russell AH, Beitler JJ, Spencer S, Asbell SO, Graham MV, Leichman LL. Chemoradiotherapy of locally advanced esophageal cancer: long-term follow-up of a prospective randomized trial (RTOG 85–01). JAMA 1999; 281(17):1623–1627.
Herskovic A, Martz K, Al-Sarraf M, Leichman L, Brindle J, Vaitkevicius V, Cooper J, Byhardt R, Davis L, Emami B. Combined chemotherapy and radiotherapy compared with radiotherapy alone in patients with cancer of the esophagus. N Eng J Med 1992; 326:1593–1598.
Bedenne L, Michel P, Bouché O, Milan C, Mariette C, Conroy T, Pezet D, Roullet B, Seitz JF, Herr JP, Paillot B, Arveux P, Bonnetain F, Binquet C. Chemoradiation followed by surgery compared with chemoradiation alone in squamous cancer of the esophagus: FFCD 9102. J Clin Oncol 2007; 25(10):1160–1168.
Stahl M, Stuschke M, Lehmann N, Meyer HJ, Walz MK, Seeber S, Klump B, Budach W, Teichmann R, Schmitt M, Schmitt G, Franke C, Wilke H. Chemoradiation with and without surgery in patients with locally advanced squamous cell carcinoma of the esophagus. J Clin Oncol 2005; 23(10):2310–2317.
Patnana SV, Murthy SB, Xiao L, Rohren E, Hofstetter WL, Swisher SG, Liao Z, Lee JH, Bhutani MS, Macapinlac HA, Wang X, Ajani JA. Critical role of surgery in patients with gastroesophageal carcinoma with a poor prognosis after chemoradiation as defined by positron emission tomography. Cancer 2010; 116(19):4487–4494
Acknowledgments
The authors would like to thank the generous support of the Stuart & Flora Mason Family Foundation and the Edwards Family Esophageal Research Fund.
Conflicts of Interest
None.
Financial Disclosures
None.
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Fig. 6
Median overall survival of trimodality-eligible patients with locally advanced esophageal adenocarcinoma with poorly differentiated tumors treated with chemoradiation followed by surgery compared to definitive chemoradiation (n = 91) (JPEG 96 kb)
Fig. 7
Median overall survival of trimodality-eligible patients with locally advanced esophageal adenocarcinoma with post-treatment SUV ≥ 8.75 treated with chemoradiation followed by surgery compared to definitive chemoradiation (n = 12) (JPEG 77 kb)
Fig. 8
Median overall survival of trimodality-eligible patients with locally advanced esophageal adenocarcinoma with post-treatment SUV = 3.45 to <8.75 treated with chemoradiation followed by surgery compared to definitive chemoradiation (n = 82) (JPEG 92 kb)
Rights and permissions
About this article
Cite this article
Murphy, C.C., Correa, A.M., Ajani, J.A. et al. Surgery Is an Essential Component of Multimodality Therapy for Patients with Locally Advanced Esophageal Adenocarcinoma. J Gastrointest Surg 17, 1359–1369 (2013). https://doi.org/10.1007/s11605-013-2223-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-013-2223-4