Abstract
Objective
The neuroprotective function of heat shock protein A5 (HSPA5) in ischemic stroke has been confirmed. This study aimed to investigate the effects of early aerobic exercise on neurological function recovery from cerebral ischemia/reperfusion and to determine whether these effects are associated with the expression level of HSPA5 in the ischemic penumbra.
Methods
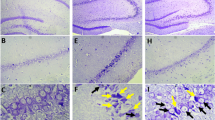
A total of 72 male Sprague-Dawley rats were randomly assigned to the ischemia and exercise group [middle cerebral artery occlusion (MCAO)-Ex, n=18], ischemia and sedentary group (MCAO-St, n=18), sham-surgery and exercise group (Sham-Ex, n=18), or sham-surgery and sedentary group (Sham-St, n=18). The MCAO-Ex and MCAO-St groups were subjected to MCAO for 60 min, whereas the Sham-Ex and Sham-St groups were subjected to an identical operation without MCAO. Rats in the MCAO-Ex and Sham-Ex groups then ran on a treadmill for 30 min once a day for 5 consecutive days. After reperfusion, the motor function of the rats was scored by the Bederson neurological function test, balance beam test, and screen test. Nissl staining was conducted to assess morphological and structural change of nerve cells in the ischemic penumbra. The reverse transcription-quantitative polymerase chain reaction was applied to detect the mRNA expression of HSPA5. Western blot analysis was conducted to determine the protein expression of HSPA5. Terminal deoxynucleotidyl transferase-mediated dUTP nick-end labeling (TUNEL) staining was carried out in the ischemic penumbra after MCAO.
Results
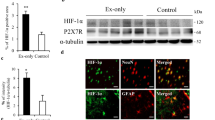
Rats receiving early treadmill exercise had lower Bederson neurological function, balance beam, and screen test scores on the 3rd, 7th, and 14th days after MCAO; in addition, more neurons survived in the ischemic penumbra after MCAO, and higher mRNA and protein expression of HSPA5 and fewer TUNEL-positive stained cells were observed.
Conclusion
Our study demonstrated that early aerobic exercise can improve neurological function recovery after ischemia/reperfusion. Furthermore, the increased level of HSPA5 in the ischemic penumbra might be one of the mechanisms of enhanced neurological function recovery.
Similar content being viewed by others
References
Xie W, Zhao ZH, Yang QM, et al. The efficacy of the seamless transfer of care model to apply for patients with cerebral apoplexy in china. Int J Nurs Sci, 2015,2(1):52–57
Gao L, Meschia JF, Judd SE, et al. What stroke symptoms tell us: association of risk factors and individual stroke symptoms in the Reasons for Geographic and Racial Differences in Stroke (REGARDS) study. J Stroke Cerebrovasc Dis, 2012,21(5):411–416
Burnett MG, Shimazu T, Szabados T, et al. Electrical forepaw stimulation during reversible forebrain ischemia decreases infarct volume. Stroke, 2006,37(5):1327–1331
Munari D, Pedrinolla A, Smania N, et al. High-intensity treadmill training improves gait ability, VO2peak and cost of walking in stroke survivors: preliminary results of a pilot randomized controlled trial. Eur J Phys Rehabil Med, 2018,54(3):408–418
Ke Z, Yip SP, Li L, et al. The effects of voluntary, involuntary, and forced exercises on brain-derived neurotrophic factor and motor function recovery: a rat brain ischemia model. PLoS One, 2011,6(2):e16643
Yoo AJ, Verduzco LA, Schaefer PW, et al. MRI-based selection for intra-arterial stroke therapy: the value of pretreatment diffusion-weighted imaging lesion volume in selecting patients with acute stroke who will benefit from early recanalization. Stroke, 2009,40(6):2046–2054
Broughton BR, Reutens DC, Sobey CG. Apoptotic mechanisms after cerebral ischemia. Stroke, 2009,40(5):e331–e338
Hendershot LM, Valentine VA, Lee AS, et al. Localization of the gene encoding human BiP/GRP78, the endoplasmic reticulum cognate of the HSP70 family, to chromosome 9q34. Genomics, 1994,20(2):281–284
Brewer JW, Hendershot LM. Building an antibody factory: a job for the unfolded protein response. Nat Immunol, 2005,6(1):23–29
Choi AY, Choi JH, Yoon H, et al. Luteolin induces apoptosis through endoplasmic reticulum stress and mitochondrial dysfunction in Neuro-2A mouse neuroblastoma cells. Eur J Pharmacol, 2011,668(1–2):115–126
Sasaki M, Honmou O, Kocsis JD. A rat middle cerebral artery occlusion model and intravenous cellular delivery. Methods Mol Biol, 2009,549:187–195
Peng ZF, Li JF, Li Y, et al. Downregulation of miR-181b in mouse brain following ischemic stroke induces neuroprotection against ischemic injury through targeting heat shock protein A5 and ubiquitin carboxyl-terminal hydrolase isozyme L1. J Neurosci Res, 2013, 91(10):1349–1362
Ding Y, Li J, Lai Q, et al. Motor balance and coordination training enhance the functional outcome in rats with transient middle cerebral artery occlusion. Neuroscience, 2004,123(3):667–674
Mátéffyová A, Otáhal J, Tsenov G, et al. Intrahippocampal injection of endothelin-1 in immature rats results in neuronal death, development of epilepsy and behavioral abnormalities later in life. Eur J Neurosci, 2006,24(2):351–360
Li J, Qu Y, Zu P, et al. Increased isoform-specific membrane translocation of conventional and novel protein kinase C in human neuroblastoma SH-SY5Y cells following prolonged hypoxia. Brain Res, 2006,1093(1):25–32
Munoz A, Nakazaki M, Goodman JC, et al. Ischemic preconditioning in the hippocampus of a knockout mouse lacking SUR1-based K(ATP) channels. Stroke, 2003,34(1):164–170
Coleman ER, Moudgal R, Lang K, et al. Early rehabilitation after stroke: a narrative review. Curr Atheroscler Rep, 2017,19(12):59
Yang L, Zhang J, Deng Y, et al. The effects of early exercise on motor, sense, and memory recovery in rats with stroke. Am J Phys Med Rehabil, 2017,96(3):e36–e43
Ye Z, Wang N, Xia P, et al. Parecoxib suppresses CHOP and Foxo1 nuclear translocation but increases GRP78 levels in a rat model of focal ischemia. Neurochem Res, 2013,38:686–693
Ouyang YB, Xu LJ, Emery JF, et al. Overexpressing GRP78 influences Ca2+ handling and function of mitochondria in astrocytes after ischemia -like stress. Mitochondrion, 2011,11(2):279–286
Nakka VP, Gusain A, Raghubir R. Endoplasmic reticulum stress plays a critical role in brain damage after cerebral ischemia/reperfusion in rats. Neurotox Res, 2010,17(2):189–202
Morimoto N, Oida Y, Shimazawa M, et al. Involvement of endoplasmic reticulum stress after middle cerebral artery occlusion in mice. Neuroscience, 2007,147(4): 957–996
Wu CX, Liu R, Gao M, et al. Pinocembrin protects the brain against ischemia/reperfusion injury by attenuating endoplasmic reticulum stress-induced apoptosis. Neurosci Lett, 2013,546:57–62
Cheng F, Zhong X, Lu Y, et al. Refined qingkailing protects MCAO mice from endoplasmic reticulum stress-induced apoptosis with a broad time window. Evid Based Complement Alternat Med, 2012,12:567872
Ferri KF, Kroemer G. Organelle-specific initiation of cell death pathways. Nat Cell Biol, 2001,3:E255–E263
Rao RV, Peel A, Logvinova A, et al. Coupling endoplasmic reticulum stress to the cell death program: role of the ER chaperone GRP78. FEBS Lett, 2002,514(2–3):122–128
Broughton BR, Reutens DC, Sobey CG. Apoptotic mechanisms after cerebral ischemia. Stroke, 2006,40(5):e331–e339
Ryou MG, Liu R, Ren M, et al. Pyruvate Protects the Brain Against Ischemia-Reperfusion Injury by Activating the Erythropoietin Signaling Pathway. Stroke, 2012,43(4):1101–1110
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
This study was funded by Basic Research Projects in Shanxi Province (No. 2015021178) and Start-up Funds for Doctoral Research at Shanxi Datong University (No. 2014-B-01).
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Peng, Zf., Zhang, Nb., Meng, J. et al. Early Aerobic Exercise Promotes Neurological Function Recovery of Rats after Cerebral Ischemia/Reperfusion by Upregulating the Expression of Heat Shock Protein A5. CURR MED SCI 42, 267–273 (2022). https://doi.org/10.1007/s11596-022-2537-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-022-2537-0