Summary
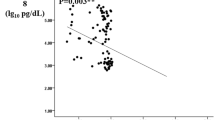
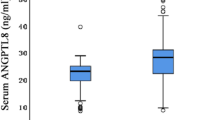
In various autoimmune diseases, Galecin-9 (Gal-9) has been shown to regulate the T-cell balance by decreasing Th1 and Th17, while increasing the number of regulatory T cells (Tregs). However, the role of Gal-9 in the patients with acute coronary syndrome (ACS) and chronic kidney disease (CKD) remains unclear. This study aims to measure the Gal-9 levels in serum and peripheral blood mononuclear cells (PBMCs) in patients with ACS plus CKD and examine their clinical implication. The serum levels of Gal-9 were determined by enzyme-linked immunosorbent assay (ELISA), the expression levels of Gal-9, Tim-3, and Foxp3 mRNA in PBMCs were detected by real-time reverse transcription-polymerase chain reaction (RT-PCR), and the expression of Gal-9 on the surface of PBMCs and in PBMCs was analyzed by flow cytometry. Furthermore, the correlation of serum Gal-9 levels with anthropometric and biochemical variables in patients with ACS plus CKD was analyzed. The lowest levels of Gal-9 in serum and PBMCs were found in the only ACS group, followed by the ACS+CKD group, and the normal coronary artery (NCA) group, respectively. Serum Gal-9 levels were increased along with the progression of glomerular filtration rate (GFR) categories of G1 to G4. Additionally, serum Gal-9 levels were negatively correlated with high-sensitivity C-reactive protein (hs-CRP), estimated GFR (eGFR), and lipoprotein(a), but positively with creatinine, age, osmotic pressure, and blood urea nitrogen (BUN). Notably, serum Gal-9 was independently associated with hs-CRP, osmotic pressure, and lipoprotein(a). Furthermore, serum Gal-9 levels were elevated in patients with type 2 diabetes (T2DM) and impaired glucose tolerance (IGT) in ACS group. It was suggested that the levels of Gal-9 in serum and PBMCs were decreased in patients with simple ACS and those with ACS plus CKD, and hs-CRP, eGFR, osmotic pressure and T2DM may have an influence on serum Gal-9 levels.
Similar content being viewed by others
References
Libby P. Inflammation in atherosclerosis. Arterioscler Thromb Vasc Biol, 2012,32(9):2045–2051
Hansson GK. Inflammation, atherosclerosis, and coronary artery disease. N Engl J Med, 2005,352:1685–1695
Hansson GK. Innate and Adaptive Immunity in the Pathogenesis of Atherosclerosis. Circ Res, 2002,91(4):281–291
Laurat E, Poirier B, Tupin E, et al. In vivo downregulation of T helper cell 1 immune responses reduces atherogenesis in apolipoprotein E-knockout mice. Circulation, 2001,104:197–202
Methe H, Brunner S, Wiegand D, et al. Enhanced T-helper-1 lymphocyte activation patterns in acute coronary syndromes. J Am Coll Cardiol, 2005,45(12):1939–1945
Cheng X, Liao YH, Ge H, et al. TH1/TH2 functional imbalance after acute myocardial infarction: coronary arterial inflammation or myocardial inflammation. J Clin Immunol, 2005,25(3):246–253
Cheng X, Yu X, Ding YJ, et al. The Th17/Treg imbalance in patients with acute coronary syndrome. Clin Immunol, 2008,127(1):89–97
Kishore U, Eggleton P, Reid KB, et al. Modular organization of carbohydrate recognition domains in animal lectins. Matrix Biology, 1997,15:583–592
Zhu C, Anderson AC, Schubart A, et al. The Tim-3 ligand galectin-9 negatively regulates T helper type 1 immunity. Nat Immunol, 2005,6(12):1245–1252
Kashio Y, Nakamura K, Abedin MJ, et al. Galectin-9 induces apoptosis through the calcium-calpain-caspase-1 pathway. J Immunol, 2003,170(7):3631–3636
Wang F, Xu J, Liao Y, et al. Tim-3 ligand galectin-9 reduces IL-17 level and accelerates Klebsiella pneumoniae infection. Cell Immunol, 2011,269(1):22–28
Oomizu S, Arikawa T, Niki T, et al. Galectin-9 suppresses Th17 cell development in an IL-2-dependent but Tim-3-independent manner. Clin Immunol, 2012,143(1):51–58
Bi S, Earl LA, Jacobs L, et al. Structural features of galectin-9 and galectin-1 that determine distinct T cell death pathways. J Biol Chem, 2008,283(18):12 248–12 258
Wang F, Wan L, Zhang C, et al. Tim-3-Galectin-9 pathway involves the suppression induced by CD4+CD25+ regulatory T cells. Immunobiology, 2009,214(5):342–349
Seki M, Oomizu S, Sakata KM, et al. Galectin-9 suppresses the generation of Th17, promotes the induction of regulatory T cells, and regulates experimental autoimmune arthritis. Clin Immunol, 2008,127(1):78–88
Chou FC, Shieh SJ, Sytwu HK. Attenuation of Th1 response through galectin-9 and T-cell Ig mucin 3 interaction inhibits autoimmune diabetes in NOD mice. Eur J Immunol, 2009,39(9):2403–2411
Kanzaki M, Wada J, Sugiyama K, et al. Galectin-9 and T cell immunoglobulin mucin-3 pathway is a therapeutic target for type 1 diabetes. Endocrinology, 2012,153(2):612–620
Leitner J, Rieger A, Pickl WF, et al. TIM-3 does not act as a receptor for galectin-9. PLoS Pathog, 2013,9(3):e1003253
Vaitaitis GM, Wagner DH, Jr. Galectin-9 controls CD40 signaling through a Tim-3 independent mechanism and redirects the cytokine profile of pathogenic T cells in autoimmunity. PLoS One, 2012,7(6):e38708
Su EW, Bi S, Kane LP. Galectin-9 regulates T helper cell function independently of Tim-3. Glycobiology, 2011,21(10):1258–1265
Foks AC, Ran IA, Wasserman L, et al. T-cell immunoglobulin and mucin domain 3 acts as a negative regulator of atherosclerosis. Arterioscler Thromb Vasc Biol, 2013,33(11):2558–2565
Kurose Y, Wada J, Kanzaki M, et al. Serum galectin-9 levels are elevated in the patients with type 2 diabetes and chronic kidney disease. BMC Nephrology, 2013,14:23
Zhu R, Liu C, Tang H, et al. Serum Galectin-9 Levels Are Associated with Coronary Artery Disease in Chinese Individuals. Mediators Inflamm, 2015,2015:457167
Levey AS, de Jong PE, Coresh J, et al. The definition, classification, and prognosis of chronic kidney disease: a KDIGO Controversies Conference report. Kidney Int, 2011,80(1):17–28
Matsuo S, Imai E, Horio M, et al. Revised equations for estimated GFR from serum creatinine in Japan. Am J Kidney Dis, 2009,53(6):982–992
Gotsman I, Grabie N, Gupta R, et al. Impaired regulatory T-cell response and enhanced atherosclerosis in the absence of inducible costimulatory molecule. Circulation, 2006,114(19):2047–2055
Mor A, Planer D, Luboshits G, et al. Role of naturally occurring CD4+ CD25+ regulatory T cells in experimental atherosclerosis. Arterioscler Thromb Vasc Biol, 2007,27(4):893–900
Xie JJ, Wang J, Tang TT, et al. The Th17/Treg functional imbalance during atherogenesis in ApoE(-/-) mice. Cytokine, 2010,49(2):185–193
Koguchi K, Anderson DE, Yang L, et al. Dysregulated T cell expression of TIM3 in multiple sclerosis. J Exp Med, 2006,203(6):1413–1418
Chabot SKY, Seki M, Shirato Y, et al. Regulation of galectin-9 expression and release in Jurkat T cell line cells. Glycobiology, 2002,12:111–118
Delacour D, Koch A, Jacob R. The role of galectins in protein trafficking. Traffic, 2009,10(10):1405–1413
Chirico WJ. C Protein release through nonlethal oncotic pores as an alternative nonclassical secretory pathway. BMC Cell Biol, 2011,12:46
Corson MA. Emerging inflammatory markers for assessing coronary heart disease risk. Current Cardiology Reports, 2009,11:452–459
Weber C, Noels H. Atherosclerosis: current pathogenesis and therapeutic options. Nat Med, 2011,17(11):1410–1422
Cheng XW, Kikuchi R, Ishii H, et al. Circulating cathepsin K as a potential novel biomarker of coronary artery disease. Atherosclerosis, 2013,228(1):211–216
Drakopoulou M, Toutouzas K, Stefanadi E, et al. Association of inflammatory markers with angiographic severity and extent of coronary artery disease. Atherosclerosis, 2009,206(2):335–339
Noren Hooten N, Ejiogu N, Zonderman AB, et al. Association of oxidative DNA damage and C-reactive protein in women at risk for cardiovascular disease. Arterioscler Thromb Vasc Biol, 2012,32(11):2776–2784
Arroyo-Espliguero R, Avanzas P, Cosin-Sales J, et al. C-reactive protein elevation and disease activity in patients with coronary artery disease. Eur Heart J, 2004,25(5):401–408
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare that there is no conflict of interests regarding the publication of this paper.
Additional information
This project was supported by grants from National Natural Science Foundation of China (No. 81270354) and Natural Science for Youth Foundation (No. 81300213).
Rights and permissions
About this article
Cite this article
Xie, Jh., Zhu, Rr., Zhao, L. et al. Down-regulation and Clinical Implication of Galectin-9 Levels in Patients with Acute Coronary Syndrome and Chronic Kidney Disease. CURR MED SCI 40, 662–670 (2020). https://doi.org/10.1007/s11596-020-2238-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-020-2238-5