Abstract
Postmortem computed tomography (CT) is an essential tool for investigating the causes of death. Postmortem CT has characteristic imaging findings and should not be interpreted in the same manner as clinical antemortem images. In analyzing the cause of death in in-hospital death cases using postmortem images, it is crucial to understand early postmortem and post-resuscitation changes. In addition, it is essential to understand the limitations of diagnosing the cause of death or significant pathology relating to death on non-contrast-enhanced postmortem CT. In Japan, there has also been social demand to establish a system for postmortem imaging at the time of death. To facilitate such a system, clinical radiologists should be prepared to interpret postmortem images and assess the cause of death. This review article provides comprehensive information regarding unenhanced postmortem CT for in-hospital death cases in daily clinical practice in Japan.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Postmortem imaging is the examination of corpses to investigate the cause of death or pathology related to death, and enables an observation of the inner body which cannot be obtained from the surface. In Japan, prior to the emergence of the concept of postmortem imaging, postmortem computed tomography (PMCT) of in-hospital deaths was reportedly performed in emergency rooms as there was a low rate of autopsy and a wide availability of CT scanners [1]. Eventually, in Japan, the concept of “Autopsy imaging” was proposed in 2000 [2]. Nowadays, it is locally abbreviated as “Ai”, and PMCT has become an essential tool for death investigation [3]. According to a 2021 report of the Japanese Ministry, 34.5% of unusual death cases (17,0174 out of 58,689 cases) underwent PMCT in 2018 [4].
The annual number of deaths in Japan began to exceed the number of births in 2005 [5]. If this trend continues, the number of deaths outside medical institutions, such as homes, is expected to increase [6]. Therefore, there is an increasing importance and need for cause-of-death investigations, which serves to improve public health and averts preventable deaths. In 2012, the law concerning the “Act to Accelerate Investigation into the Cause of Death and Identification” was enacted in Japan [7]. Further, in 2015, the “Medical Accident Investigation System” was established to prevent repeated medical accidents [8]. The Japanese system for investigating causes of death is being strengthened, and the use of imaging modalities at the time of death is expected to improve the accuracy of death-cause diagnosis. In this context, diagnostic guidelines for postmortem imaging in Japan were established and translated into English and Italian to standardize the interpretation of postmortem imaging [9].
This narrative review article describes essential knowledge required of clinical radiologists when interpreting postmortem non-contrast-enhanced CT of in-hospital death cases.
Practical operation of postmortem CT in in-hospital death cases
In most cases of postmortem imaging for in-hospital deaths in Japan, the time from death to postmortem CT scanning is several hours to 1 day, and the patient is kept in a supine position in a mortuary until postmortem CT scanning [10,11,12]. Postmortem CT of the deceased does not produce respiratory or motion artifacts. Dose reduction techniques are not necessary to reduce radiation exposure. Therefore, there is no problem with imaging under conditions that provide the best image quality, but an increased dose does not always improve image quality [13]. It is essential to include coverage from head to pelvis during the scanning, and the scanning range may be divided between the head and trunk. If the lower extremities are presumed to be less involved in the cause of death, they could be omitted [13]. During scanning, the standard position for the body is supine, with both upper extremities in the drooping position. Artifacts on the trunk caused by drooping of the upper extremities may degrade image quality [14]. In cases where medical malpractice is suspected, it is crucial to preserve the circumstances at the time of death by scanning CT images with medical devices, such as catheters and tubes, left in place [15].
CT findings of normal postmortem change
CT imaging can reveal the various changes that occur in the body after death, known as postmortem changes. These changes are classified into early (occurring within 24 h of death) and late postmortem changes [16]. Examples of early postmortem changes include pallor mortis (the earliest change which reflects the external skin color change), algor mortis (the progressive decrease in body temperature), livor mortis as known postmortem hypostasis (the non-circulating blood and other fluids settle with gravity toward the dependent parts of the body), and rigor mortis (stiffening of muscle). However, some changes, such as pallor mortis and algor mortis, cannot be visualized on CT scans [17], although low temperature induces density changes in tissue and associated CT value changes. Meanwhile, findings reflecting the cessation of circulation are observed after death. In cases of in-hospital deaths, early postmortem changes are typically observed because of the relatively short time between death and CT scanning. Decomposition (autolysis and gas production) is a late postmortem change, but it can occur during the early postmortem period. Understanding these changes and their mechanisms can aid in interpreting postmortem CT images and determining the cause of death (Table 1).
Livor mortis/postmortem hypostasis
Livor mortis, also known as postmortem hypostasis, is a postmortem change that occurs as a result of circulatory arrest, which leads to a deprivation of blood supply. On CT images, this can manifest as hyper-attenuation in the dorsal venous sinuses, such as the superior sagittal and transverse venous sinuses (Fig. 1a and 1b) [18], and in the lumens of the heart and aorta, especially in the thoracic aorta (sometimes also in the inferior vena cava). This is due to the concentration of the hemoglobin molecule in sedimented erythrocytes and can sometimes lead to the formation of a fluid–fluid level (Fig. 1c) [19]. In the lungs, increased attenuation can be seen on the gravity side, resembling ground glass opacity with horizontal plane formation (Fig. 1d) [20], and this finding becomes more conspicuous over time [21]. Additionally, a distinct blood deposition with a horizontal plane in the lumens of the heart and aorta can indicate a hyper-fibrinolytic system due to acute death. In contrast, a cast-like hyperattenuating area can be seen in the pulmonary artery and aorta during the long death phase and in the agonal stage of chronic disease (Fig. 2) [22]. Subcutaneous edema and skin thickening can also occur and are generally symmetrical [23].
Representative CT images of early regular postmortem changes. a, b increased density in the superior sagittal sinus (arrowheads), brain edema (dorsally dominant), and loss of the differentiation between the gray and white matter. c thickened left ventricular wall (white arrowheads), hyperattenuating in the gravity side (black arrowhead), and dilatation in the right heart cavities (arrows). d fluid in the bilateral main bronchus (arrows) and the increased attenuation, such as ground-glass opacities with the horizontal plane formation on the gravity-dependent side of the lungs (arrowheads). e narrowed short diameter of the aorta and the hyperattenuating and thickened aortic wall (arrow). f intrahepatic portal venous gas (8 h elapsed from death without cardiopulmonary resuscitation). g obscuring of outline and swelling in the pancreas (arrowheads)
Rigor mortis
Rigor mortis is a postmortem change that occurs as a result of muscle stiffness caused by chemical changes in the muscles after death. On CT images, this can manifest as an increase in the heart's ventricular wall thickness compared with antemortem images (Fig. 1c) [24], narrowing of short arterial diameters, and hyper-attenuation and thickening of the arterial wall, reflecting smooth muscle contraction. These findings are most prominent in large arteries, such as the aorta and pulmonary artery (Fig. 1e) [25,26,27]. Skeletal muscle, which is striated muscle, also exhibits increased attenuation on postmortem CT than on antemortem CT [28].
Changes secondary to the cessation of circulation
After circulatory arrest, the vasculature is at a mean circulatory filling pressure, which is hydrostatic pressure (approximately 7 mmHg). This pressure is slightly higher than the antemortem right heart system diastolic pressure, which leads to a shift of blood volume to the right heart system after death and can result in dilatation of the right heart system and superior vena cava (Fig. 1c) [29] and narrowing of the aorta (Fig. 1e) [30, 31].
Additionally, there are other characteristic postmortem CT findings of brain parenchyma associated with cessation of circulation, such as edematous changes that increase the water content of the gray matter and obscure the distinction between gray and white matter (Fig. 1a) [32], narrowing of sulci and ventricles, and brain swelling when vasogenic edema is prominent (Fig. 1a) [33].
Loss of air in the trachea can also occur as fluid exudates from the lung interstitium into the alveoli and airways (Fig. 1d). This is a common finding when pleural effusion, lung consolidation, and atelectasis are present. Membrane hyperpermeability is thought to be one of the mechanisms [34].
The CT value of cerebrospinal fluid is higher in postmortem images than in antemortem images, but the degree of change can vary depending on the cause of death, antemortem pathology, and environmental conditions. In addition, there is a positive correlation between the CT values of the cerebrospinal fluid and elapsed time after death [35, 36].
There are few reports of postmortem CT findings for pleural effusion and ascites; however, the rate in pleural space fluid increased at 30 h, and the volume continued to increase until 40 h after death [37]. In addition, an elevated level of ascites attenuation on postmortem CT within 24 h after death relative to antemortem CT (AMCT) was confirmed in individuals who underwent enhanced AMCT shortly before death [38].
Gas production
Gas production can occur in blood vessels and organs (Fig. 1f) because of the decomposition or presence of gas-producing bacteria in the vasculature and intestinal tract, even in cases where cardiopulmonary resuscitation was not performed [39]. (Gas production reflecting post-cardiopulmonary resuscitation changes is discussed later.) Gas is typically more prevalent 24 h after death but can also be seen in the early postmortem period, and its presence does not correlate with the time elapsed since death [40].
Autolysis
Autolysis is a phenomenon in which tissues are digested by their enzymes after death, resulting in obscuring of organ outlines and changes in volume. In the early postmortem period, autolysis of the pancreas, spleen, and adrenal gland can occur (Fig. 1g) [41]. In the stomach, a condition called gastromalacia can occur, which can also cause gastric perforation because of the gastric juice. This postmortem change may be suspected on CT images when abdominal free air is observed despite the absence of other postmortem changes, such as intravascular or intra-organic gas [42], and must be distinguished from antemortem gastric perforation. Additionally, after 24 h postmortem, the brain shows a decrease in gray matter density due to autolysis [41].
Cardiopulmonary resuscitation-related postmortem CT findings
Postmortem CT images may also show findings reflecting post-cardiopulmonary resuscitation changes performed before death. The effects of cardiopulmonary resuscitation before death are reflected in the postmortem CT findings (Table 1).
Artificial ventilation
Manual ventilation with a bag–valve–mask or similar device supplies air to the trachea, lungs, and gastrointestinal tract, resulting in extensive dilation of the gastrointestinal tract, primarily the stomach and small intestine (Fig. 3a). If the intestinal mucosa becomes ischemic owing to the cessation of circulation, gas from the lumen of the gastrointestinal tract can easily enter the intestinal wall (Fig. 3a). If the pressure on the ischemic intestinal wall is increased by the dilation and gas in the intestinal tract, intestinal gasses can pass through the ischemic wall, through the mesentery (Fig. 3a), and into the portal gas [43].
Representative CT images reflecting cardiopulmonary resuscitation. a extensive dilation of the gastrointestinal tract, gas in the intestinal wall and mesenteric vasculature. b the dilated medullary veins (arrows) near the lateral ventricles. c intraorganic and intravascular (broadly observed in the liver, abdominal aorta, vertebral body, and spinal canal) gas (scanned 10 h after death and also observed abdominal free air due to possible autolysis (unidentified bowel perforation by autopsy)). d gas in the arteries and veins in the brain (same case as Fig. 3c). e rib fractures in the anterior thoracic regions (arrowheads). f hemopericardium (arrows) due to cardiac rupture and sequel left hemothorax (black arrowheads) (also observed left rib fractures in the anterior thoracic regions (white arrowhead)). g right retroperitoneal hemorrhage (arrows) due to unknown cause confirmed by autopsy (same case as Fig. 3f)
Chest compression
Elevated white matter CT values cause the loss of gray–white matter differentiation in sudden death cases in which cardiopulmonary resuscitation was performed before death [44]. It is believed that the dilated medullary veins in the hypoxic brain under cardiac arrest become more congested by chest compressions, increasing white matter density (Fig. 3b) [45]. However, elevated white matter CT values can be seen even without cardiopulmonary resuscitation [46, 47].
Intravascular and intra-organic gas is frequently observed after chest compressions. The causes of this observation include the vaporization of dissolved gasses in the blood, air entry through the infusion route, and the formation of broncho-vascular fistulas (pressure trauma to the bronchi and pulmonary vessels) in combination with artificial ventilation [48, 49]. Chest compressions can spread intravascular gasses in the arteries in a prograde fashion and the veins in a retrograde fashion. The longer the duration of cardiopulmonary resuscitation, the more frequently gas is seen in multiple organs (Fig. 3c and d) [40].
Chest compression can easily cause rib fractures in the anterior and lateral thoracic regions (Fig. 3e) [50], and posterior rib fractures are usually considered inconsistent with chest compression. Chest compression can also complicate small pneumothorax, hemothorax, lung contusion, hemopericardium, and cardiac rupture (Fig. 3f) [51,52,53]. Intrapericardial and mediastinal hematomas may also occur. Owing to chest compression, sternal fractures can sometimes occur, and subcutaneous pre-sternal hematoma is sometimes observed following the use of a mechanical chest compression device [54]. Using mechanical chest compression devices can also result in unusual fractures of the mid-thoracic spine [55]. Abdominal hemorrhages, including peri-hepatic, peri-splenic, and retroperitoneal, are potential complications from chest compression attempts (Fig. 3g) [54, 56]. These findings can be explained when there is no other reason for antemortem pathology.
Interpretation of postmortem CT
When interpreting postmortem CT images, it is important to consider normal postmortem changes and post-cardiopulmonary resuscitation changes that have already been discussed. The use of a checklist can aid in this process [57]. To interpret the images accurately, it is important to gather information such as the date and time of death, the date and time of CT imaging, medical history, and clinical course, and whether or not resuscitation was performed (if so, the details of the resuscitation). If a sequential autopsy is planned after the CT, it can be helpful to inform the autopsy physician of any relevant findings, even if they are negative. In summary, while the cause of death can sometimes be determined from postmortem CT images, it is important to recognize that the pathologist plays a central role in determining the cause of death, and the CT should be used as an adjunct [58].
Diagnosable or non-diagnosable pathology on non-contrast-enhanced postmortem CT in in-hospital deaths
Head and neck
Gross intracranial hemorrhage and cerebral herniation, which are lethal intracranial lesions, can almost certainly be detected on postmortem CT images (Fig. 4a) [59, 60]. However, cessation of circulation reduces the amount of subdural hemorrhage in adults. This can be observed as a reduction in the volume of the subdural hematoma and midline deviation and an increase in hematoma CT value on postmortem CT images, compared with antemortem CT images [61, 62].
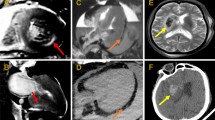
Examples of diagnosable fatal pathology on postmortem CT. a cerebral hemorrhage in the left parietal lobe with subfalcine herniation. b right hemothorax (arrow) due to ruptured thoracic aortic aneurysm after endovascular aortic repair (arrowheads). c ruptured abdominal aortic aneurysm (arrow) and massive retroperitoneal hemorrhage (arrowheads)
Additionally, significant diffuse cerebral swelling and obscuration of gray-white matter differentiation at a very early postmortem stage may indicate antemortem hypoxic-ischemic encephalopathy [63, 64]. These findings can sometimes be observed immediately after death and may be difficult to distinguish from normal postmortem changes. However, the brain in the very early postmortem period is in a state similar to hyperacute cerebral infarction. Cellular edema causes the swelling of individual brain cells and the narrowing of the spaces between cells, making it difficult for the overall brain volume to change significantly [65]. In the very early postmortem period, loss of gray–white matter differentiation because of cellular edema is rarely accompanied by severe brain swelling [32]. Changes in basal ganglia density may help distinguish these from postmortem changes [66]. In addition, a gross neck mass that narrows the airway can be determined by postmortem CT [67].
When performing diagnostic imaging on neonates and fetuses at the time of death, it is important to be aware that their skulls, and other organs and structures, such as the abdominal organs and eyeballs, are susceptible to severe deformation because of their lack of fixation. This can occur as a result of being placed on a CT cradle or suturing, leading to inaccuracies in the imaging results. Therefore, care should be taken to minimize mechanical deformation and ensure accurate imaging in these cases. [68, 69].
Chest
Evaluating diffusely elevated pulmonary concentrations on postmortem CT can be challenging because of the high occurrence of postmortem pulmonary edema and hypostasis, which can obscure antemortem pathologies such as inflammation or tumors [12]. In children with established respiration, the air in the lungs may not be visible on postmortem CT images, making it difficult to diagnose conditions such as pneumonia before death [70]. However, certain conditions, such as ruptured thoracic aortic aneurysms, thoracic aortic dissection, pleural effusion, pneumothorax, and pneumo-mediastinum, can be diagnosed with high sensitivity on postmortem CT (Fig. 4b) [71]. Cardiac tamponade can also be diagnosed [72, 73]. Iatrogenic hematomas in the pericardium may occur owing to chest compression and must be distinguished from antemortem conditions [74]. Pulmonary arterial thrombo-embolization can be difficult to evaluate [33], but fat emboli in the pulmonary artery can be identified [74].
Abdomen and pelvis
The detection of ruptured abdominal aortic aneurysms, intra-abdominal hemorrhage, gross abdominal masses, and gastrointestinal perforations, can be facilitated through postmortem CT imaging (Fig. 4c). However, the evaluation of gastrointestinal contents and walls can be challenging [75]. Postmortem CT images can be difficult to determine gastrointestinal hemorrhagic changes.
Bones and soft tissues
Postmortem CT imaging can be used to elucidate bony lesions, such as the presence or absence of fractures and osteoplastic or osteolytic changes (Fig. 5) [23]. Additionally, postmortem CT imaging can reveal subcutaneous edema, large peripheral lacerations, and soft tissue hematomas [76]. These findings can aid in determining the cause of death and identifying any potential injuries and pathologies that occurred before death.
Benefits of postmortem CT for pathologic autopsy in in-hospital death
Postmortem CT can provide detailed images of the body in its current state, allowing for a comprehensive evaluation of the cause of death [77]. The advantages of postmortem CT include: 1. The ability to determine the need for an autopsy by identifying key findings before autopsy. 2. The ability to plan autopsies by capturing images of the patient's condition before autopsy, reducing the risk of infection and exposure to hazardous materials for pathologists. 3. The ability to identify findings that may not be easily detected through traditional dissection methods, such as bony lesions. In some cases, postmortem CT may be sufficient to omit skeletal dissection during the subsequent autopsy [23].
Furthermore, postmortem CT allows for the preservation of the “as is” state of the body, which cannot be achieved once an autopsy has been performed. Additionally, postmortem CT images can be stored semi-permanently and reviewed again, providing superior objectivity. Furthermore, the reproducibility of the images can be helpful in discussions during investigations of medical accidents. Various devices and tubes should not be removed. They should remain in place for postmortem imaging, especially when medical malpractice is suspected or following resuscitation attempts [78].
Diagnostic accuracy of non-contrast-enhanced postmortem CT to estimate the cause of death in cases of in-hospital deaths
Postmortem CT can aid in diagnosing the cause of death and major diagnoses related to death, surpassing clinical diagnosis alone in cases of in-hospital deaths (Table 2) [79,80,81,82,83,84,85]. The correlation rate of the immediate cause of death determined by postmortem CT and hospital autopsy is approximately 60‒70% [79, 82]. Similarly, the sensitivity for the cause of death is approximately 60‒70% with postmortem CT [79, 80, 84]. When combined with clinical information, postmortem CT can accurately diagnose respiratory failure [82]. On the contrary, in a large prospective cohort study validating postmortem images in diagnosing medicolegal deaths in adults, postmortem CT was more accurate than magnetic resonance imaging (MRI) in providing the cause of death, and postmortem CT could be acceptable for medicolegal purposes. However, the most common errors in identifying the cause of death by CT or MRI images based on conventional autopsy were ischemic heart disease, pulmonary embolism, pneumonia, and intra-abdominal lesions. Some common causes of sudden death are often omitted on postmortem CT and MRI and these imaging techniques cannot replace conventional autopsy [86].
When focusing specifically on in-hospital deaths in children, the detection rate of the main pathology related to the immediate cause of death using unenhanced postmortem CT in combination with clinical evaluation is approximately 30% [83, 85]. In cases of non-traumatic in-hospital deaths in children aged < 3 years, postmortem CT had a higher diagnostic sensitivity for respiratory system-related causes of death compared with clinical evaluation alone. However, regarding the causes of death related to cardiac and multi-organ systems, the diagnostic sensitivity of postmortem CT was significantly lower than that of clinical evaluation [85]. In children, an autopsy alone is not the best method for estimating the cause of death, and it is considered that autopsies should be used in combination with postmortem images. Postmortem CT is indispensable, especially for determining death due to trauma. On the other hand, in traumatic death cases of children aged 0 to 12 years, an autopsy alone is not the most reliable method for estimating the cause of death, and it is considered that autopsies should be used in combination with postmortem CT. Particularly, postmortem CT is superior to autopsy for detecting bone lesions in children [87].
Conclusions
Postmortem CT plays a crucial role in postmortem investigations, particularly in cases of in-hospital death. It allows the accurate diagnosis of the cause of death and major diagnoses for specific cases, surpassing clinical diagnosis alone. Therefore, clinical radiologists are expected to have a role in the interpretation of postmortem CT images and the determination of the cause of death. This presents an opportunity and need for radiologists to expand their knowledge and experience in postmortem imaging and to contribute toward the understanding of death causes.
References
Ohashi N. Detection of the cause of death in DOA patients: utility and problems of postmortem computed tomography. KANTO J Jpn Assoc Acute Med. 1989;10:24–5.
Ezawa H, Yoneyama R, Kandatsu S, Yoshikawa K, Tsujii H, Harigaya K. Introduction of autopsy imaging redefines the concept of autopsy: 37 cases of clinical experience. Pathol Int. 2003;53:865–73.
Shiotani S, Hamabe Y, Ohashi N, Ezawa H. The ground swell of postmortem computed tomography in Japan: the harbinger of widespread use of autopsy imaging? Accessed Dec 26, 2022 from https://www.bmj.com/rapid-response/2011/10/31/ground-swell-postmortem-computed-tomography-japan-harbinger-widespread-use
Administrative Evaluation Bureau, Ministry of Internal Affairs and Communications. Policy evaluation regarding promotion of death-cause investigation. Accessed Mar 25, 2023 from https://www.soumu.go.jp/main_content/000737589.pdf
Ministry of Health, Labour, and Welfare of Japan. Vital statistics in Japan. Accessed Dec 26, 2022 from https://www.mhlw.go.jp/toukei/list/81-1a.html
Cabinet Office, Government of Japan. Annual Report on the Aging Society. Accessed Dec 26, 2022 from https://www8.cao.go.jp/kourei/whitepaper/index-w.html
The House of Representatives, Japan. Accessed Dec 26, 2022 from https://www.shugiin.go.jp/internet/itdb_housei.nsf/html/housei/18020120622033.htm
Ministry of Health, Labour, and Welfare of Japan. Vital statistics in Japan. Accessed Dec 26, 2022 from https://www.mhlw.go.jp/stf/seisakunitsuite/bunya/0000061201.html
Japan Radiological Society and Center for Cause of Death Investigation, Faculty of Medicine, Hokkaido University ed. Post-Mortem Imaging Interpretation Guidelines 2020. Kanehara & Co., Ltd
Gullo RL, Mishra S, Lira DA, Padole A, Otrakji A, Khawaja RD, et al. Quantification of interstitial fluid on whole body CT: comparison with whole body autopsy. Forensic Sci Med Pathol. 2015;11:488–96.
Takahashi N, Yajima K, Otaki M, Yoshikawa Y, Ishihara A, Sato Y, et al. Postmortem volume change of the spleen and kidney on early postmortem computed tomography: comparison with antemortem computed tomography. Jpn J Radiol. 2019;37:534–42.
Gonoi W, Watanabe Y, Shirota G, Abe H, Okuma H, Shintani-Domoto Y, et al. Pulmonary postmortem computed tomography of bacterial pneumonia and pulmonary edema in patients following non-traumatic in-hospital death. Leg Med (Tokyo). 2020;45: 101716.
Shenton A, Kralt P, Suvarna SK. Practical considerations of post mortem computed tomography and report writing. In: Shenton A, Kralt P, Suvarna SK, editors. Post mortem CT for non-suspicious adult death. 1st ed. Cham, Switzerland: Springer Cham; 2021. p. 19–23.
Ishida M, Gonoi W, Shirota G, Okuma H, Yamazawa S, Hinata M, et al. Diffusely decreased liver attenuation on post-mortem computed tomography: Comparison with ante-mortem computed tomography and autopsy findings. J Forensic Radiol Imaging. 2019;16:1–4.
Japan Medical Safety Research Organization. Accessed May 26, 2022 from https://www.medsafe.or.jp/modules/public/index.php?content_id=7
Zerbo S, Scopelliti L, Vernuccio F, Re GL, Argo A, Kharoshah M. Radiology for postmortem. In: Radiology in forensic medicine. Cham: Springer. 2020:265–72.
Shenton A, Kralt P, Suvarna SK. Death, Post mortem changes and decomposition on post mortem computed tomography. In: Shenton A, Kralt P, Suvarna SK, editors. Post mortem CT for non-suspicious adult death. 1st ed. Cham, Switzerland: Springer Cham; 2021. p. 38–42.
Takahashi N, Satou C, Higuchi T, Shiotani M, Maeda H, Hirose Y. Quantitative analysis of intracranial hypostasis: comparison of early postmortem and antemortem CT findings. AJR Am J Roentgenol. 2010;195:W388–93.
Shiotani S, Kohno M, Ohashi N, Yamazaki K, Itai Y. Postmortem intravascular high-density fluid level (hypostasis): CT findings. J Comput Assist Tomogr. 2002;26:892–3.
Shiotani S, Kohno M, Ohashi N, Yamazaki K, Nakayama H, Watanabe K, et al. Non-traumatic postmortem computed tomographic (PMCT) findings of the lung. Forensic Sci Int. 2004;139:39–48.
Shiotani S, Kobayashi T, Hayakawa H, Kikuchi K, Kohno M. Postmortem pulmonary edema: a comparison between immediate and delayed postmortem computed tomography. Leg Med (Tokyo). 2011;13:151–5.
Shenton A, Kralt P, Suvarna SK. Death, Post mortem changes and decomposition on post mortem computed tomography. In: Shenton A, Kralt P, Suvarna SK, editors. Post mortem CT for non-suspicious adult death. 1st ed. Cham, Switzerland: Springer Cham; 2021. p. 56–7.
Shenton A, Kralt P, Suvarna SK. Post mortem computed tomography of the bones and soft tissues. In: Shenton A, Kralt P, Suvarna SK, editors. Post mortem CT for non-suspicious adult death. 1st ed. Cham, Switzerland: Springer Cham; 2021. p. 274–5.
Okuma H, Gonoi W, Ishida M, Shintani Y, Takazawa Y, Fukayama M, et al. Heart Wall Is Thicker on Postmortem Computed Tomography Than on Antemortem Computed Tomography: The First Longitudinal Study. PLoS ONE. 2013;8: e76026.
Shiotani S, Kohno M, Ohashi N, Yamazaki K, Nakayama H, Ito Y, et al. Hyperattenuating aortic wall on postmortem computed tomography (PMCT). Radiat Med. 2002;20:201–6.
Okuma H, Gonoi W, Ishida M, Shintani Y, Takazawa Y, Fukayama M, et al. Greater thickness of the aortic wall on postmortem computed tomography compared with antemortem computed tomography: the first longitudinal study. Int J Legal Med. 2014;128:987–93.
Gonoi W, Watanabe Y, Nyunoya K, Shirota G, Okimoto N, Fujimoto K, et al. Early postmortem changes in the pulmonary artery and veins: An antemortem and postmortem computed tomography study. Forensic Imaging. 2022;28: 200497.
Okuma H, Gonoi W, Ishida M, Shirota G, Shintani Y, Abe H, et al. Comparison of attenuation of striated muscle between postmortem and antemortem computed tomography: results of a longitudinal study. PLoS ONE. 2014;9: e111457.
Shiotani S, Kohno M, Ohashi N, Yamazaki K, Nakayama H, Watanabe K, et al. Dilatation of the heart on postmortem computed tomography (PMCT): comparison with live CT. Radiat Med. 2003;21:29–35.
Takahashi N, Higuchi T, Hirose Y, Yamanouchi H, Takatsuka H, Funayama K. Changes in aortic shape and diameters after death: comparison of early postmortem computed tomography with antemortem computed tomography. Forensic Sci Int. 2013;225:27–31.
Hyodoh H, Sato T, Onodera M, Washio H, Hasegawa T, Hatakenaka M. Vascular measurement changes observed using postmortem computed tomography. Jpn J Radiol. 2012;30:840–5.
Takahashi N, Satou C, Higuchi T, Shiotani M, Maeda H, Hirose Y, et al. Quantitative analysis of brain edema and swelling on early postmortem computed tomography: comparison with antemortem computed tomography. Jpn J Radiol. 2010;28:349–54.
Ishida M, Gonoi W, Okuma H, Shirota G, Shintani Y, Abe H, et al. Common postmortem computed tomography findings following atraumatic death: Differentiation between normal postmortem changes and pathologic lesions. Korean J Radiol. 2015;16:798–809.
Ishida M, Gonoi W, Hagiwara K, Okuma H, Shintani Y, Abe H, et al. Fluid in the airway of nontraumatic death on postmortem computed tomography: relationship with pleural effusion and postmortem elapsed time. Am J Forensic Med Pathol. 2014;35:113–7.
Hasegawa I, Shimizu A, Saito A, Suzuki H, Vogel H, Püschel K, et al. Evaluation of post-mortem lateral cerebral ventricle changes using sequential scans during post-mortem computed tomography. Int J Legal Med. 2016;130:1323–8.
Fujimoto K, Gonoi W, Ishida M, Okimoto N, Nyunoya K, Abe H, et al. Association between postmortem computed tomography value of cerebrospinal fluid and time after death: A longitudinal study of antemortem and postmortem computed tomography. J Forensic Leg Med. 2023;93: 102461.
Hyodoh H, Shimizu J, Watanabe S, Okazaki S, Mizuo K, Inoue H. Time-related course of pleural space fluid collection and pulmonary aeration on postmortem computed tomography (PMCT). Leg Med (Tokyo). 2015;17:221–5.
Ishida M, Gonoi W, Abe H, Shirota G, Fujimoto K, Okimoto N, et al. Longitudinal comparison of ascites attenuation between antemortem and postmortem computed tomography. Forensic Sci Int. 2021;321: 110727.
Shenton A, Kralt P, Suvarna SK. Death, Post mortem changes and decomposition on post mortem computed tomography. In: Shenton A, Kralt P, Suvarna SK, editors. Post mortem CT for non-suspicious adult death. 1st ed. Cham, Switzerland: Springer Cham; 2021. p. 50–6.
Ishida M, Gonoi W, Hagiwara K, Takazawa Y, Akahane M, Fukayama M, et al. Intravascular gas distribution in the upper abdomen of non-traumatic in-hospital death cases on postmortem computed tomography. Leg Med (Tokyo). 2011;13:174–9.
Levy AD, Harcke HT, Mallak CT. Postmortem imaging: MDCT features of postmortem change and decomposition. Am J Forensic Med Pathol. 2010;31:12–7.
O’Donnell CJ, Baker MA. Postmortem CT findings of gastromalacia: a trap for the radiologist with forensic interest. Forensic Sci Med Pathol. 2010;6:293–7.
Shiotani S, Kohno M, Ohashi N, Yamazaki K, Nakayama H, Watanabe K. Postmortem computed tomographic (PMCT) demonstration of the relation between gastrointestinal (GI) distension and hepatic portal venous gas (HPVG). Radiat Med. 2004;22:25–9.
Torbey MT, Selim M, Knorr J, Bigelow C, Recht L, et al. Quantitative analysis of the loss of distinction between gray and white matter in comatose patients after cardiac arrest. Stroke. 2000;31:2163–7.
Lida K, Satoh H, Arita K, Nakahara T, Kurisu K, Ohtani M, et al. Delayed hyperemia causing intracranial hypertension after cardiopulmonary resuscitation. Crit Care Med. 1997;25:971–6.
Nishiyama Y, Kanayama H, Mori H, Tada K, Yamamoto Y, Katsube T, et al. Whole brain analysis of postmortem density changes of grey and white matter on computed tomography by statistical parametric mapping. Eur Radiol. 2017;27:2317–25.
Shirota G, Gonoi W, Ishida M, Okuma H, Shintani Y, Abe H, et al. Brain swelling and loss of gray and white matter differentiation in human postmortem cases by computed tomography. PLoS ONE. 2015;10: e0143848.
Shiotani S, Kohno M, Ohashi N, Atake S, Yamazaki K, Nakayama H. Cardiovascular gas on non-traumatic postmortem computed tomography (PMCT): the influence of cardiopulmonary resuscitation. Radiat Med. 2005;23:225–9.
Yamaki T, Ando S, Ohta K, Kubota T, Kawasaki K, Hirama M. CT demonstration of massive cerebral air embolism from pulmonary barotrauma due to cardiopulmonary resuscitation. J Comput Assist Tomogr. 1989;13:313–5.
Yang KM, Lynch M, O’Donnell C. “Buckle” rib fracture: an artifact following cardio-pulmonary resuscitation detected on postmortem CT. Leg Med (Tokyo). 2011;13:233–9.
Miller AC, Rosati SF, Suffredini AF, Schrump DS, et al. A systematic review and pooled analysis of CPR-associated cardiovascular and thoracic injuries. Resuscitation. 2014;85:724–31.
Ondruschka B, Baier C, Bayer R, Hammer N, Dreßler J, Bernhard M, et al. Chest compression-associated injuries in cardiac arrest patients treated with manual chest compressions versus automated chest compression devices (LUCAS II) - a forensic autopsy-based comparison. Forensic Sci Med Pathol. 2018;14:515–25.
Kashiwagi Y, Sasakawa T, Tampo A, Kawata D, Nishiura T, Kokita N, et al. Computed tomography findings of complications resulting from cardiopulmonary resuscitation. Resuscitation. 2015;88:86–91.
Baumeister R, Held U, Thali MJ, Flach PM, Ross S. Forensic imaging findings by post-mortem computed tomography after manual versus mechanical chest compression. J Forensic Radiol Imaging. 2015;3:167–73.
Bohara M, Ohara Y, Mizuno J, Matsuoka H, Hattori N, Arita K. Cardiopulmonary Resuscitation-induced Thoracic Vertebral Fracture: A Case Report. NMC Case Rep J. 2015;3:106–8.
Pinto DC, Haden-Pinneri K, Love JC, et al. Manual and automated cardiopulmonary resuscitation (CPR): a comparison of associated injury patterns. J Forensic Sci. 2013;58:904–9.
Takahashi N, Higuchi T, Shiotani M, Satou S, Hirose Y. Effectiveness of a worksheet for diagnosing postmortem computed tomography in emergency departments. Jpn J Radiol. 2011;29:701–6.
Shenton A, Kralt P, Suvarna SK, et al. Practical considerations of post mortem computed tomography and report writing. In: Shenton A, Kralt P, Suvarna SK, editors., et al., Post mortem CT for non-suspicious adult death. 1st ed. Cham, Switzerland: Springer Cham; 2021. p. 26–31.
Rutty GN, Morgan B, Robinson C, Raj V, Pakkal M, Amoroso J, et al. Diagnostic accuracy of post-mortem CT with targeted coronary angiography versus autopsy for coroner-requested post-mortem investigations: a prospective, masked, comparison study. Lancet. 2017;390:145–54.
Tappero C, Thali MJ, Schweitzer W. The possibility of identifying brain hemorrhage in putrefied bodies with PMCT. Forensic Sci Med Pathol. 2020;16:571–6.
Inokuchi G, Yajima D, Hayakawa M, Motomura A, Chiba F, Makino Y, et al. Is acute subdural hematoma reduced during the agonal stage and postmortem? Int J Legal Med. 2013;127:263–6.
Berger N, Ebert LC, Ampanozi G, Flach PM, Gascho D, Thali MJ, et al. Smaller but denser: Postmortem changes alter the CT characteristics of subdural hematomas. Forensic Sci Med Pathol. 2015;11:40–6.
Fujioka M, Okuchi K, Sakaki T, Hiramatsu K, Miyamoto S, Iwasaki S, et al. Specific changes in human brain following reperfusion after cardiac arrest. Stroke. 1994;25:2091–5.
Dedouit F, Sévely A, Costagliola R, Otal P, Loubes-Lacroix F, Manelfe C, et al. Reversal sign on ante- and postmortem brain imaging in a newborn: report of one case. Forensic Sci Int. 2008;182:e11–4.
Liang D, Bhatta S, Gerzanich V, Simard JM, et al. Cytotoxic edema: mechanisms of pathological cell swelling. Neurosurg Focus. 2007;22:E2.
Shirota G, Ishida M, Shintani Y, Abe H, Ikemura M, Fukayama M, et al. Can postmortem computed tomography detect antemortem hypoxic-ischemic encephalopathy? Forensic Sci Med Pathol. 2016;12:267–75.
Shenton A, Kralt P, Suvarna SK, et al. Post mortem computed tomography of the extra-cranial head and neck. In: Shenton A, Kralt P, Suvarna SK, editors., et al., Post mortem CT for non-suspicious adult death. 1st ed. Cham, Switzerland: Springer Cham; 2021. p. 158.
Klein WM, Bosboom DGH, Koopmanschap DHJLM, Nievelstein RAJ, Nikkels PGJ, van Rijn RR, et al. Normal pediatric postmortem CT appearances. Pediatr Radiol. 2015;45:517–26.
Arthurs OJ, Barber JL, Taylor AM, Sebire NJ. Normal perinatal and paediatric postmortem magnetic resonance imaging appearances. Pediatr Radiol. 2015;45:527–35.
Kawasumi Y, Usui A, Hosokai Y, Igari Y, Hosoya T, Hayashizaki Y, et al. Post-mortem computed tomography findings of the lungs: Retrospective review and comparison with autopsy results of 30 infant cases. Eur J Radiol. 2015;84:721–5.
Shenton A, Kralt P, Suvarna SK, et al. Post mortem computed tomography of the chest. In: Shenton A, Kralt P, Suvarna SK, editors., et al., Post mortem CT for non-suspicious adult death. 1st ed. Cham, Switzerland: Springer Cham; 2021. p. 175–8.
Mychajlowycz M. The armored heart: Differentiating the etiology of hemopericardium on postmortem computed tomography. J Forensic Radiol Imaging. 2017;9:6–7.
Yamaguchi R, Makino Y, Chiba F, Torimitsu S, Yajima D, Shinozaki T, et al. Fluid-Fluid Level and Pericardial Hyperdense Ring Appearance Findings on Unenhanced Postmortem CT Can Differentiate Between Postmortem and Antemortem Pericardial Hemorrhage. AJR Am J Roentgenol. 2015;205:W568–77.
Makino Y, Kojima M, Yoshida M, Motomura A, Inokuchi G, Chiba F, et al. Postmortem CT and MRI findings of massive fat embolism. Int J Legal Med. 2020;134:669–78.
Shenton A, Kralt P, Suvarna SK, et al. Post mortem computed tomography of the abdomen and pelvis. In: Shenton A, Kralt P, Suvarna SK, editors., et al., Post mortem CT for non-suspicious adult death. 1st ed. Cham, Switzerland: Springer Cham; 2021. p. 252–331.
Jalalzadeh H, Giannakopoulos GF, Berger FH, Fronczek J, van de Goot FRW, Reijnders UJ, et al. Post-mortem imaging compared with autopsy in trauma victims–A systematic review. Forensic Sci Int. 2015;257:29–48.
Shenton A, Kralt P, Suvarna SK, et al. Introduction to the investigation of death and post mortem computed tomography. In: Shenton A, Kralt P, Suvarna SK, editors., et al., Post mortem CT for non-suspicious adult death. 1st ed. Cham, Switzerland: Springer Cham; 2021. p. 1–13.
Shenton A, Kralt P, Suvarna SK, et al. External findings, tubes and devices on post mortem computed tomography. In: Shenton A, Kralt P, Suvarna SK, editors., et al., Post mortem CT for non-suspicious adult death. 1st ed. Cham, Switzerland: Springer Cham; 2021. p. 72–5.
Takahashi N, Higuchi T, Shiotani M, Hirose Y, Shibuya H, Yamanouchi H, et al. The effectiveness of postmortem multidetector computed tomography in the detection of fatal findings related to cause of non-traumatic death in the emergency department. Eur Radiol. 2012;22:152–60.
Westphal SE, Apitzsch J, Penzkofer T, Mahnken AH, Knuchel R. Virtual CT autopsy in clinical pathology: feasibility in clinical autopsies. Virchows Arch. 2012;461:211–9.
Wichmann D, Obbelode F, Vogel H, Hoepker WW, Nierhaus A, Braune S, et al. Virtual autopsy as an alternative to traditional medical autopsy in the intensive care unit: a prospective cohort study. Ann Intern Med. 2012;156:123–30.
Inai K, Noriki S, Kinoshita K, Sakai T, Kimura H, Nishijima A, et al. Postmortem CT is more accurate than clinical diagnosis for identifying the immediate cause of death in hospitalized patients: a prospective autopsy-based study. Virchows Arch. 2016;469:101–9.
Arthurs OJ, Guy A, Thayyil S, Wade A, Jones R, Norman W, et al. Comparison of diagnostic performance for perinatal and paediatric post-mortem imaging: CT versus MRI. Eur Radiol. 2016;26:2327–36.
Sonnemans LJP, Kubat B, Prokop M, Klein WM, et al. Can virtual autopsy with postmortem CT improve clinical diagnosis of cause of death? A retrospective observational cohort study in a Dutch tertiary referral centre. BMJ Open. 2018;8: e018834.
Ishida M, Gonoi W, Shirota G, Abe H, Shintani-Domoto Y, Ikemura M, et al. Utility of unenhanced postmortem computed tomography for investigation of in-hospital nontraumatic death in children up to 3 years of age at a single Japanese tertiary care hospital. Medicine(Baltimore). 2020;99:e20130.
Roberts ISD, Benamore RE, Benbow EW, Lee SH, Harris JN, Jackson A, et al. Post-mortem imaging as an alternative to autopsy in the diagnosis of adult deaths: a validation study. Lancet. 2012;379:136–42.
Krentz BV, Alamo L, Grimm J, Dédouit F, Bruguier C, Chevallier C, et al. Performance of post-mortem CT compared to autopsy in children. Int J Legal Med. 2016;130:1089–99.
Acknowledgements
Part of this article was initially presented at the 79th Annual Meeting of the Japan Radiological Society in Yokohama in 2020. This work was supported by JSPS KAKENHI Grant Numbers JP19K19482, JP20K07989 and Daiwa Securities Health Foundation Grant 2021-2022.
Funding
Open access funding provided by The University of Tokyo. JSPS KAKENHI Grant Numbers JP19K19482, JP20K07989 and Daiwa Securities Health Foundation Grant 2021–2022.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical statement
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Ishida, M., Gonoi, W., Abe, H. et al. Essence of postmortem computed tomography for in-hospital deaths: what clinical radiologists should know. Jpn J Radiol 41, 1039–1050 (2023). https://doi.org/10.1007/s11604-023-01443-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11604-023-01443-w